What is estradiol cypionate powder?
Understanding raw material characteristics is crucial for finding high-quality pharmaceutical intermediates. Estradiol cypionate powder is a pharmaceutical-grade API that binds 17β-estradiol to a C17 cypionate ester. lengthy-acting injectable formulations that give estrogenic action over lengthy durations use this white to off-white crystalline powder. The cypionate esterification increases lipophilicity and biological half-life, distinguishing it from other estradiol derivatives in formulation and performance.
Understanding Estradiol Cypionate: Chemical Identity and Core Positioning
Estradiol cypionate differs from basic estradiol compounds in molecular structure. By attaching a cyclopentylpropionate chain to the hydroxyl group at position 17, pharmaceutical chemists create a prodrug that undergoes gradual hydrolysis after intramuscular administration. This structural alteration turns a rapidly metabolized hormone into a depot formulation that lasts one to two weeks after injection.
Raw Estradiol Cypionate Powder is usually a microcrystalline solid with a melting point between 149-153°C, demonstrating purity and structure. The chemical has a molecular weight of 396.56 g/mol and weak water solubility but great solubility in organic vehicles, including ethyl oleate, sesame oil, and cottonseed oil, which guide formulation techniques.

Pharmaceutical companies emphasize this API because it solves a major hormone delivery problem: steady blood levels without frequent doses. As endogenous esterases slowly break the cypionate ester link, bioactive estradiol is released into circulation. The powder's pharmacokinetic characteristics make it ideal for medicinal applications needing predictable, persistent estrogenic actions.
The positioning within pharmaceutical portfolios typically falls under specialized hormone APIs rather than commodity chemicals. Batch-to-batch uniformity, crystalline purity above 98%, and extensive analytical documentation distinguish pharmaceutical-grade material from research-grade. HPLC, GC, and spectroscopic analysis are used by manufacturers to ensure pharmacopeial compliance.
The Critical Industry Problems Estradiol Cypionate Powder Solves
Hormone formulations remain difficult for pharmaceutical and CDMO businesses. Daily drug compliance plummets, especially for chronic illnesses requiring long-term treatment. Estradiol cypionate powder allows weekly or bimonthly injections instead of a daily oral dose, addressing this adherence issue.
Another issue is bioavailability fluctuation. Oral estradiol undergoes extensive first-pass hepatic metabolism, resulting in unpredictable serum levels influenced by individual metabolic differences, food intake, and drug interactions. Estradiol cypionate injectables bypass hepatic metabolism and enter the systemic circulation via the intramuscular depot. First-pass variability is eliminated, resulting in more uniform pharmacokinetic profiles across patient groups.
Hormone product formulation stability is an issue. Unesterified estradiol degrades oxidatively during storage and is unstable in several vehicle systems. The cypionate ester chemically stabilizes the steroid nucleus, preventing degradation and sustaining potency over time. Product formulations employing high-quality raw estradiol cypionate powder typically have 24-36 month stability profiles under proper storage.
Another benefit of this API is manufacturing scalability. The crystalline powder format facilitates accurate weighing, blending, and dissolution in oil vehicles during compounding operations. Estradiol cypionate dissolves well in pharmaceutical-grade oils at concentrations from 1 mg/mL to 5 mg/mL, making scale-up from laboratory batches to commercial production quantities easy.
The compound's analytical simplicity pleases quality control teams. HPLC assays measure APIs in raw materials and final formulations, whereas related substances testing detects contaminants and degradation products. For GMP-compliant production, analytical access provides comprehensive quality assurance processes.
Core Features and Functional Characteristics Deep Dive
Estradiol cypionate powder's estrogen API performance is influenced by various linked factors. Understanding these factors helps manufacturers improve formulation development and set specs.
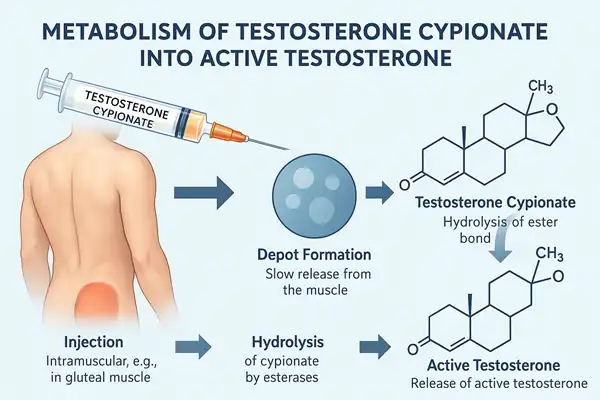
Enhanced Lipophilicity and Tissue Retention: Cypionate ester partition coefficient is much higher than that of unmodified estradiol. Because the chemical is lipophilic, it accumulates at the injection site in muscle tissue and adipose tissue. This depot's slow release controls the extended duration of effect, with tissue retention kinetics depending on injection site blood flow, muscle mass, and vehicle composition.
