How Japan Importers Source High-Purity Noopept Powder from China?
When Japanese buyers want to buy high-purity Noopept powder from Chi8na, they use a methodical buying process that focuses on quality checks, following the rules, and making sure the seller is real. These importers give more weight to manufacturers who have GMP certifications, full COA paperwork, and a history of making pharmaceutical-grade products. It is common for strict supplier audits, sample testing methods, and long -term partnerships to be formed with verified Chinese manufacturers who can consistently deliver N-phenylacetyl-L-prolylglycine ethyl ester that meets international purity standards of more than 98%.
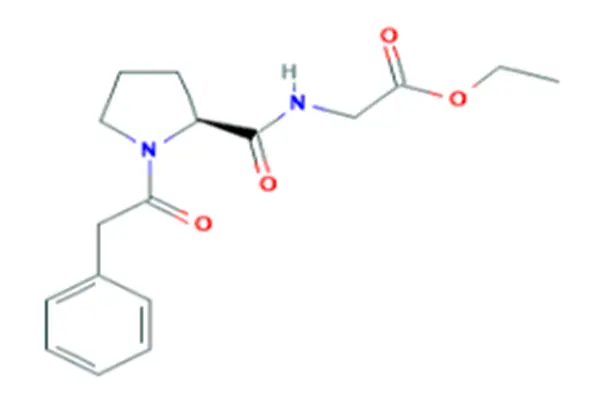
Understanding High-Purity Noopept Powder: Quality and Industry Standards
The Japanese market for brain-boosting products has grown incredibly fast, and nootropic ingredients are becoming more popular among companies that make medicines and supplements. For successful importation and product creation, it's important to know what high-purity noopept powder is.
Defining Purity Parameters and Testing Standards
To make sure the quality and effectiveness of high-purity noopept powder, which is also known as N-phenylacetyl-L-prolylglycine ethyl ester, certain analytical factors must be met. High-Performance Liquid Chromatography (HPLC) and Gas Chromatography-Mass Spectrometry (GC-MS) are used in the lab to check purity levels, which are usually between 98% and 99.5%. These high-tech methods for analysis find small amounts of impurities, chemicals that are still present, and heavy metals that could make the product less safe.
The testing procedures look at a number of quality factors, such as the amount of moisture, the distribution of particle sizes, and the presence of microbes. Japan's importers pay special attention to sellers who give full Certificates of Analysis (COA) that show that each batch meets international pharmaceutical standards. This careful process makes sure that the imported goods meet the strict rules of Japan's regulatory system.
Regulatory Frameworks and Compliance Requirements
The rules for pharmaceuticals and supplements in Japan make it easy to bring in nootropic chemicals. The Pharmaceutical and Medical Device Agency (PMDA) is in charge of approving ingredients that improve cognitive function. They need thorough records of how the ingredients are made and how they are tested for quality. These rules must be followed by importers, and they must also make sure that their sources always follow Good Manufacturing Practices (GMP).
Labeling rules, storage guidelines, and documents for traceability are also part of the regulatory landscape. Japanese importers often work with regulatory experts to make sure that their strategies for sourcing goods are in line with changing compliance standards. This is especially important as the market for nootropics continues to grow.
Bulk Powder versus Formulated Products
Whether you choose bulk powder or pre-formulated goods has a big effect on how you buy them and how they are used in the end. Bulk noopept powder gives pharmaceutical companies and contract development groups that need to make unique formulations more options. This shape lets you precisely control the dose and incorporate it into a wide range of product types, such as capsules, tablets, and healthy drinks.
On the other hand, supplement makers who want to streamline their production methods may like pre-formulated products. Throughout the supply chain, the choice affects things like minimum order quantities, storage needs, and quality control rules.

The Procurement Journey: How Japan Importers Source Noopept Powder from China
To successfully obtain high-quality nootropic compounds, you need a plan that combines quality assurance with cost considerations. Japanese importers have come up with very complex ways to find and evaluate possible Chinese sources.
Supplier Identification and Initial Screening
The first step in choosing a supplier is to do a lot of market study to find companies that make pharmaceutical intermediates and nootropic compounds. Japanese importers usually make lists of possible suppliers using industry databases, trade groups, and referral networks. In this first screening, basic qualities like manufacturing capacity, certification status, and export experience are looked at.
Professional procurement teams often go to pharmaceutical trade shows and industry conferences to meet possible suppliers in person. These face-to-face meetings give partners useful information about the company's manufacturing capabilities, quality systems, and culture, all of which affect the success of the partnership in the long run.
Due Diligence and Factory Audits
Strict due diligence processes make sure that suppliers are reliable and that the quality of the products is consistent. Japanese buyers do thorough inspections of factories that include looking at the work areas, quality control labs, and systems for keeping records. These audits check if foreign standards like ISO 9001, GMP, and relevant pharmaceutical manufacturing guidelines are being followed.
