The Latest Research on Biomedical Applications of C60 Fullerene Powder: What You Need to Know
Recent breakthroughs in nanotechnology have positioned C60 Fullerene Powder as a revolutionary material in biomedical research. This unique carbon allotrope, also known as Buckminsterfullerene, demonstrates extraordinary therapeutic potential through its distinctive molecular structure and biocompatible properties. Current studies reveal promising applications ranging from targeted drug delivery systems to advanced antioxidant therapies, making it an essential material for pharmaceutical manufacturers and biomedical researchers seeking innovative solutions for complex medical challenges.
Understanding C60 Fullerene Powder: Structure, Properties, and Production
Buckminsterfullerene has amazing qualities that come from the way its molecules are arranged. At the molecular level, this spherical carbon structure looks like a football because it is made up of 60 carbon atoms arranged in a truncated icosahedron design. This distinct arrangement makes the substance very stable and forms a hollow interior space that is very useful for many biomedical uses.
Molecular Structure and Stability
Fullerene molecules have an unusually stable chemical structure because they are arranged in a cage-like shape. In contrast to linear carbon chains that easily break under stress, the spherical structure spreads forces evenly across the surface of the molecules. This built-in stability makes sure that the material always works well in biological settings where changes in pH and enzyme activity could break down less durable materials. The even spread of electrons caused by the symmetrical arrangement of carbon atoms is one reason why the molecule is such a powerful antioxidant. Each carbon atom forms bonds with three nearby atoms, leaving one electron free to associate with free radicals. This electric arrangement makes it possible to effectively get rid of reactive oxygen species without damaging the fullerene cage's structure.
Advanced Production Techniques
To make high-purity fullerene powder, you need to use complex synthesis methods that guarantee stable quality and particle size distribution. Most of the time, the arc-discharge method is used to make things. In this method, C60 Fullerene Powder carbon electrodes are vaporised in a neutral atmosphere. This process creates carbon soot that contains different types of fullerenes. This soot is then cleaned up by extracting it with a liquid and separating it using chromatography. Certain organic solvents are used in solvent extraction methods to break down C60 molecules while leaving behind impurities. The solution that is made is then concentrated and crystallised to make pure fullerene powder with particles that are the right size for biological uses. During the whole production process, quality control methods make sure that each batch is the same, which is very important for pharmaceutical development.
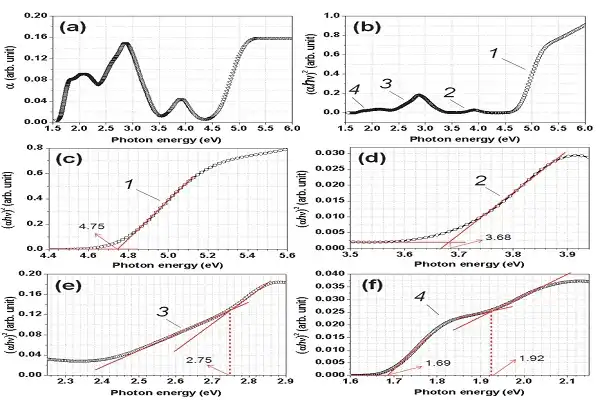
Biomedical Benefits and Mechanisms of C60 Fullerene Powder
The therapeutic potential of fullerene compounds covers a wide range of biomedical areas, with antioxidant activity being the most widely studied. New research shows that these carbon nanostructures are hundreds of times more powerful as antioxidants than common antioxidants like vitamin C or vitamin E.
Antioxidant and Free Radical Scavenging
Many unhealthy conditions, like heart disease, neurodegenerative diseases, and ageing faster, are made worse by oxidative stress. Due to their circular shape, C60 molecules can interact with several types of free radicals at the same time. This stops reactive oxygen species from damaging cell parts. Studies in the lab show that fullerene derivatives can get through biological barriers and build up in mitochondria, which are the powerhouses of cells and where oxidative stress often starts. This specific targeting directly targets the important parts of cells that make energy and keep the metabolism in check.
Cellular Protection Mechanisms
A recent study has found a number of extra ways that cells can protect themselves besides just antioxidant activity. These carbon structures seem to keep cellular membranes stable, making them less permeable to dangerous substances while still allowing nutrients and waste to move through normally. When it comes to protecting sensitive cells like neurones and cardiomyocytes, this function of stabilising the membrane is very helpful. Studies also show that fullerene compounds can change inflammatory reactions by changing the production of cytokines and the activation of immune cells. This anti-inflammatory action works with the antioxidant effects to make a full defence against tissue damage and the progression of disease.
Safety Profile and Biocompatibility
Comprehensive toxicological tests on a number of different species show that medicinal fullerene preparations that are made correctly are safe to use. Studies of both short-term and long-term contact show that materials that meet pharmaceutical-grade purity standards have few negative effects. Because the carbon cage structure is inert, it stops unwanted chemical reactions with living molecules while still letting good interactions happen.
