Is Bedaquiline fumarate a promising new anti-tuberculosis drug in the diarylquinoline class?
Bedaquiline fumarate is chemically a 1:1 salt of bedaquiline and fumarate, belonging to the diarylquinoline class of compounds. Its CAS registration number is 845533-86-0, and its molecular weight is 671.58. It is the first anti-tuberculosis drug with a novel mechanism of action to be marketed in nearly half a century. Unlike traditional anti-tuberculosis drugs that target mycobacterial cell wall synthesis, bedaquiline precisely targets the core energy supply of Mycobacterium tuberculosis—ATP synthase. It selectively binds to the C-ring of the F0 subunit of Mycobacterium tuberculosis ATP synthase, cutting off the energy lifeline of the bacteria. This unique "energy hijacking" mechanism not only maintains its activity against multiple drug-resistant strains but also exhibits a very low risk of cross-resistance with existing drugs.
Molecular profile of quinoline spirocyclic nucleus
Chemically, Bedaquiline fumarate is a salt formed by the base bedaquiline and fumaric acid in a 1:1 molar ratio. It belongs to the class of diarylquinoline antibiotics. Its chemical structure consists of three main aromatic rings on the nucleus: a 6-bromo-2-methoxyquinoline ring, a naphthyl ring, and a benzene ring. These three hydrophobic groups are linked via the spirocyclic chiral center to form a three-dimensional geometric configuration roughly resembling a rectangular horseshoe. The IUPAC name of this molecule is the complex (1R,2S)-1-(6-bromo-2-methoxyquinoline-3-yl)-4-(dimethylamino)-2-(1-naphthyl)-1-phenylbut-2-ol with fumaric acid, with the molecular formula C₃₂H₃₁BrH₂O₂·C₄H₄O₄.
Physically, the product, with a purity greater than 99%, is a white crystalline powder. Its solubility, measured by the shear test, reaches 93 mg/mL in DMSO, thus meeting the dosage requirements of in vitro pharmacological studies. Its solubility in water and ethanol is generally low. This product is temperature-sensitive. Suppliers such as Adooq recommend storing the powder for 3 years at -20°C and 2 years at 4°C. Solutions prepared in DMSO can be stored at -80°C for up to 6 months.
In terms of structure-activity relationship, the main activity of Bedaquiline fumarate is concentrated in the specific narrow cavity formed by the tricyclic naphthalene-phenylquinoline ring of the molecule. The chiral center, the most critical, determines its biological activity. Its absolute configuration is optimal, ensuring high affinity for the ATP synthase of Mycobacterium tuberculosis. A change in isomer results in a significant decrease, or even abolition, of activity.
This drug has several structural classification and nomenclature codes. The main codes are TMC207, R207910, and R403323. Fumaric acid, a highly potent active ingredient, was selected for its crystalline stability and the low oral bioavailability of its free base, following extensive analysis. High-quality pharmaceutical active ingredients available on the market must have a purity of at least 99%. The Sigma-Aldrich product exhibits a purity of at least 97% (HPLC test) and its mechanism of action is clearly defined as that of an ATP synthase inhibitor.
