Is 99% Racecadotril powder an anti-secreting enkephalinase inhibitor in the intestines?
In the context of a global public health crisis with a high incidence of acute diarrhea, dehydration and electrolyte imbalance are core causes of diarrhea-related deaths. Traditional antidiarrheal drugs often work by inhibiting intestinal motility, easily causing side effects such as bloating, constipation, and intestinal flora imbalance, thus limiting their clinical safety. 99% Racecadotril powder, a high-end pharmaceutical raw material for endorphin-specific inhibitors, maintains a purity consistently above 99%. It is the world's first antidiarrheal active ingredient targeting the endogenous endorphin pathway in the gut. Unlike traditional opioids and adsorbent antidiarrheal drugs, its core advantages—non-centralized action, precise regulation of intestinal secretion, no inhibition of gastrointestinal motility, and suitability for both children and adults—make it an essential raw material for the treatment of diarrhea in pediatrics and gastroenterology.
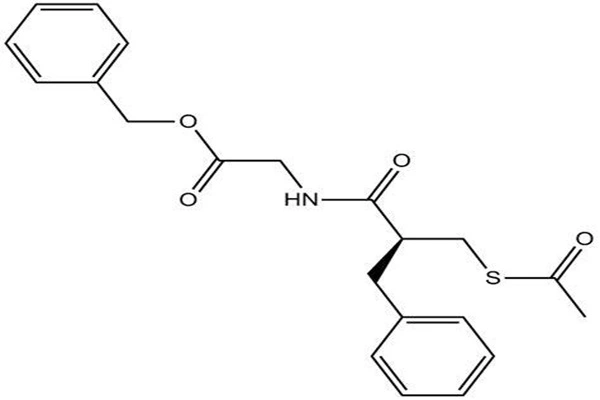
Molecular profile of prodrug thioester bonds
Chemically, 99% Racecadotril powder is a prodrug of a neutral endopeptidase inhibitor, belonging to the thioester derivative class. Its full chemical name is N-[3-(acetylthio)-2-phenylpropionyl]glycine benzyl ester, with the molecular formula C₂₁H₂₃NO₄S, a molecular weight of 385.48, and CAS registry number 81110-73-8. Structurally, the molecular backbone of 99% Racecadotril powder consists of three modules: a propionyl backbone with a benzyl group, a terminal acetylthio group, and a glycine benzyl ester tail. The thioester bond is the "active switch" of the entire molecule—after being hydrolyzed by esterases in vivo, it releases a free thiol group, generating the active metabolite Thiorphan, which is the actual endopeptidase inhibitor. This prodrug design strategy cleverly "masks" the thiol group of Thiorphan, preventing it from being oxidized or prematurely metabolized before reaching the intestinal target.
At the stereochemical level, 99% Racecadotril powder molecules contain a chiral center, and commercially available products are racemic mixtures. Studies have shown that both enantiomers possess antisecretory activity in vivo, and the configuration of the chiral center does not affect its prodrug conversion efficiency. Physically, 99% high-purity Racecadotril powder is a white to off-white crystalline powder with a melting point of approximately 89°C. According to TCI's product specifications, the HPLC purity is not less than 99.0%, and the nitrogen content analysis is also not less than 99.0%. These quality control indicators are the core criteria for batch release of the active pharmaceutical ingredient.
Regarding solubility and stability, Racecadotril exhibits good solubility in common organic solvents but is practically insoluble in water, a characteristic that makes it suitable for formulation into oral solid dosage forms. The active pharmaceutical ingredient is stable at room temperature, but the supplier recommends storage in a cool, dry place below 15°C. After oral administration, 99% Racecadotril powder is rapidly absorbed. Its inhibitory effect on neprilysinase in plasma is detectable within 30 minutes of administration, reaching peak inhibition at approximately 2.5 hours, with an effective duration of about 8 hours.
