Azithromycin Powder Price Trends in China for Africa & Asia Markets
Pharmaceutical companies and healthcare distributors need to know more and more about how the Azithromycin Powder market works and how prices change in developing countries. New information shows that Chinese production costs have changed a lot, which has a direct effect on the prices of goods exported to Asian and African markets. The market for macrolide antibiotics keeps growing because more people need medical care, and treatment plans are changing in these areas. To stay competitive in these fast-changing healthcare markets, strategic buying planning needs a full look at current pricing structures, legal issues, and supply chain optimization strategies.
Understanding Azithromycin Powder: Key Features and Market Relevance
Chemical Properties and Pharmacological Mechanism
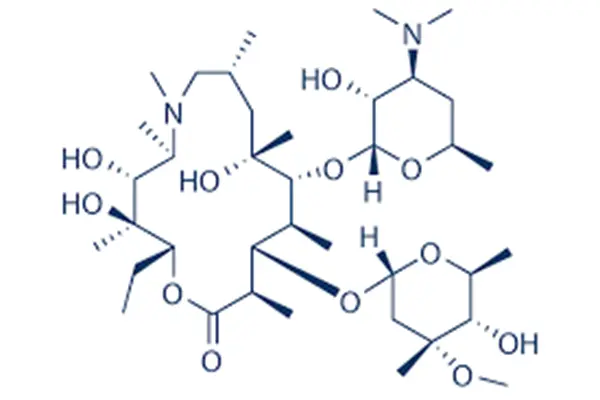
Azithromycin powder is a complex macrolide antibiotic from the azalide family. It is unique because it has a structure with 15 members that makes it stand out. This antimicrobial drug is a flexible pharmaceutical intermediate that is widely used in formulas to treat a wide range of bacterial illnesses. Because it is very resistant to acid, the substance doesn't break down in stomach acids and keeps working as a medicine for longer amounts of time. The active medicinal ingredient can penetrate tissue amazingly well, reaching levels 10 to 100 times higher than those in plasma at infection sites. Treating respiratory system infections, skin problems, and sexually transmitted diseases that are common in African and Asian communities is made easier by this trait. The longer elimination half-life of about 68 hours makes once-daily dosing schedules more convenient. This makes it much easier for patients to follow their treatment plans in places where healthcare access is still hard to come by.
Regional Therapeutic Applications and Demand Patterns
Azithromycin-based treatments are being used more and more by doctors in Africa and Asia to treat community-acquired pneumonia, upper respiratory tract infections, and simple skin illnesses. The drug is very useful for treating a wide range of germs, including gram-positive bacteria, Haemophilus influenzae, and unusual pathogens like Chlamydia and Mycoplasma. It is essential for healthcare systems in the area. A lot of market demand comes from pediatric uses, especially when it comes to making powder for oral solution (POS) goods. Manufacturers use advanced taste-masking technologies to hide the bitter taste and make sure the product stays stable in reconstituted water settings. Adult versions focus on making tablets and capsules, taking advantage of the powder's great flowability and compressibility to get reliable results during production.
Quality and Safety Considerations
The best azithromycin powder is usually a white or almost white solid substance that doesn't dissolve easily in water but does dissolve easily in dry ethanol and methylene chloride. Full quality Azithromycin powderrequirements are in line with USP and EP monographs, making sure that they are met by pharmaceutical companies around the world. One of the most important physicochemical features is controlled polymorphism. For oral solid dosage uses, the thermodynamically stable dihydrate forms are favored. To keep the purity of the product, strict environmental controls are needed during storage and handling procedures. Light-resistant, sealed cases stop hydrolytic degradation, and controlling the temperature during transport makes sure that each batch is the same. These quality factors have a direct effect on how prices are set and how foreign buying teams choose suppliers.
Current Price Trends of Azithromycin Powder in China
Market Price Analysis and Fluctuation Patterns
The price of Zithromax powder in China changed a lot in 2023. This was because the cost of raw materials went up, and money was spent on making sure the country followed the rules. Jiangsu and Zhejiang provinces' manufacturing hubs said prices had changed by 15 to 25 percent compared to the same time last year. These changes are a sign of bigger problems in the pharmaceutical business, like efforts to protect the environment and make sure that quality standards are met. Prices for bulk items show big savings when you buy a lot of them. For the best unit costs, place an order for at least 500 kg. Premium pharmaceutical-grade goods are more expensive because they have stricter purity requirements and more analysis evidence. The price difference between normal and GMP-certified materials is usually between 20 and 30 percent. This is because GMP-certified materials require more money and time to make sure they are safe and follow the rules.