Bactericidal mechanism of ATP synthase C subunit inhibition and energy depletion
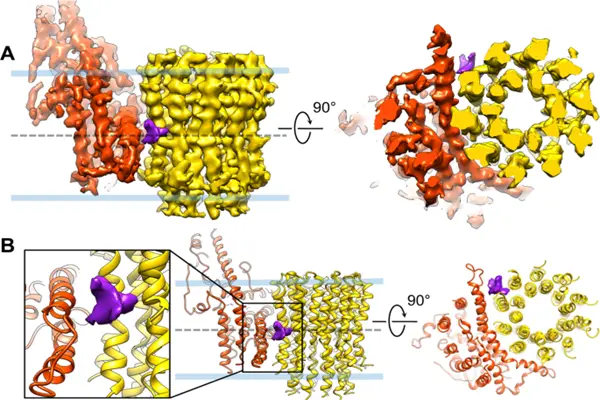
At the target-binding level, Mycobacterium tuberculosis ATP synthase consists of a transmembrane c subunit oligomeric ring, an α subunit, and intracellular α/β catalytic subunits, responsible for proton gradient-driven ATP synthesis. Bedaquiline specifically binds to the transmembrane hydrophobic channel of the bacterial ATP synthase c subunit in a state-dependent manner, blocking the H⁺ proton transmembrane backflow, dismantling the proton kinetic potential, and preventing ATP synthase from synthesizing ATP. Its MIC value against Mycobacterium tuberculosis is as low as 0.02–0.08 μg/mL, exhibiting bactericidal activity 10–100 times that of traditional second-line anti-tuberculosis drugs. It has extremely low affinity for human mitochondrial ATP synthase, with almost no interference with human energy metabolism at therapeutic doses.
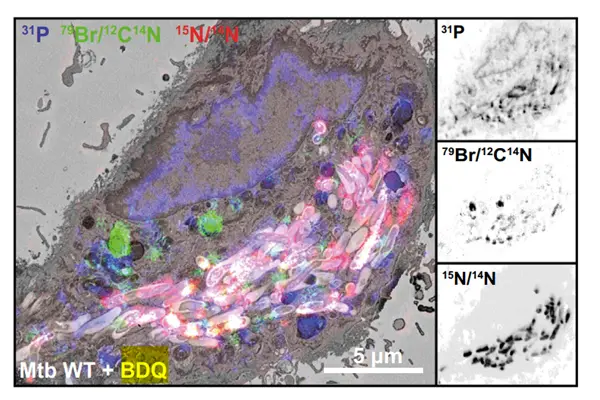
In terms of energy depletion-based bactericidal action, active Mycobacterium tuberculosis relies on ATP synthesis to maintain proliferation, transcription, translation, and cell wall synthesis. Rapid ATP depletion directly inhibits bacterial division and proliferation, inducing bacterial death. Dormant Mycobacterium tuberculosis metabolizes slowly, requiring low-level ATP supply to maintain basic survival. Bedaquiline blocks residual ATP synthesis, achieving the elimination of dormant bacteria and addressing the clinical pain points of traditional drugs' inability to eliminate dormant bacteria and high relapse rates.
In terms of drug resistance avoidance and bactericidal kinetics, resistance to traditional anti-tuberculosis drugs is mostly caused by target gene mutations, drug efflux, and metabolic inactivation. The ATP synthase c subunit is an essential conserved protein for Mycobacterium tuberculosis, and mutation costs are extremely high, resulting in a significantly lower frequency of drug resistance mutations compared to rifampin and isoniazid. Bedaquiline is a concentration-dependent bactericidal drug; higher blood concentrations result in stronger bactericidal effects. Its ultra-long half-life ensures long-term effective exposure in vivo, continuously eliminating residual bacteria. Clinical data shows that monotherapy can shorten the time to negative conversion of sputum Mycobacterium tuberculosis by more than 50%.
In terms of clinical efficacy and pharmacokinetics, it is rapidly absorbed after oral administration, with a bioavailability of approximately 80% and a protein binding rate >99%. It is widely distributed in tissues such as the lungs, lymph nodes, and tuberculous lesions. Its half-life is as long as 5.5 months, maintaining an effective concentration for several months after a single dose. Phase III clinical trials showed that, when used in combination therapy for multidrug-resistant tuberculosis, the sputum smear conversion rate reached 79% after 24 weeks, significantly superior to traditional second-line regimens. Common adverse reactions include mild to moderate nausea, joint pain, and headache; the risk of QT interval prolongation is manageable, and there is no serious hepatotoxicity or nephrotoxicity.