Esterases hydrolyze the cypionate moiety after intramuscular delivery, raising blood estradiol levels over 24-72 hours. Peak concentrations occur 3-5 days post-injection, then gradually drop as the depot depletes. The pharmacokinetic curve is different from oral administration, where estradiol levels spike within hours and rapidly decline.
Esterase-mediated bioactivation: Prodrugs must be converted to active estradiol. Tissue and plasma esterases hydrolyze ester bonds, producing estradiol and cyclopentylpropionic acid. Only the active hormone reaches estrogen receptors throughout the body due to this biological activation process, whereas the cleaved ester component undergoes ordinary metabolic disposition.
Crystalline Stability Profile: High-quality powder is stable as a solid at 2-8°C, sheltered from light and moisture. Steroid hormone oxidation and breakdown processes are blocked by the crystalline lattice. Professional packaging keeps material potent for 3-5 years, helping producers manage inventories.
While mostly used in oil-based injectable solutions, the powder's solubility allows formulation scientists to investigate other vehicle systems. Benzyl benzoate increases concentration in compact injectable volumes, whereas sesame, cottonseed, and ethyl oleate oil bases manage injection site tolerability and release kinetics.
The substance is easily analyzed using pharmaceutical methods. Reverse-phase columns with UV detection at 280 nm improve HPLC sensitivity and specificity. Gas chromatography verifies ester structure, whereas mass spectrometry identifies molecules. This analytical compatibility facilitates quality control lab technique creation and validation.
Technical Foundations Supporting Performance
The pharmaceutical use of estradiol cypionate powder is based on steroid hormone chemistry and pharmacokinetics. Esterification is a well-known organic synthesis that involves acylation of the 17β-hydroxyl group with cypionate acid under controlled circumstances employing catalysts and reaction parameters.
Pharma companies assess raw material quality using several technological lenses. XRPD verifies crystalline structure, confirming polymorphism. DSC checks thermal characteristics and finds amorphous material that may impair dissolution and stability. These solid-state characterisation methods ensure incoming material meets standards and performs consistently during formulation.
The intramuscular depot mechanism depends on numerous biological aspects that formulation scientists must consider. Gluteal muscles have great volume capacity and moderate blood flow, while deltoid administration has higher perfusion rates and quicker absorption. Injectability and depot formation are affected by vehicle viscosity, which increases residence length but may reduce patient comfort.
Generic versions and formulation modifications must address bioequivalence. The pharmacokinetic profile depends on vehicle composition, concentration, injection volume, and needle gauge. The formulation's estradiol exposure must be carefully controlled by manufacturers to match reference items.
Advanced manufacturing facilities use environmental controls to avoid processing contamination and deterioration. To prevent moisture absorption and hydrolytic breakdown, hormone APIs must be handled under regulated humidity. Nitrogen blanketing prevents oxidation during weighing and compounding. These technical controls demonstrate the advanced knowledge needed to operate with steroid hormone intermediates.
Key Advantages for Pharmaceutical Manufacturing Operations
Suppliers of estradiol cypionate powder should be evaluated for their strategic benefits to production. The compound's chemical stability lowers processing potency loss, waste, and rework. Estradiol cypionate may be compounded using normal equipment and methods with proper safeguards, unlike certain labile medicinal compounds.
The established regulatory process is another benefit. Estradiol cypionate products have thorough USP and EP monographs and a long regulatory history in key markets. This regulatory maturity streamlines documentation for manufacturers seeking new formulation or production site approvals.
The compound's defined synthesis pathways and internationally certified sources improve supply chain dependability. Second-source vendors offer procurement security that proprietary or freshly built APIs lack, but they require thorough validation. Competition in the supply chain enables cost control without compromising quality.
Clinical adaptability increases this API's formulation market potential. Manufacturers can create varied product portfolios from a single intermediate since applications cover multiple therapeutic areas. The multi-indication potential justifies process development, analytical methodologies, and regulatory documentation.
Many markets charge more for injections than oral medications due to their pharmacological complexity and patient convenience. Estradiol cypionate powder is favorable to firms seeking high-value pharmaceutical markets rather than generic markets due to this economic reality.