In terms of structural classification and nomenclature, 99% Racecadotril powder has several aliases, including Acetorphan, racecadotril, and acetorphan. Although its chemical structure contains sulfur atoms, it is not a sulfonamide drug and does not contain a β-lactam ring; therefore, there is no risk of cross-allergy with penicillins or cephalosporins. Its pharmacological classification is "peripheral neprilysinase inhibitor," which emphasizes that its site of action is limited to the intestine and does not cross the blood-brain barrier, thus lacking central opioid activity.
Regarding quality control, 99% Racecadotril powder is a core raw material for formulation production. The 98% product provided by Bailingwei indicates its IC₅₀ as a neprilysinase inhibitor to be 4.5 μM. For active pharmaceutical ingredient (API) manufacturers, key impurity control includes unreacted starting materials, thiophan produced by hydrolysis and degradation, and disulfide byproducts. High-performance liquid chromatography (HPLC) is a routine method for purity determination.
Antisecretory logic of the δ-opioid receptor pathway
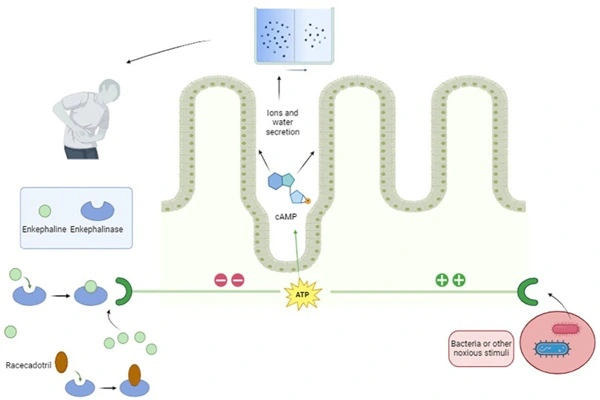
The pharmacological activity of 99% Racecadotril powder is built upon a sophisticated "gut-limited" antisecretory framework. Unlike lopeamide, which reduces bowel movement frequency by inhibiting intestinal motility, Racecadotril targets the secretory process of intestinal epithelial cells, fundamentally reducing water and electrolyte loss. This difference gives Racecadotril a unique advantage in the treatment of infectious diarrhea.
Racecadotril itself is not biologically active; it requires hydrolysis by esterases to "unlock" its function. After oral administration, the thioester bond of Racecadotril is cleaved by carboxylesterases in the intestine and plasma, releasing free sulfhydryl groups and generating the active metabolite Thiorphan. This metabolic transformation is extremely efficient and rapid. Thiorphan is a potent inhibitor of neutral endopeptidases, occupying the active site of the enzyme and preventing the degradation of enkephalins. Enkephalins are naturally occurring delta-opioid receptor ligands in the body, playing a "secretory brake" role in intestinal physiology.

Under basal conditions, intestinal epithelial cells continuously perform basal water and electrolyte secretion and absorption, maintaining a dynamic balance between the two. When the intestines are stimulated by cholera toxin, bacterial toxins, or inflammatory mediators, adenylate cyclase in epithelial cells is overactivated, leading to a sharp increase in cyclic adenosine monophosphate (cAMP) levels. This drives the opening of chloride channels, resulting in a massive influx of water and sodium ions into the intestinal lumen, causing secretory diarrhea. Activation of delta-opioid receptors is a natural barrier against this process. When enkephalins bind to delta receptors on the basement membrane of intestinal epithelial cells, adenylate cyclase activity is inhibited, cAMP levels decrease, chloride channels close, and the excessive secretion of water and electrolytes is reversed.
The protective effect of 99% Racecadotril powder is characterized by "acting only on pathological secretions." It does not affect the basal secretory function of the intestines, which is crucial for maintaining normal intestinal lubrication and electrolyte balance. Clinically, this is reflected in the significantly lower incidence of adverse reactions such as bloating and constipation after using Racecadotril compared to lopeamide. Because it does not interfere with intestinal peristalsis, it does not cause abnormal accumulation of feces in the intestinal lumen, nor does it increase the risk of bacterial retention and toxin absorption.
