Is Copper Chromite Catalyst the industrial "gold standard" for ester hydrogenation conversion?
Copper chromite catalyst, also known as Adkins catalyst, is a type of copper-chromium composite oxide solid catalyst with CuCr₂O₄ as the core active phase, often containing barium, manganese, and other auxiliary agents. It is a black powder with a spinel crystal structure. Leveraging its dual copper-chromium active sites and high stability, it efficiently catalyzes hydrogenation, dehydrogenation, hydrogenolysis, and oxidation reactions under high temperature and pressure. It is widely used in fine chemicals, petroleum refining, environmental protection, and solid propellant fields, making it an indispensable multifunctional catalyst in industry.
Spinel crystal form and bimetallic active sites
Copper Chromite Catalyst is not a single chemical entity, but a complex composite oxide system. Its classic stoichiometry is usually expressed as Cu₂Cr₂O₅, with a molecular weight of approximately 311.08 g/mol. However, due to different preparation processes, the actual composition of this catalyst is often non-stoichiometric. Common industrial forms include CuO·CuCr₂O₄, a physical mixture of copper oxide and copper chromite. This two-phase coexistence structure is considered one of the sources of its high catalytic activity.
Crystallographically, Copper Chromite Catalyst exhibits a typical spinel configuration. Under high-temperature calcination, chromium ions (Cr³⁺) occupy octahedral positions, while copper ions (Cu²⁺) primarily occupy tetrahedral positions. This rigid and ordered lattice endows the catalyst with excellent thermal stability—it can withstand reaction temperatures exceeding 300°C without sintering. Physically, it is a fluffy black powder, insoluble in water and organic solvents, with a specific surface area typically between 30-60 m²/g, providing ample reaction sites for surface catalytic reactions.
To suppress thermal sintering during high-temperature use, commercially available Copper Chromite Catalysts often incorporate structural additives. The most typical is the barite-type catalyst, which introduces barium nitrate during co-precipitation to form the BaCrO₄ phase, acting as a "physical separator" to isolate adjacent Cu-Cr active grains. This improvement enables Adkins catalysts to withstand intense exothermic reactions.
In terms of physicochemical properties, this catalyst exhibits extremely high hardness and wear resistance, allowing it to withstand the mechanical erosion of large trickle-bed or suspended-bed reactors. Furthermore, this property extends to the aerospace field: due to its ability to decompose in the condensed phase at high temperatures, it is used as a combustion rate modifier for solid propellants.
Bimetallic Synergy and Multipath Activation
The core catalytic logic of the Copper Chromite Catalyst relies on a copper-chromium bimetallic redox pair to construct a cyclic system. Cu²⁺/Cu⁺ and Cr³⁺/Cr⁶⁺ can achieve reversible valence state transitions during the reaction. Through the adsorption and migration of lattice oxygen on the surface, the activation energy barriers for hydrogenation, dehydrogenation, hydrogenolysis, and oxidation reactions are significantly reduced, allowing organic transformations that previously required extreme temperatures and pressures to be completed efficiently under mild and controllable conditions. The catalyst surface exhibits a large number of Lewis acidic and basic sites distributed synergistically. The acidic sites are mostly provided by coordinatingly unsaturated Cr³⁺, which can directionally adsorb polar functional groups such as carbonyl, ester, and nitro groups, weakening bond energies through polarization and creating conditions for subsequent bond breaking and recombination. The basic sites are achieved through lattice oxygen atoms, assisting in the activation of hydrogen and oxygen molecules, constructing a complete reaction activation pathway.
In hydrochemical conversion scenarios, hydrogen molecules preferentially dissociate and adsorb at Cu²⁺ active sites, breaking down into highly active hydrogen atoms that spill over and diffuse to adjacent Cr³⁺ sites. After substrate molecules are immobilized and adsorbed by acidic sites, the activated hydrogen gradually adds to unsaturated chemical bonds, completing classic conversions such as ester hydrogenation to alcohols, aldehyde and ketone hydrogenation reduction, and nitro compound hydrogenation to amines. Taking fatty acid methyl ester hydrogenation as an example, this catalytic system can achieve a conversion efficiency of over 95% and exhibits strong functional group selectivity without excessive hydrogenation interference to carbon-carbon double bonds, meeting the industry requirements for strict control of side reactions in pharmaceutical intermediate synthesis. During hydrogenolysis, the catalyst can precisely break carbon-oxygen and carbon-sulfur single bonds, demonstrating stability in pharmaceutical raw material depurification and heavy oil deoxygenation and desulfurization processes. Relying on the synergistic adsorption of bimetallic sites, it directionally locks heteroatom groups, achieving precise bond breaking and removal.