Important Considerations and Potential Limitations
Estradiol cypionate powder procurement and use limits must be considered for objective evaluation. Some chemical vendors lack the technical expertise and regulatory compliance to regularly supply pharmaceutical-grade hormone APIs.
Analytical technique validation is costly. Standard methodologies apply, but pharmaceutical producers must design and evaluate formulation matrix-specific stability-indicating methods. It needs thorough technique development and specificity to distinguish the intact ester from free estradiol and breakdown products.
Injectables have formulation restrictions that solid oral dose forms do not. Oil vehicle selection, multi-dose preservative systems, and main container compatibility must be considered. Compatibility tests on glass syringes, rubber stoppers, and closure mechanisms assure no extractables or leachables affect product quality.
Finished product requirements may need cold chain logistics. Some completed formulas require refrigerated storage to enhance shelf life, raw powder is stable at room temperature. For global supply chains, this demand affects distribution infrastructure and inventory management.
Regulatory monitoring of hormone products is growing worldwide. Pharmaceutical companies must have strong quality systems for raw material certification, in-process controls, completed product testing, and stability. Batch genealogies, impurity profiles, and manufacturing controls documentation must fulfill stricter regulatory requirements.
Hormone product regulations may limit market access in some places. When considering market entry and investment, manufacturers should consider regional regulations.
Estradiol Cypionate Powder versus Alternative Estrogen APIs
When choosing APIs for novel formulations, pharmaceutical development teams consider many choices. Comparing estradiol cypionate to alternatives reveals its benefit.
Estradiol valerate is the injectable formulation's main rival. Valerate ester effect is shorter and requires more frequent dosing. Some formulation scientists prefer this shorter period for commencement or tighter circulation level control. Maintenance regimens that prioritize convenience and compliance benefit from the cypionate ester's prolonged activity.
Another option with pharmacokinetics between valerate and cypionate is estradiol enanthate. The seven-carbon chain of the enanthate ester differs from cypionate's cyclopentylpropionate structure, affecting tissue retention. Regional preferences, previous market presence, and product positioning tactics influence enanthate or cypionate formulation.
The simplest basic analogy is oral estradiol. Oral delivery is convenient, but first-pass metabolism is insurmountable for hormone-dependent individuals. Injectable versions employing estradiol cypionate powder eliminate this constraint; they need injection and may cause injection site responses.
Another competitor is transdermal estradiol, delivered by patches or gels. They escape first-pass metabolism like injectables but require daily administration and might cause application site responses. The decision between transdermal and injectable approaches frequently depends on patient desire, lifestyle, and provider experience.
Estradiol cypionate powder's injectable method, prolonged duration, and regulatory status set it apart. This distinctive profile gives it a market position that meets patient demands better than alternatives.
Ideal Applications and Target Manufacturing Operations
Estradiol cypionate powder excels in some pharmaceutical production applications. Sterile injectable CDMOs with aseptic oil-based solution filling capabilities are the main customers. Estradiol cypionate and other injectable hormone medicines are made by these enterprises using existing equipment and knowledge.
This API helps mid-size specialist endocrinology pharmaceutical businesses differentiate their product ranges. The molecule allows long-acting formulations to complement oral medication lines, giving prescribers a variety of patient-specific alternatives.
Pharmacy compounding operations for specialty patients use pharmaceutical-grade estradiol cypionate powder to make bespoke formulas. Compounding regulations differ by jurisdiction, but a high-quality API is necessary for safe, effective formulations.
The compound's potency and ease of synthesis relative to newer hormone delivery systems assist international pharmaceutical producers serving emerging countries. These items are valuable in injectable administration markets with cold chain infrastructure.
Veterinary pharmaceutical businesses are emerging, but regulatory constraints and market dynamics differ from those in the human pharmaceutical industry. Estradiol cypionate's pharmacological qualities make it useful in human medicine and veterinary applications that need persistent estrogenic actions.
Pharmacokinetic, formulation development, and bioequivalence research companies need reference-grade material with analytical documentation. These consumers value Certificates of Analysis, impurity profiles, and material traceability over cost.