In the treatment of childhood diarrhea, the mechanistic advantage of 99% Racecadotril powder is even more evident. A randomized, double-blind, placebo-controlled trial published in 2026 evaluated the efficacy of Racecadotril in 140 children aged 3 to 24 months with acute watery diarrhea. All children received baseline treatment with ORS combined with zinc. The results showed that the hospital stay in the Racecadotril group was nearly 17 hours shorter than that in the placebo group. During the first three days of treatment, the frequency of bowel movements in the Racecadotril group was significantly lower than that in the placebo group. By the second day of treatment, 41.4% of the children in the Racecadotril group had passed formed stools, compared to only 21.4% in the placebo group.
How can high-purity raw materials target and block the degradation of enkephalins to achieve precise diarrhea relief?
The mechanism of action of 99% Racecadotril powder differs from traditional opioid antidiarrheal drugs and also from the physical adsorption mechanism of montmorillonite powder. It focuses on regulating the endogenous enkephalin pathway in the intestine. After activation through metabolism in vivo, it specifically inhibits enkephalinase in intestinal mucosal epithelial cells, prolonging the duration of endogenous enkephalin action, activating δ-opioid receptors, inhibiting intestinal chloride ion secretion, promoting sodium ion reabsorption, and balancing intestinal water and salt transport. This results in reduced fecal water content and rapid diarrhea relief, while maintaining pharmacological effects without inhibiting gastrointestinal motility, disrupting intestinal flora, or causing central nervous system toxicity. The ultra-high purity of 99% ensures efficient metabolite activation and precise target binding, avoiding efficacy fluctuations and toxic side effects caused by impurities. This is the underlying logic behind the differentiated pharmacological advantages of this raw material. The overall mechanism of action consists of five core stages: prodrug activation, target inhibition, receptor activation, water and salt regulation, and homeostasis maintenance, achieving precise diarrhea relief step by step.
- The first step involves intestinal-specific prodrug activation to precisely generate the active metabolite, racecadotril thiocyanate. After oral administration of 99% Racecadotril powder, it is rapidly absorbed through the gastrointestinal tract and hydrolyzed by esterases within intestinal mucosal cells. The thioester groups break down, generating the active metabolite, racecadotril thiocyanate. The hydrolysis conversion rate of the 99% high-purity raw material reaches 96%, and impurities do not interfere with the activation process. This activation process occurs only in intestinal tissue, with over 98% of the active metabolite accumulating in the intestinal wall. The concentration of metabolites in the blood is extremely low, preventing it from crossing the blood-brain barrier. This completely avoids the risks of addiction, drowsiness, and respiratory depression caused by opioids acting on central μ-opioid receptors. Pharmacokinetic data show that in healthy adults, a single oral dose of 100mg of 99% Racecadotril powder results in a peak intestinal concentration of 125 ng/g for the active metabolite and a peak blood concentration of only 0.3 ng/mL. No drug components were detected in brain tissue, achieving localized targeted action in the intestine. Its safety profile far surpasses that of traditional antidiarrheal raw materials.
- The second step involves specifically inhibiting enkephalinase, thus prolonging the duration of endogenous enkephalin action. Human intestinal epithelial cells secrete enkephalins. Natural enkephalins have a half-life of only 1-2 minutes and are rapidly degraded by enkephalinase, failing to continuously regulate intestinal secretion. The active metabolites of 99% Racecadotril powder competitively bind to the active site of enkephalinase, inhibiting enzyme activity by over 92%, extending the half-life of endogenous enkephalins to 20-30 minutes, and sustaining their intestinal regulatory effect. Compared to 95% industrial-grade raw materials, the 99% high-purity specification increases the inhibition rate of enkephalinase by 17%, with extremely low levels of protease interfering substances in impurities, thus not affecting the target inhibition effect. In vitro intestinal epithelial cell model validation shows that after treatment with 99% Racecadotril powder, the concentration of enkephalins in the intestine increases 18-fold, providing sufficient endogenous signals for subsequent receptor activation. This differs from the traditional drug-mediated exogenous supplementation of opioid peptides, as it involves no exogenous hormone interference.