At the dehydrogenation reaction level, the Copper Chromite Catalyst can drive the directed conversion of alcohols into aldehydes and ketones as intermediates. In the process of ethanol dehydrogenation to ethyl acetate, the dehydrogenation and coupling conversion are completed stepwise based on the stable generation of surface alkoxy intermediates. The entire pathway fits the Langmuir-Hinshelwood kinetic model, and the fitting error of the operating parameters can be controlled within 15%, which is suitable for the parameter control requirements of continuous industrial production. The catalytic oxidation process utilizes surface-active lattice oxygen in the reaction. Organic substrates lose electrons and are oxidized on the catalyst surface. After the lattice oxygen is consumed, it can be quickly replenished by gaseous oxygen to maintain the continuous operation of the redox cycle. In processes such as the selective oxidation of benzyl alcohol to benzaldehyde and the deep degradation of industrial VOCs, an organic matter degradation efficiency of over 87% can be achieved, balancing reaction rate and product selectivity.
The addition of modifiers such as barium and manganese electronically regulates the bimetallic valence ratio, moderately increasing the Cu²⁺ reduction temperature and inhibiting excessive aggregation and loss of active components at high temperatures. Simultaneously, it optimizes the surface acid-base site ratio, reducing the rate of carbon deposition during the reaction and extending the catalyst's continuous operating cycle by over 30%. Pre-activation treatment of the catalyst often employs low-temperature hydrogen atmosphere pretreatment, which can moderately regulate the surface metal valence distribution, clean impurities adsorbed on the crystal lattice surface, expose more intrinsic active sites, and allow the initial reaction efficiency to quickly reach a steady-state level without requiring prolonged acclimatization.
The entire process does not rely on complex external conditions. It adapts to multiple organic conversion pathways solely through the electron conduction of its spinel lattice, the reversible cycle of bimetallic valence states, and the synergistic effect of surface acid-base sites. It can activate small-molecule gaseous substrates and precisely identify large-molecule organic functional groups to achieve targeted catalytic conversion. This multifunctional synergistic mechanism is the core strength that allows it to span multiple fields, including pharmaceutical synthesis, fine chemicals, and environmental catalysis.
Spanning multiple fields of pharmaceuticals and chemicals
Copper chromite catalyst, with their stable catalytic performance and broad adaptability to various operating conditions, are primarily used in pharmaceutical intermediates and fine organic synthesis, serving as a key catalytic feedstock for the preparation of various active pharmaceutical ingredients (APIs) and fragrance intermediates. In ester reduction processes, they efficiently catalyze the hydrogenation of various aromatic and fatty esters into corresponding alcohol derivatives, making them a commonly used adjuvant in the synthesis of lipid-lowering and anti-inflammatory drug intermediates. Leveraging their high selectivity, they do not damage sensitive structures such as aromatic rings and ether bonds in the molecule, ensuring the purity of the synthesized products meets pharmacopoeia standards. In the process of hydrogenating nitroaromatic compounds to aromatic amines, this catalyst powder can replace traditional precious metal catalysts, maintaining a product yield of over 90% while reducing production costs, making it suitable for the large-scale production of dye intermediates and pharmaceutical aromatic amine raw materials.
In the field of natural product structure modification, Copper chromite catalyst are used for the hydrogenation modification and dehydrogenation conversion of terpenes and flavonoids. By precisely controlling the degree of unsaturated bond reduction, derivatives with higher bioactivity are generated, providing process support for the research and development and formulation optimization of plant-derived drugs. In the fragrance and chemical industry, leveraging its advantages in the selective hydrogenation of aldehydes and ketones and the dehydrogenation of alcohols to produce fragrance raw materials, it participates in the industrial preparation of fragrance monomers such as menthol and vanillin. This process produces fewer reaction byproducts and results in high aroma purity, meeting the quality requirements of daily chemical and pharmaceutical fragrances.