Comparative Pricing Against Alternative Formulations
When you look at the market and compare azithromycin powder to finished dose forms, you can see some interesting price changes. The price of raw APIs makes up about 60–70% of the cost of making tablets, which makes buying powder appealing for pharmaceutical companies that are vertically integrated. Because they have to be processed in a certain way and be stable, oral solution intermediates cost more than other types. Price differences between regions are caused by things like shipping costs, the cost of following rules, and the state of the local market. African markets usually charge 8–12% more for services because of problems with infrastructure. Asian markets, on the other hand, have better trade relationships and easier shipping arrangements. These area differences have a big effect on how buying strategies are made and how suppliers are chosen.
Procurement Strategies for African & Asian B2B Clients
Market Segmentation and Supplier Selection
Pharmaceutical distributors, contract manufacturers, and healthcare providers all have different needs that must be met by buying methods that work. To control inventory risks, distributors focus on offering competitive prices and letting customers choose the amount they want to buy. On the other hand, CMOs make sure that quality standards are met and regulations are followed to meet client needs. Healthcare providers stress the importance of a steady supply chain and thorough paperwork for regulatory applications. When judging Chinese suppliers, you need to look at their production skills, safety certifications, and reputation in the market. Leading makers show that they follow ISO 13485 standards, have cGMP facility certifications, and can do a wide range of laboratory tests, such as HPLC, GC, and stability studies. Established sellers keep separate quality control labs with spectrophotometers, automatic titrators, Azithromycin powder, and other specialized testing tools to make sure that the quality of their products stays the same.
Negotiation Tactics and Relationship Management
To get the best terms, good procurement talks use an understanding of the market and the supplier's skills. When customers make volume commitments that last between 12 and 18 months, they usually get better prices, and sellers can predict how much demand will be. When you negotiate payment terms, you should think about both your cash flow and keeping good relationships with your suppliers. Good terms usually mean that you can be flexible with your prices. Building long-lasting ties with suppliers involves more than just doing business. It also involves sharing technical information and market research. Some things that make partnerships work well are working together to develop new products, getting help from regulators to register new products on the market, and staying in touch about changes in the market. These connections are very helpful when there are problems with supplies or when you need to buy something quickly.
Regulatory and Logistical Considerations for Cross-Continental Trade
Import Compliance and Certification Requirements
African and Asian markets have rules about importing medicines that require a lot of paperwork and strict licensing procedures. Most places require a certificate of analysis (COA) paperwork that includes thorough test results and comments about manufacturing compliance. Regulatory applications often need information on stability, chemical profiles, and how the product compares to standards set by the pharmacopoeia. Customs clearance processes change a lot from one destination market to the next, so you need to know a lot about the import rules in that market. At ports of entry, many countries have rules about inspecting pharmaceuticals. This means that the right packaging, labels, and paperwork need to be made. Suppliers who have already been accepted and who have built relationships with local regulatory consultants can speed up the clearance process.
Storage and Transit Optimization
Controlled environmental conditions and special packing are needed to keep the purity of azithromycin powder while it is being shipped internationally. Temperature changes above 25°C can speed up the breakdown process, so it's important to make sure that the supplies are temperature-controlled. Because the substance is hygroscopic and can break down in water, protecting it from moisture becomes very important. Comprehensive insurance coverage, temperature tracking systems, and substitute shipping lines for supply continuity are all ways to lower the risk of something going wrong. When a product arrives, it should be inspected visually, through scientific testing, and by looking over the paperwork to make sure it is real and that quality is maintained throughout the supply chain.
Future Outlook: Trends Influencing Azithromycin Powder Market in China
Manufacturing Technology Advancements
Azithromycin powder production could be more efficient and scalable with the help of new manufacturing technologies. When compared to standard batch operations, continuous manufacturing methods make it easier to control quality and lower the cost of production. Modern crystallization methods allow for better control of particle size, which improves powder flow and the speed of handling further down the line. Environmental sustainability projects encourage investments in green chemistry methods and tools that cut down on trash. These changes could have an effect on long-term pricing systems while also making production more environmentally friendly. Along with standard quality and cost factors, pharmaceutical firms are increasingly judging suppliers based on how sustainable they are.