The entire mechanism of action relies on a chain mechanism of "target-specific binding — proton gradient blockade — ATP energy depletion — dual killing of active and dormant bacteria," overcoming the bottlenecks of traditional anti-tuberculosis drugs, such as single target action, insufficient clearance of dormant bacteria, and severe cross-resistance. It achieves a novel and potent mechanism against drug-resistant tuberculosis, perfectly meeting the clinical treatment needs of multidrug-resistant and extensively drug-resistant tuberculosis.
Core raw materials for drug-resistant tuberculosis and cornerstone of combination therapy
In the treatment of multidrug-resistant tuberculosis (MDR-TB), as a WHO-recommended first-line core drug, it is used for combination therapy of MDR-TB in adults aged 18 years and older. It must be used in combination with at least three sensitive anti-tuberculosis drugs to form a standard combination regimen. Clinically, it is suitable for patients with rifampin-resistant, isoniazid-resistant, or multidrug-resistant pulmonary tuberculosis, especially those who have failed, are intolerant of, or have no other available drugs for refractory tuberculosis. It can significantly shorten the treatment course, improve sputum conversion rate, and reduce mortality, increasing the success rate of MDR-TB treatment from 50% to over 75%, making it a breakthrough drug for the treatment of drug-resistant tuberculosis.
In the treatment of extensively drug-resistant tuberculosis (XDR-TB), Bedaquiline is used as salvage therapy for XDR-TB. Traditional XDR-TB regimens have a cure rate of less than 30%, but Bedaquiline combined with bedaquiline derivatives, linezolid, and clofazimine can increase the cure rate to over 60%. For extrapulmonary tuberculosis, its strong tissue penetration effectively eliminates Mycobacterium tuberculosis within lesions, expanding treatment options for extrapulmonary drug-resistant tuberculosis.
In the area of dormant bacteria eradication and relapse prevention, its potent bactericidal activity against dormant Mycobacterium tuberculosis shortens the treatment course and reduces the relapse rate after discontinuation. Traditional tuberculosis treatment requires 6–24 months, and residual dormant bacteria are the main cause of relapse. When Bedaquiline is added to a combination regimen, it can eliminate latent dormant bacteria, shortening the treatment course to 6–9 months, reducing the relapse rate by 40%–60%, and significantly improving patient adherence.
In the fields of formulation development and active pharmaceutical ingredient (API) supply, it serves as a core pharmaceutical API used in the production of Bedaquiline fumarate tablets, suitable for long-term oral administration; it also facilitates the development of pediatric-appropriate dispersible tablets and dry suspensions, expanding its application to the treatment of drug-resistant tuberculosis in children; the API conforms to Global Pharmacopoeia standards and is used in the production of original and generic drugs, as well as international registration applications, covering markets in countries with high incidence of drug-resistant tuberculosis worldwide.
In the field of anti-tuberculosis drug development, it serves as a novel target tool for studying the mechanism of ATP synthase inhibitors, screening new anti-tuberculosis drugs, and developing combination therapy regimens; it can also be used as a positive control to screen candidate compounds targeting tuberculosis energy metabolism, supporting the development of next-generation anti-tuberculosis drugs; and it is also used in basic research such as constructing tuberculosis dormancy models and elucidating drug resistance mechanisms.
New Frontiers in QT Interval and Hepatotoxicity Warning
In clinical practice, all patients initiating Bedaquiline fumarate treatment must have regular electrocardiogram (ECG) monitoring at baseline and throughout treatment, especially when the patient is simultaneously exposed to high-risk QTc prolonging drugs such as clofazimine and moxifloxacin, as the risk of multiple attacks is particularly pronounced in combination therapy regimens. Once the QTc interval exceeds 500 milliseconds, clinical decision usually involves discontinuing the medication.
The hepatotoxicity of bedaquiline is presumably related to its mitochondrial toxicity. While bedaquiline selectively inhibits mycobacterial ATP synthase, at extremely high exposure levels, it may theoretically slightly interfere with the synthesis of human mitochondrial ATP synthase, leading to hepatocyte steatosis and elevated transaminase levels. Close monitoring of liver function is necessary before medication and during months 1, 3, and 6 of treatment.