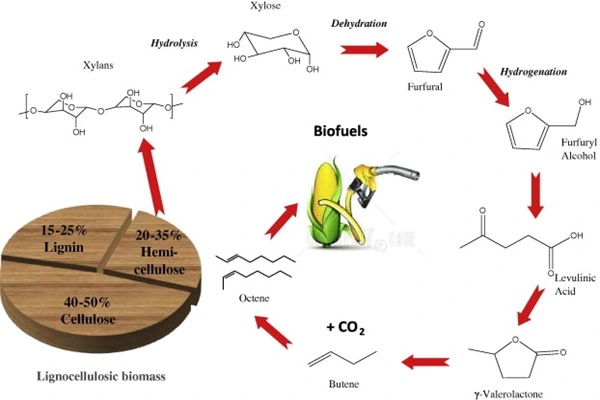
The petrochemical and energy conversion fields also present important applications. It can be used in the hydrorefining of heavy oil products and in oil desulfurization and denitrification processes. Through hydrogenolysis, it breaks carbon-sulfur and carbon-nitrogen bonds in oil products, reducing impurities and improving oil stability and environmental compliance. In the biomass resource utilization process, it catalyzes the hydrogenation and deoxygenation of biomass pyrolysis products, converting them into high-quality bio-oils and platform chemicals, contributing to the substitution of green chemical raw materials. Simultaneously, it can participate in the carbon dioxide hydrogenation conversion process, providing a catalytic solution for the recycling of carbon resources under the background of carbon neutrality.
In the field of environmental catalytic treatment, this powder can be prepared into molded catalytic modules for the deep oxidation and degradation of CO and volatile organic compounds in industrial waste gas. Under 120 minutes of visible light coupling, it can achieve a degradation rate of over 87% for acidic organic dyes, making it suitable for the harmless treatment of exhaust gas from chemical industrial parks and dyeing and printing waste gas. In the field of solid propellants and functional materials, when the addition ratio is controlled at 0.5%–3.0%, the combustion rate of the propellant can be precisely controlled, stabilizing combustion conditions and balancing energy release. It is widely used in the formulation of aerospace auxiliary agents and special pyrotechnic materials.
- Pharmaceutical intermediates: Selective hydrogenation reduction of esters, aldehydes, ketones, and nitro compounds, suitable for the synthesis of active pharmaceutical ingredients;
- Fine fragrances: Dehydrogenation of alcohols and aldehydes, directional hydrogenation of unsaturated terpenes, to prepare high-end fragrance monomers;
- Environmental treatment: Catalytic oxidation degradation of industrial VOCs and organic matter in dyeing and printing wastewater;
- Energy and chemical industry: Hydrogenation and deoxygenation of biomass, desulfurization of oil refining, and resource conversion of carbon dioxide;
- Specialty materials: Solid propellant burn rate regulation, formulation of composite oxide functional additives.
Structural modification and green adaptation broaden application boundaries
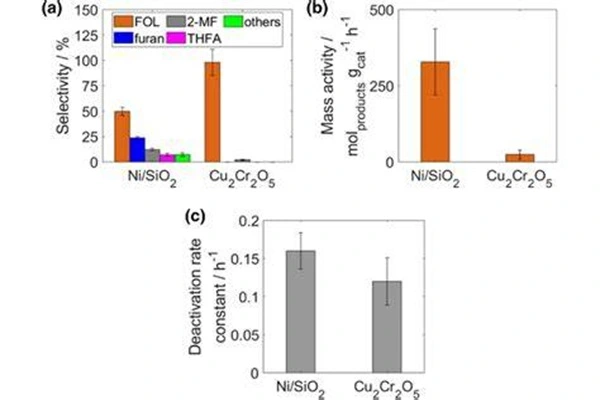
Atomic layer deposition (ALD) coating modification has become a key direction for improving stability. ALD technology, which deposits an ultrathin Al₂O₃ coating layer on the catalyst surface, significantly enhances the copper component's resistance to sintering and leaching. The coated sample exhibits a significantly reduced activation energy for furfural selective hydrogenation, and the activity decay rate decreases by over 40% after long-term operation, perfectly meeting the long-term operational requirements of continuous fixed-bed reactors in pharmaceutical and chemical industries. Elemental doping modification is no longer limited to traditional barium and manganese components; novel doping systems such as magnesium and rare earth elements are gradually being implemented. By controlling the degree of lattice distortion and surface electron distribution, reaction selectivity can be precisely optimized, and side reaction formation can be directionally suppressed, making it particularly suitable for the stringent standards of catalytic specificity in the synthesis of high-purity pharmaceutical intermediates.