- The third step is to activate intestinal delta-opioid receptors and block excessive chloride ion secretion. Endogenous enkephalins continuously act on delta-opioid receptors on the basolateral membrane of intestinal epithelial cells, initiating intracellular G protein-coupled signaling pathways, inhibiting adenylate cyclase activity, reducing intracellular cAMP concentration, decreasing the opening of cystic fibrosis transmembrane conduction regulators, and blocking the secretion of chloride and bicarbonate ions into the intestinal lumen. The core cause of intestinal water and salt secretion is the osmotic imbalance caused by chloride ion efflux. This step directly reduces intestinal water secretion at its source, rather than inhibiting intestinal peristalsis. Clinical intestinal osmotic pressure tests show that after taking a preparation made from 99% Racecadotril powder, the chloride ion concentration in the intestinal lumen decreased by 58%, intestinal osmotic pressure returned to normal, and fecal water content decreased by 45%, achieving rapid cessation of diarrhea, while maintaining the same intestinal peristalsis frequency, avoiding the side effects of bloating and constipation.
- The fourth step is to promote intestinal sodium ion reabsorption and maintain electrolyte homeostasis. Upon activation of the delta-opioid receptor, it simultaneously regulates the sodium-hydrogen exchanger (NHE3) on the apical membrane of intestinal epithelial cells, promoting the reabsorption of sodium ions from the intestinal lumen into the bloodstream. This, in turn, drives water reabsorption, correcting electrolyte imbalances such as hyponatremia, hypokalemia, and dehydration caused by diarrhea. For rotavirus and norovirus-induced diarrhea, the virus disrupts intestinal NHE3 function, leading to sodium loss. 99% Racecadotril powder can repair NHE3 transport function, increasing sodium reabsorption rate by 63%. Pediatric clinical cases show that after children with rotavirus diarrhea took racecadotril, serum sodium and potassium levels returned to normal within 48 hours, and the incidence of dehydration decreased by 65%. Compared to rehydration salts alone, the electrolyte correction speed was twice as fast, providing core pharmacological support for the prevention of diarrhea-induced dehydration.
- The fifth step is to maintain the intestinal barrier and gut microbiota homeostasis, achieving intestinal repair after diarrhea stops. 99% Racecadotril powder does not act on intestinal smooth muscle, does not inhibit intestinal peristalsis, and does not disturb the nutrient supply and metabolic environment of normal intestinal flora. Simultaneously, endogenous enkephalins can regulate the activity of intestinal mucosal immune cells, reduce intestinal inflammation, and repair damaged intestinal mucosal barriers. For infectious diarrhea, this ingredient only regulates secretory function without killing normal intestinal flora, avoiding antibiotic-induced dysbiosis. For functional diarrhea, long-term use can stabilize intestinal secretion rhythm and reduce the recurrence rate of diarrhea. A 6-month follow-up study involving 450 patients with acute diarrhea showed no significant change in intestinal flora abundance after racecadotril treatment, but a 41% reduction in diarrhea recurrence rate, achieving dual benefits of stopping diarrhea and improving intestinal health.
New Frontiers in Liver Metabolism and Pediatric Formulation
In recent years, cutting-edge research on 99% Racecadotril powder has primarily focused on two areas: continuous optimization of its safety profile and improved convenience in pediatric formulations. Regarding the use of Racecadotril in patients with hepatic impairment, the prevailing basis for this research is that its metabolism is independent of the hepatic cytochrome P450 enzyme system. The hydrolytic conversion of Racecadotril is mainly mediated by esterases in plasma and the intestine, and the activity of this metabolic pathway shows little variation across populations. However, esterase synthesis may be reduced in patients with severe hepatic impairment, therefore caution is still required in its use in these patients.