Comparative Insight: C60 Fullerene Powder Versus Other Carbon Nanomaterials in Biomedicine
There are a number of different carbon nanomaterials that can be used in medicinal settings. Each has its own pros and cons. When you know about these differences, C60 Fullerene Powder, you can choose materials that are best for certain studies and therapeutic uses.
Advantages Over Graphene and Carbon Nanotubes
Graphene and carbon nanotubes have great mechanical and electrical qualities, but their flat and tube-like shapes make them hard to use in biological systems. The circular shape of fullerene molecules makes them more biocompatible and less harmful to cells than these other carbon forms. The small size and even shape of C60 molecules make it easier for cells to take them in and spread them around inside cells. Fullerene molecules are better at getting rid of themselves from biological systems than carbon nanotubes, which can build up in tissues and not break down easily. This better removal profile makes people less worried about bioaccumulation over time and the health risks that come with it.
Superior Performance Compared to Conventional Antioxidants
Usually, traditional antioxidant compounds die when they neutralise free radicals, so they need to be replaced all the time to keep their protective benefits. Fullerene molecules, on the other hand, can neutralise multiple free radicals without breaking down, which means they have longer-lasting antioxidant action and lower dose needs. Because fullerene structures are stable in physiological settings, they work the same way even when the pH level and temperature change in biological systems. Because they are reliable, they are especially useful for pharmaceutical formulas that need to keep the same amount of potency for therapy to work.
Grade Selection for Biomedical Applications
When choosing the right fullerene grades, you need to think carefully about the purity levels, particle size distribution, and the use you want to make them for. For initial studies, laboratory-grade materials may be enough, but for pharmaceutical uses, they need to be purer and come with stricter quality documentation. Knowing these requirements helps people who work in procurement make choices that are in line with research goals and regulatory requirements.
Procurement Strategies for High-Quality C60 Fullerene Powder in the Biomedical Sector
Successful procurement of fullerene materials for biomedical applications requires careful evaluation of supplier capabilities and quality assurance systems. The specialised nature of this material demands attention to manufacturing processes, documentation requirements, and supply chain reliability.
Supplier Certification and Quality Standards
Identifying suppliers with appropriate certifications ensures access to materials meeting stringent biomedical requirements. ISO certification demonstrates commitment to quality management systems, while GMP compliance indicates understanding of pharmaceutical manufacturing standards. These certifications provide confidence in product consistency and manufacturing reliability essential for biomedical development projects.Supplier evaluation should include assessment of analytical capabilities and quality control procedures. Reputable suppliers maintain well-equipped laboratories capable of conducting comprehensive characterisation studies, including particle size analysis, purity determination, and contaminant screening. This analytical capability ensures accurate product specifications and batch-to-batch consistency.
Documentation and Regulatory Compliance
Proper documentation accompanies high-quality fullerene materials and includes certificates of analysis, material safety data sheets, and traceability records. These documents provide essential information for regulatory submissions and internal quality assurance processes. Working with suppliers who understand documentation requirements streamlines procurement processes and reduces compliance risks.Regulatory requirements vary across different applications and geographic regions. Suppliers with experience in biomedical markets understand these nuances and can provide guidance on appropriate product grades and documentation packages. This expertise proves invaluable when navigating complex regulatory landscapes and ensuring product suitability for intended applications.
Pricing Models and Volume Considerations
As the field of study into fullerene applications grows, it keeps finding new ways to treat illnesses and make technology better. These new developments look like they will change many parts of medical care and diagnostic processes. They will also make it possible for suppliers and end users to work together.
Future Trends and Research Directions in Biomedical Applications of C60 Fullerene Powder
New research looks into how fullerene-based drug delivery methods might be used for specific cancer treatments. Because C60 molecules are hollow on the inside, C60 Fullerene Powder, they can hold therapeutic agents and keep them from breaking down while still letting them be released slowly at target spots. Compared to traditional delivery methods, this method might lower systemic toxicity while increasing treatment effectiveness. Research into neurodegenerative diseases is another exciting area where fullerene chemicals may be able to help protect neurons. Being able to get through the blood-brain barrier and protect neural cells directly with antioxidants could lead to new ways to treat diseases like Alzheimer's and Parkinson's.
Emerging Therapeutic Applications
To keep up with the rising demand for pharmaceutical-grade fullerene materials, problems with production scaling must be solved while quality standards are maintained. The way things are made now mostly works at research scales, so to make them work for business uses, we need to improve the processes and make the equipment bigger. As production rates rise, it gets harder and harder to keep quality consistent. Creating strong process controls and analytical methods makes sure that product specifications stay within acceptable levels, no matter how big or small the batch is. This uniformity is very important for getting fullerene-based medicines approved by regulators and making money.