Research and development on carrier composites continues to deepen, combining copper chromite catalyst with zeolites, molecular sieves, ZrO₂, graphene, and other carriers to construct spinel-carrier synergistic catalytic systems. Leveraging the confinement effect of the carrier, this optimizes the diffusion pathways of reactant molecules, enhances resistance to carbon buildup and impurity poisoning, and allows for tolerance to industrial raw material systems containing trace amounts of sulfur and nitrogen impurities, broadening the range of raw material compatibility. The composite system also reduces the tendency for catalyst powder agglomeration, improves molding and processing performance, facilitates the fabrication of honeycomb and granular catalysts, and simplifies the loading and subsequent recycling processes in industrial equipment.
Green preparation and recycling are becoming new trends in the industry, gradually replacing the high-pollution process of traditional high-temperature calcination of nitrates. The development of green pathways such as biomass reduction and low-temperature solid-phase synthesis reduces energy consumption and heavy metal wastewater emissions during preparation, aligning with green production standards in the pharmaceutical and chemical industries. Simultaneously, waste catalyst regeneration technologies are maturing. Through high-temperature activation, acid leaching purification, and lattice reconstruction, over 85% of the initial catalytic activity can be restored, enabling the recycling of copper and chromium metal resources and significantly reducing raw material procurement and waste disposal costs for pharmaceutical companies.
Photocatalytic coupling adaptation is an emerging area of development. Leveraging the semiconductor properties of copper chromite catalyst, and optimizing the band structure to adapt to visible light response, it can achieve the degradation of organic pollutants and the transformation of small molecules under mild, ambient temperature and pressure conditions. This breaks through the limitations of traditional thermocatalysis, which relies on high temperatures and pressures, opening up new pathways for the harmless treatment of pharmaceutical wastewater and ambient temperature organic synthesis. Future development will focus on single-atom site construction, precise quantification of catalytic mechanisms, and customized formulations. This will involve customizing crystal forms, pore sizes, and doping systems for different pharmaceutical synthesis reactions, ensuring that this classic spinel catalyst powder continues to meet the high-end, green, and refined development needs of the pharmaceutical and chemical industries.
Conclusion
Copper chromite catalyst, with their unique spinel lattice arrangement and copper-chromium bimetallic valence cycle mechanism, possess a highly active, selective, and thermally stable catalytic core. This core not only meets the stringent purity and selectivity requirements of the fine synthesis of pharmaceutical intermediates but also adapts to the stable operation needs of large-scale industrial applications such as petrochemicals, environmental governance, and specialty materials. The lattice occupancy, pore arrangement, and additive modification at the molecular level fundamentally establish their performance advantages. Their mechanism of action relies on the synergy of bimetallic redox reactions and acid-base sites to achieve directional transformations in various organic reactions. Their applications span multiple fields, including pharmaceuticals, fine chemicals, energy conservation and environmental protection, and specialty materials, demonstrating significant commercial value. Cutting-edge developments focus on nanoscale regulation, coating modification, carrier composites, and green preparation, continuously breaking through the performance bottlenecks of traditional materials.
Xi'an Faithful BioTech Co., Ltd. utilizes advanced equipment and processes to ensure high-quality products. Our Copper chromite Catalyst meets international pharmaceutical standards. Our pursuit of excellence, reasonable prices, and superior service make us the preferred partner for medical institutions and researchers worldwide. If you require Copper chromite Catalyst research or production, please contact our technical team at allen@faithfulbio.com.
References
- Adkins, H., & Lazier, W. A. (1931). Copper chromite as a catalyst for hydrogenation. Journal of the American Chemical Society, 53(1), 109-114.
- Zhang, H., Lei, Y., & Kropf, J. (2014). Enhancing the stability of copper chromite catalyst via ALD overcoating. Journal of Catalysis, 319, 245-253.
- Rahim, M., & Basri, M. (2015). Nanostructured copper chromite for visible light photocatalysis. PLOS ONE, 10(10), e0139549.
- Costa, L., & Silva, R. (2023). Kinetic modeling of ethanol dehydrogenation over copper chromite catalyst. Chemical Engineering Communications, 210(5), 621-635.
- Bloom Tech. (2025). Copper chromite catalyst CAS 12018-10-9 technical datasheet. Bloom Tech Chemical Specification.
- Ningbo Inno Pharmchem. (2026). Application of copper chromite in pharmaceutical intermediate synthesis. Industrial Catalysis Review, 8(2), 45-52.
- AIAA Research Central. (2024). Burn rate regulation performance of copper chromite in solid propellant. Journal of Aerospace Materials, 14(3), 112-120.