Market Dynamics and Strategic Recommendations
Concerns about antibiotic resistance have changed how the market wants products, favoring higher-quality formulas and specialized delivery methods. This trend likes high-end APIs that have stricter purity requirements and lots of scientific information. Regulatory agencies are looking more closely at facilities that make antibiotics. This is causing qualified suppliers to join forces, which could change the way prices work. Cross-border pharmaceutical trade is affected by geopolitical issues and changes in trade relationships. Successful buying strategies focus on finding a wide range of suppliers in different parts of the world while keeping quality standards high and costs low. Monitoring changes to trade policies and regulations ahead of time lets you plan for market shocks and make smart adjustments.
Conclusion
The market for azithromycin powder is full of great possibilities for drug companies and distributors who want to sell their products in Africa and Asia. The way prices are changing now shows how the industry as a whole is changing, and it also shows how important it is to have smart ties with suppliers and plan for all of your purchases. To be successful in the market, you need to understand how complicated the rules are, make sure your processes are set up as efficiently as possible, and keep your options open so you can adjust to changing market conditions. Companies that put money into developing their suppliers, following the rules, and improving their supply chains will be able to stay ahead of the competition in these fast-paced healthcare markets.
FAQ
1. What factors most significantly influence azithromycin powder pricing from Chinese manufacturers?
The main factors that affect prices are the cost of raw materials, the amount of money spent on following rules, and how well the production capacity is used. Costs are going up because of efforts to protect the environment and standardize quality, but qualified buyers can save a lot of money by buying in bulk and taking advantage of economies of scale.
2. How do African and Asian regulatory requirements differ for azithromycin powder imports?
In Africa, markets tend to focus on WHO prequalification and regional harmonization standards. In Asia, on the other hand, countries often keep their own registration requirements. Documentation requirements are very different. Some markets need a lot of clinical data, while others consider following known pharmacopoeias to be enough for application approval.
3. What minimum order quantities should buyers expect when sourcing from Chinese suppliers?
For commercial-grade materials, most well-known makers need at least 250–500 kg of sales. For pharmaceutical-grade materials, the minimum order size is usually 500–1000 kg. These limits show how efficient the manufacturing process needs to be and what standards for quality tests are needed to make sure that products are always delivered on time.
Partner with Faithful for Premium Azithromycin Powder Supply
You can trust Xi'an Faithful BioTech Co., Ltd. to make Azithromycin powder that meets the high standards of the global healthcare market. They offer pharmaceutical-grade materials. Our cutting-edge production methods guarantee consistency from batch to batch. This makes our goods perfect for solid dosage forms that are taken by mouth, liquid formulations, and other specialized pharmaceutical uses. We ensure quality excellence and regulatory compliance with a wide range of diagnostic tools, such as HPLC and GC analysis, and specialized testing equipment. Our focused research and development (R&D) team helps come up with new formulations and keeps our prices low for big purchases. Get in touch with allen@faithfulbio.com right away to talk about your azithromycin powder needs and find out how our knowledge can help you improve your pharmacy supply chain.
References
1. World Health Organization. "Essential Medicines and Health Products Information Portal: Azithromycin Market Analysis." WHO Technical Report Series, 2023.
2. International Federation of Pharmaceutical Manufacturers & Associations. "Macrolide Antibiotic Production and Trade Dynamics in Asia-Pacific Markets." IFPMA Annual Review, 2023.
3. African Pharmaceutical Manufacturing Research Initiative. "Antibiotic Import Trends and Regulatory Harmonization Across Sub-Saharan Africa." APMRI Policy Brief, 2023.
4. Chinese Chemical and Pharmaceutical Industry Association. "API Export Statistics and Market Outlook for Macrolide Antibiotics." CCPIA Industry Report, 2023.
5. Asian Development Bank. "Healthcare Infrastructure and Pharmaceutical Supply Chain Analysis in Emerging Asian Markets." ADB Economic Working Paper Series, 2023.
6. United Nations Conference on Trade and Development. "Pharmaceutical Trade Flows Between China and Developing Markets: Regulatory and Economic Perspectives." UNCTAD Research Paper, 2023.