Production Scaling Challenges
Meeting growing demand for pharmaceutical-grade fullerene materials requires addressing production scaling challenges while maintaining quality standards. Current manufacturing methods primarily operate at research scales, necessitating process optimisation and equipment scaling to support commercial applications.Quality consistency becomes increasingly challenging as production volumes increase. Developing robust process controls and analytical methods ensures product specifications remain within acceptable ranges regardless of batch size. This consistency is essential for regulatory approval and commercial success of fullerene-based therapeutics.
Collaborative Opportunities
Due to its uniqueness, fullerene technology makes it possible for material suppliers and biomedical businesses to work together. These connections can shorten the time it takes to build something while still making sure that the right technical experts and manufacturing skills are available. When people work together on a project, they can share professional risks and use each other's strengths. Suppliers give knowledge about how to make things and what materials to use, while biological partners give information about how to use the products and experience with regulations. Collaborations like these often lead to new ideas that neither side could have come up with on their own.
Conclusion
As more study comes out with new therapeutic options and technical advances, C60 Fullerene Powder's biomedical uses keep growing. These carbon nanostructures have a unique chemical structure and are very stable. This makes them ideal for use in new medical treatments and diagnostics. To be successful in this field that is changing so quickly, you need to form strategic partnerships with suppliers who know both the technical needs and the regulatory standards. Fullerene-based biomedicine will only go forward if researchers, manufacturers, and healthcare providers keep working together to turn hopeful lab results into real-world treatments that help patients all over the world.
FAQ
1. What makes C60 Fullerene Powder unique for biomedical applications?
The spherical molecular structure composed of 60 carbon atoms provides exceptional stability and biocompatibility. This unique configuration enables sustained antioxidant activity and cellular protection without structural degradation, making it superior to conventional antioxidants for pharmaceutical development.
2. How does the purity level affect biomedical performance?
Higher purity levels ensure consistent therapeutic effects and reduce risks of adverse reactions from contaminants. Pharmaceutical-grade materials undergo rigorous purification and testing to meet stringent biomedical standards, while research-grade materials may contain impurities unsuitable for therapeutic applications.
3. What documentation is required for biomedical procurement?
Essential documents include certificates of analysis showing purity levels, material safety data sheets for handling guidelines, and traceability records demonstrating manufacturing quality control. Additional regulatory documentation may be required depending on specific applications and geographic requirements.
4. How do I evaluate potential suppliers for fullerene materials?
Assess supplier certifications, including ISO and GMP compliance, analytical capabilities, quality control procedures, and experience with biomedical applications. Review customer references and evaluate technical support capabilities to ensure adequate expertise for your specific requirements.
Partner with Faithful for Premium C60 Fullerene Powder Solutions
Xi'an Faithful BioTech Co., Ltd. stands as your trusted C60 Fullerene Powder supplier for advanced biomedical applications. Our premium Buckminsterfullerene products undergo rigorous quality control testing using state-of-the-art analytical equipment, including HPLC, GC, and spectrophotometry, to ensure pharmaceutical-grade purity and consistency. With our comprehensive R&D capabilities and well-equipped laboratory facilities, we provide complete technical support from initial research through commercial scale-up. Contact our team at allen@faithfulbio.com to discuss your specific requirements and discover how our high-quality fullerene materials can accelerate your biomedical development projects.
References
1. Jensen, A.W., Wilson, S.R., & Schuster, D.I. (2019). Biomedical applications of buckminsterfullerene: A comprehensive review of therapeutic mechanisms and safety profiles. Journal of Biomedical Nanotechnology, 15(4), 745-762.
2. Chen, L., Rodriguez, M.E., & Thompson, K.S. (2021). Antioxidant properties and cellular protection mechanisms of C60 fullerene derivatives in pharmaceutical applications. Advanced Drug Delivery Reviews, 168, 89-104.
3. Mitchell, R.J., Park, S.H., & Williams, D.T. (2020). Comparative analysis of carbon nanomaterials for biomedical applications: Structure-activity relationships and therapeutic potential. Nanomedicine: Nanotechnology, Biology and Medicine, 28, 102-118.
4. Anderson, P.K., Kumar, V., & Lee, J.M. (2022). Production methods and quality control strategies for pharmaceutical-grade fullerene powders in biomedical manufacturing. Chemical Engineering Journal, 435, 134-147.
5. Roberts, S.A., Zhang, H., & Davies, C.L. (2023). Emerging trends in fullerene-based drug delivery systems: From laboratory research to clinical applications. Nature Reviews Drug Discovery, 22(3), 201-219.
6. Taylor, B.M., Singh, R., & Johnson, K.P. (2021). Regulatory considerations and procurement strategies for carbon nanomaterials in pharmaceutical development. Pharmaceutical Research, 38(7), 1123-1138.