Conclusion
Estradiol cypionate powder occupies a specialized but significant position within pharmaceutical intermediate markets. Its unique pharmacokinetic properties enable long-acting injectable formulations addressing genuine patient needs for sustained hormone delivery without frequent dosing. Pharmaceutical manufacturers evaluating this API should prioritize supplier qualification, analytical capabilities, and regulatory support alongside pricing considerations. The compound's established clinical history, favorable stability profile, and formulation versatility make it a valuable component of diversified pharmaceutical portfolios targeting specialty endocrinology markets. As regulatory standards continue evolving globally, partnerships with quality-focused suppliers become increasingly critical for maintaining competitive advantage and ensuring an uninterrupted supply of this important pharmaceutical intermediate.
FAQ
Q1: What analytical documentation should accompany pharmaceutical-grade estradiol cypionate powder shipments?
A: Comprehensive documentation includes a Certificate of Analysis showing HPLC purity (typically ≥98%), identification testing via spectroscopic methods, related substances profile, residual solvents analysis, heavy metals testing, microbial limits, and physical properties including appearance, melting point, and specific rotation. Pharmaceutical manufacturers should also request batch manufacturing records, stability data, and reference standard information supporting the analytical methods employed.
Q2: How does storage condition affect the stability of estradiol cypionate powder?
A: Properly packaged estradiol cypionate powder stored at controlled room temperature (15-25°C) or refrigerated conditions (2-8°C), protected from light and moisture, typically maintains stability for 3-5 years. Exposure to elevated temperatures, humidity, or light accelerates oxidative degradation and ester hydrolysis. Manufacturers should implement appropriate environmental controls during warehousing and establish stability monitoring programs confirming continued conformance to specifications throughout the retest period.
Q3: What formulation factors most significantly influence the release kinetics of estradiol cypionate injections?
A: Vehicle composition exerts primary influence, with higher viscosity oils producing slower release rates. Concentration affects depot size and dissolution kinetics—higher concentrations create larger depots with extended release. Injection site selection matters due to blood flow differences between muscle groups. Co-solvents like benzyl benzoate modify solubility and diffusion characteristics. Pharmaceutical manufacturers must carefully control these parameters to achieve target pharmacokinetic profiles and ensure batch-to-batch consistency.
Q4: What are the critical quality attributes for qualifying a new estradiol cypionate powder supplier?
A: Pharmaceutical procurement teams should verify GMP compliance through site audits or third-party certifications. Analytical capabilities must include HPLC, GC, spectroscopy, and appropriate physical testing. Suppliers should demonstrate batch-to-batch consistency through comparative analytical data across multiple lots. Regulatory support, including Drug Master Files or Certificate of Suitability to European Pharmacopoeia, proves essential for regulated markets. Supply chain stability, including raw material sourcing transparency and manufacturing capacity, determines long-term viability as a strategic supplier.
Ready to Source Pharmaceutical-Grade Estradiol Cypionate Powder?
Securing a dependable Estradiol cypionate powder supplier capable of meeting GMP requirements remains crucial for pharmaceutical manufacturers developing long-acting injectable formulations. Xi'an Faithful BioTech Co., Ltd. provides pharmaceutical-grade Raw Estradiol Cypionate Powder supported by complete HPLC/GC analytical documentation, batch-specific Certificates of Analysis, and full traceability. Our quality control laboratory employs advanced analytical instrumentation, ensuring every batch meets stringent pharmaceutical specifications. Contact our technical team at allen@faithfulbio.com to discuss your API requirements, review documentation, and explore how Faithful can become your trusted estradiol cypionate manufacturer for a reliable long-term supply.
References
1. Kuhl H. (2005). Pharmacology of estrogens and progestogens: influence of different routes of administration. Climacteric, 8(Sup1), 3-63.
2. Stanczyk FZ, Archer DF, Bhavnani BR. (2013). Ethinyl estradiol and 17β-estradiol in combined oral contraceptives: pharmacokinetics, pharmacodynamics and risk assessment. Contraception, 87(6), 706-727.
3. Düsterberg B, Nishino Y. (1982). Pharmacokinetic and pharmacological features of oestradiol valerate. Maturitas, 4(4), 315-324.
4. Schindler AE, Campagnoli C, Druckmann R, Huber J, Pasqualini JR, Schweppe KW, Thijssen JH. (2003). Classification and pharmacology of progestins. Maturitas, 46, S7-S16.
5. Fotherby K. (1996). Bioavailability of orally administered sex steroids used in oral contraception and hormone replacement therapy. Contraception, 54(2), 59-69.
6. Thorneycroft IH, Stanczyk FZ, Bradshaw KD, Ballagh SA, Nichols M, Weber ME. (1999). Effect of low-dose oral contraceptives on androgenic markers and acne. Contraception, 60(5), 255-262.