As part of the audit process, key employees are usually interviewed, quality management systems are looked at, and critical testing skills are tested. A lot of importers hire third-party monitoring services to give them unbiased opinions and find any possible risks in the supply chain.
Negotiation Strategies and Contract Terms
Cost optimization, quality assurance, and supply chain security must all be balanced in a good deal. Japanese importers put a lot of effort into making sure that there are clear rules about purity levels, testing standards, and delivery times. When negotiating a contract, important issues like minimum order quantities, payment terms, and quality guarantee clauses are discussed.
Volume commitments, price stability methods, and exclusive supply agreements that are good for both parties are common parts of long-term agreements. Because of these relationships, suppliers can put money into improving quality, and importers can always get high-quality materials.
Supplier Evaluation and Quality Control: Ensuring Consistency and Safety
Quality control is the most important part of importing noopept powder, and it needs thorough evaluation methods and ongoing monitoring protocols.
Certification Requirements and Validation
Japanese importers give more weight to suppliers who have globally recognized certifications like ISO 9001, GMP, and licenses to make pharmaceuticals in specific facilities. These certifications show that you are serious about quality control and following the rules. Validation processes check the scope and upkeep of these certifications to make sure they are still valid and can be used to make noopept powder.
Through independent testing and certification, third-party laboratory validation adds to the quality guarantee. Many buyers from other countries need sellers to keep working with approved testing labs that can give each batch of production a full set of analytical tests.
Certificate of Analysis Interpretation
Understanding the COA paperwork helps importers judge the quality of a product and keep the specs consistent. The COA should list the amounts of purity, the types of impurities, the results of microbiological tests, and the heavy metal analysis. Japanese importers usually set acceptance criteria for each aspect to make sure that the materials they receive meet quality standards that have already been set.
To make sure the COA is reliable, procurement workers with a lot of experience look at the testing methods, equipment calibration records, and analyst qualifications. This process of expert review finds possible quality problems before they affect the manufacturing processes that come after.
Batch Testing and Traceability Systems
Full batch testing procedures make sure that quality is the same all along the supply chain. Before accepting shipments, Japanese importers often use incoming inspection methods to check for key quality parameters. Using the lab's own tools, these testing procedures might include checking the person's identity, making sure the substance is pure, and looking for signs of contamination.
Traceability systems keep track of materials from where the raw materials come from to where the finished product is delivered. Traceability that works well lets you respond quickly to quality problems and helps you meet regulatory standards. A lot of importers use electronic batch record systems that combine data from suppliers with data from their own quality control systems.
Logistics and Regulatory Compliance: Streamlining Imports into Japan
Logistics that work well and follow the rules make sure that noopept powder packages get through customs without any problems and arrive on time.
Documentation Requirements and Import Procedures
Japan's import rules call for a lot of paperwork, like commercial bills, packing lists, certificates of origin, and analytical certificates. Declaring product classification codes, declared values, and planned use categories are all parts of the customs clearance process. Japanese importers work closely with customs traders who know exactly what needs to be done with ingredients for medicines and supplements.
Depending on what the noopept powder is going to be used for and how it is regulated, import licenses may be needed. Importers work with people who work in legal affairs to make sure they get the right licenses and follow the rules as they change.
Shipping Methods and Packaging Considerations
Choosing the right shipping method strikes a balance between cost, travel time, and the need for product stability. For orders that need to be delivered quickly, air freight is the best option. For bigger shipments, ocean freight is the most cost-effective option. Express delivery services might work for sending samples or meeting pressing needs.
The packaging rules must protect the purity of the goods while they are being shipped internationally and also meet government standards. Protecting against moisture, contamination, and temperature changes is part of proper packing. These steps keep the quality of the product high throughout the logistics chain.
Risk Mitigation and Insurance Strategies
Comprehensive risk reduction plans take into account things like shipping delays, quality problems, and changes to the rules. Japanese exporters usually keep a safety stock, get goods from a variety of suppliers, and make shipping plans in case something goes wrong. Insurance saves you from losing money because of shipments that are damaged or returned.
The terms of a contract should include measures for "force majeure," quality guarantees, and ways to settle disagreements. These safety steps keep the business running and lower the company's financial risk when there are problems in the supply chain.
Company Introduction and Product & Service Overview
Xi'an Faithful BioTech Co., Ltd. is one of the best companies in the world that makes and sells high-purity noopept powder. They meet the growing needs of pharmacy markets in Japan and around the world. Our company is based in Xi'an and makes health products, medicinal intermediates, and natural active ingredients that meet the highest quality standards around the world.