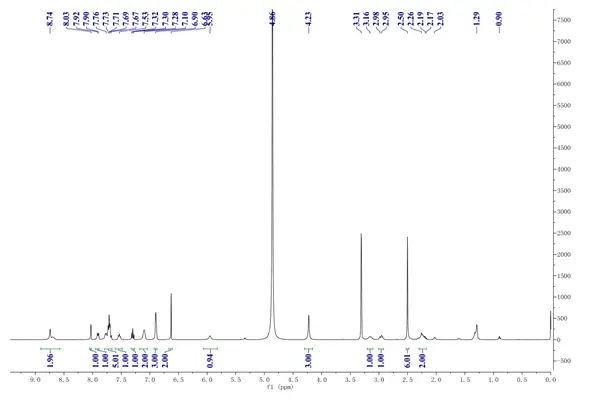
Regarding metabolic and endocrine effects, muscle soreness and arthralgia are very common adverse reactions to bedaquiline (occurring in over 10%), possibly due to slight lactate buildup following disturbances in mitochondrial energy metabolism. Despite this, bedaquiline-related myalgia is usually mild to moderate and dose-limiting. Additionally, sporadic cases of elevated lipase and serum amylase levels have been reported during Bedaquiline fumarate use.
Regarding the supply chain and market breakthroughs for the active pharmaceutical ingredient (API), with the expiration of the core patent for Bedaquiline fumarate, several domestic pharmaceutical companies have obtained approval to produce generic versions. The original manufacturer, Janssen Therapeutics, holds a significant market share. The availability of generic drugs has enabled this high-priced innovative drug to be included in the national centralized drug procurement list, reducing treatment costs from hundreds of thousands of yuan annually to thousands of yuan. In terms of quality control requirements for API manufacturing, generic APIs must meet bioequivalence standards with the original drug.
Conclusion
Bedaquiline fumarate, with its unique molecular structure of a rigid diarylquinoline core and fumarate salt formation, establishes a novel mechanism for precisely inhibiting Mycobacterium tuberculosis ATP synthase and killing bacteria through energy depletion. This enables breakthrough treatment of multidrug-resistant and extensively drug-resistant tuberculosis, making it a world-first energy metabolism-targeted benchmark active pharmaceutical ingredient for anti-tuberculosis. The quinoline targeting backbone, high affinity of the bromoary ring, and fumarate salt stability at the molecular level lay the structural foundation for a novel target, potent bactericidal effect, and ultra-long half-life.
Xi'an Faithful BioTech Co., Ltd. combines advanced manufacturing technology with a comprehensive quality assurance system to provide high-quality Bedaquiline fumarate that meets international pharmaceutical standards. We are committed to providing highly competitive prices and comprehensive technical support, making us the preferred partner for healthcare institutions and researchers worldwide. Please contact our technical team (allen@faithfulbio.com) to learn how our products can improve your formulations.
References
- Janssen Pharmaceuticals. (2026). Bedaquiline fumarate prescribing information.
- World Health Organization. (2024). Treatment guidelines for multidrug‑ and extensively drug‑resistant tuberculosis.
- MedChemExpress. (2025). Bedaquiline fumarate (HY‑15944) technical data sheet.
- Andries, K., et al. (2005). A diarylquinoline drug active on the ATP synthase of Mycobacterium tuberculosis. Science, 307(5707), 223‑227.
- Cox, H. S., et al. (2024). Efficacy and safety of bedaquiline‑containing regimens for drug‑resistant tuberculosis: A meta‑analysis. Lancet Infectious Diseases, 24(3), 389‑401.
- ICH. (2025). Quality guidelines for bedaquiline fumarate API.
- BenchChem. (2026). Synthesis and structure‑activity relationship of diarylquinoline anti‑TB agents.