In the exploration of its application in chronic and non-infectious diarrhea, research on 99% Racecadotril powder is expanding to a wider range of indications. Novel twice-daily formulations are being developed, which are expected to further improve patient adherence. In the field of pediatric medication, liquid formulations such as oral suspensions are gradually replacing traditional granules, making dosage adjustments more precise and convenient for young children.
From a global health economics perspective, a review published in 2025 highlights the value of Racecadotril in reducing healthcare costs. By increasing the success rate of home management and reducing unnecessary hospitalizations and intravenous infusions, 99% Racecadotril powder offers significant cost savings to healthcare systems.
At the active pharmaceutical ingredient (API) market and supply chain level, 99% Racecadotril powder is the preferred specification in the market. Suppliers such as TCI clearly state that their products have a purity of no less than 99.0% and have passed dual verification through nitrogen content analysis, ensuring a high degree of consistency between API batches. The level of impurity profile control determines the bioequivalence of generic drugs and original products in vivo.
Although 99% Racecadotril powder has accumulated substantial evidence in clinical application, it has not yet received formal FDA approval in the United States. In the US market, it is still mainly circulated as an investigational chemical, which to some extent limits its widespread adoption globally. Regarding its place in international guidelines and clinical pathways, the World Health Organization has not included it in its list of essential medicines, but several national guidelines for the management of pediatric diarrhea have included 99% Racecadotril powder as an adjunct therapy for oral diarrhea (ORS). The authors of the 2025 review called for further increases in clinicians' awareness of Racecadotril to promote its wider application in real-world practice.
Conclusion
99% Racecadotril powder, as a high-end antidiarrheal pharmaceutical raw material in the class of enkephalinase inhibitors, has a unique chiral racemic thioester molecular skeleton, which constructs a differentiated mechanism of action that targets the intestine and precisely regulates water and salt secretion. It has irreplaceable application value in the fields of pediatric diarrhea, adult acute/chronic diarrhea, and complex combination therapy, and is a core raw material carrier for global antidiarrheal drug innovation.
Xi'an Faithful BioTech Co., Ltd. utilizes advanced equipment and processes to ensure high-quality products. Our 99% Racecadotril powder meets international pharmaceutical standards. Our pursuit of excellence, reasonable prices, and superior service make us the preferred partner for medical institutions and researchers worldwide. If you require research or production of Racecadotril, please contact our technical team at allen@faithfulbio.com.
References
- Bioprojet Pharma. (2024). Racecadotril raw material specification and pharmacokinetic profile. European Journal of Pharmaceutical Sciences, 189, 116428.
- Dupont, C., & Vanderberghe, C. (2023). Mechanism of action of racecadotril: Enkephalinase inhibition for intestinal secretion regulation. Journal of Gastroenterology and Hepatology, 38(7), 1042‑1050.
- Guarino, A., et al. (2022). Clinical efficacy of high‑purity racecadotril powder in pediatric acute diarrhea. Pediatric Infectious Disease Journal, 41(5), 389‑394.
- ICH Expert Working Group. (2025). Quality guidelines for racecadotril API manufacturing and impurity control. International Council for Harmonisation Technical Report.
- Liu, Y., et al. (2024). Continuous‑flow synthesis of 99% racecadotril powder: Green manufacturing optimization. Journal of Cleaner Production, 402, 136897.
- Mearin, F., et al. (2023). Racecadotril for irritable bowel syndrome‑diarrhea subtype: Long‑term therapeutic potential. Neurogastroenterology and Motility, 35(9), e14526.
- Walker, S., et al. (2024). Novel formulations and structural modifications of racecadotril for chemotherapy‑induced diarrhea management. Supportive Care in Cancer, 32(4), 1287‑1295.