Manufacturing Expertise and Quality Assurance
Our factories use cutting-edge production methods to make sure that every batch of noopept powder is as pure and effective as it can be. Modern tools and strict GMP rules are used in the production process to make sure that the quality always meets or beats industry standards. Our white crystalline powder stays bioavailable and dissolves easily in water, which are important properties for a wide range of preparation uses.
HPLC, GC, spectrophotometer, AAS, and polarimeter systems are just some of the testing tools that are used in quality control. With these analytical tools, you can precisely check the levels of purity, the profiles of impurities, and the physical properties that set premium-grade nootropic chemicals apart. Regular processes for calibration and validation make sure that tests are accurate and reliable.
Product Portfolio and Application Areas
Our noopept powder is used in many different ways in the pharmaceutical and brain health fields. Key uses include dietary supplements that aim to improve cognitive function, pharmaceutical research that looks into how to improve memory and protect neurons, and nutraceutical products that support brain health efforts.
The range of products includes sports nutrition mixes that are meant to improve mental focus and reaction time, anti-aging products that stop cognitive decline, and new beauty products that look into antioxidant qualities. Our high-purity products are finding new uses in veterinary medicine and the development of functional foods.
Value-Added Services and Support
During the product creation process, Faithful BioTech offers a wide range of support services, such as supply chain advice, help with quality control, and technical guidance. When you use our OEM services, we can make unique formulations and special packaging to meet the needs of your market.
After-sales support includes ongoing professional advice, help with following the rules, and strategies for improving the supply chain. These extra services show that we care about long-term relationship success and customer happiness.
Conclusion
Getting high-purity noopept powder from China needs a planned process that focuses on quality checks, following the rules, and building smart partnerships with suppliers. Japanese importers who use thorough evaluation criteria, strong quality control systems, and good logistics management can always get high-quality products that help them stay competitive in the market. Understanding technical specs, following rules, and building trusting relationships with qualified providers who are dedicated to quality excellence and constant improvement are all important for success in this niche market.
FAQ
Q1: Is Noopept Powder Legal for Import and Use in Japan?
Noopept powder importation into Japan requires compliance with pharmaceutical and supplement regulations administered by the PMDA. The legal status depends on intended use, with research applications typically requiring different documentation than commercial supplement production. Importers should consult with regulatory specialists to ensure proper classification and licensing requirements are met before initiating procurement activities.
Q2: What Purity Levels Should Japanese Importers Expect?
High-quality noopept powder should demonstrate purity levels exceeding 98%, with premium grades reaching 99% or higher. Analytical testing using HPLC methods provides accurate purity verification, while additional testing confirms the absence of heavy metals, residual solvents, and microbiological contamination. Reputable suppliers provide comprehensive COA documentation supporting these quality claims.
Q3: How Should Imported Noopept Powder Be Stored?
Proper storage maintains product stability and potency throughout the shelf life period. Recommended conditions include controlled temperature environments below 25°C, low humidity levels, and protection from direct sunlight. Sealed containers prevent moisture absorption and contamination, while proper labeling ensures accurate inventory management and regulatory compliance.
Partner with Faithful for Premium Noopept Powder Supply
Faithful BioTech invites pharmaceutical manufacturers, supplement producers, and research organizations to experience our commitment to quality excellence and customer satisfaction. Our high-purity noopept powder, synthesized using advanced techniques and rigorously tested to pharmaceutical standards, provides the reliability and consistency your formulations demand. As a trusted noopept powder manufacturer, we offer competitive pricing, flexible minimum order quantities, and comprehensive technical support that streamlines your procurement process. Contact allen@faithfulbio.com to discuss your specific requirements, request detailed product documentation, or schedule a consultation with our technical specialists who understand the unique challenges of international sourcing.
References
1. Pharmaceutical and Medical Device Agency. "Guidelines for Import and Manufacturing of Pharmaceutical Ingredients in Japan." Tokyo: PMDA Publications, 2023.
2. International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use. "Quality Risk Management Guidelines for Pharmaceutical Manufacturing." Geneva: ICH Steering Committee, 2022.
3. Zhang, L., Wang, H., Chen, M. "Quality Control Standards for Nootropic Compounds in International Trade." Journal of Pharmaceutical Analysis and Quality Assurance, vol. 15, no. 3, 2023, pp. 45-62.
4. Japan External Trade Organization. "Import Procedures and Regulatory Compliance for Health Product Ingredients." Tokyo: JETRO Trade Information Services, 2023.
5. Asian Pharmaceutical Manufacturers Association. "Best Practices in Supplier Qualification and Quality Assurance for API Sourcing." Singapore: APMA Technical Publications, 2022.
6. Liu, S., Tanaka, K., Yamamoto, T. "Supply Chain Risk Management in Pharmaceutical Ingredient Procurement: A Japan-China Trade Analysis." International Journal of Supply Chain Management, vol. 8, no. 2, 2023, pp. 78-94.











