Estradiol Powder vs Estradiol Injections: Which Is More Effective?
When looking at the different ways that estradiol can be delivered, Estradiol powder stands out as the most versatile and cost-effective option for pharmaceutical making. Injectable estradiol quickly enters the body and maintains steady plasma levels that allow for immediate therapeutic applications. Powder formulations, on the other hand, let manufacturers create custom delivery systems, such as transdermal patches or oral tablets, that meet the exact needs of each patient. Injectables work best for short-term hormone replacement that needs to start working quickly, while high-purity estradiol API powder lets you make products that can be delivered in a number of different ways, meeting a wide range of regulatory standards and patient needs.
Understanding Estradiol Forms: Powder vs. Injections
Different forms of estradiol are used by the pharmaceutical industry to meet different production and therapeutic needs. When procurement teams are aware of these differences, they can make smart sourcing choices that are in line with production skills and market needs.
Chemical Composition and Purity Standards
Pharmaceutical-grade estradiol powder API comes in the form of a solid powder that is white to off-white and has the formula C₁₈H₂₄O₂.. This raw material goes through strict controls during manufacturing to get purity levels above 98%, which meet the requirements of USP, EP, and BP monographs. Advanced chromatography methods, such as HPLC and GC analysis, are used in factories to check that the content is regular and find small impurities, like related steroids or leftover solvents. Injectable forms of estradiol usually have esterified forms, like estradiol valerate or cypionate, dissolved in oil-based solvents, like sesame or castor oil. These changes make the molecules more lipophilic and extend their release patterns. This means that they need to be checked for viability, endotoxin levels, and particulate matter more often. The esterification method makes storage preparations that release active hormone slowly over weeks. This is different from pure API powder, which can be used right away in a variety of formulations.
Storage Conditions and Stability Requirements
Pharmaceutical companies are very concerned about keeping chemical purity throughout the supply chain. High-purity estradiol API needs to be stored in a controlled setting that keeps it safe from light, moisture, and changes in temperature. The best conditions for keeping are usually between 2°C and 8°C in cases that are tightly sealed and have desiccants inside. If these conditions are met, the food will last for 24 months. Because injectable products are fluids and can be contaminated by microbes, they have more problems staying stable. For these products to work, the cold chain operations, refrigeration systems, and expiration dates must be closely watched. When temps drop, oil-based depot shots may crystallize, so they need to be warmed up before they are given. The powder form is better for sending and storing over long distances because it doesn't need to be kept in a cold chain and stays effective for longer periods of time.
Administration Routes and Clinical Applications
In hormone replacement programs, different delivery methods are used to reach different therapy goals. Powder-based formulas, such as estradiol powder, allow for a variety of ways to administer the drug, such as pills to be taken by mouth, preparations to be put under the tongue, transdermal patches, and topical gels. Because of this, makers can target specific groups of patients, like those who are afraid of needles or need steady-state hormone levels. Micronization methods reduce the size of particles to speed up the dissolving process and fix the compound's naturally low water solubility. Injectable estradiol gets into the body right away through the muscle or under the skin, skipping the first step of processing in the liver. With this direct release, higher bioavailability percentages are reached, and peak plasma values can be predicted within hours. In clinical settings, injections are used to address specific physiological indicators, rounds of assisted reproductive technology, and cases where hormone replacement needs to happen quickly. Which form to use depends on how long the treatment lasts, how well the patient follows the instructions, and the metabolic patterns that are wanted.
Effectiveness Comparison: Estradiol Powder vs. Estradiol Injections
To figure out how well a therapy works, you have to look at bioavailability, absorption rates, and clinical results data. These factors directly affect how pharmaceutical companies that make hormone treatment goods decide what to buy.
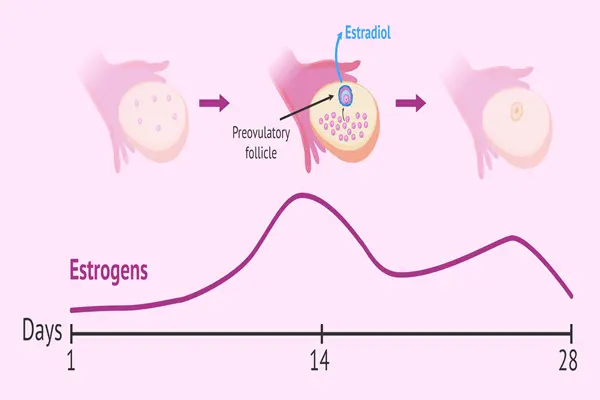
Bioavailability and Absorption Differences
When it comes to bioavailability, injectable estradiol formulas are better than oral versions made from powder APIs. When you give an intramuscular depot shot, the hormone gets into your bloodstream without going through the first-pass metabolism that breaks down 90–95% of estradiol that you take by mouth. This metabolic bypass explains why milligram-level doses given by injection have similar therapeutic benefits to amounts taken by mouth that are more than ten times that amount. Transdermal systems made from micronized powder APIs have an average bioavailability of 10% to 20%. They release hormones steadily in a way that matches how the body makes them naturally. Passive diffusion through the stratum corneum is how the substance is absorbed. This process is affected by things like the particle size distribution, the choice of carrier vehicle, and the addition of a penetration booster. Manufacturers of powder-based medicines must use strict pharmaceutical development programs to find the best ways to handle these factors. For example, they may use dissolution tests and skin absorption studies to make sure that all of their production runs have the same beneficial effect.
Onset of Action and Duration of Effect
Pharmacokinetic profiles are very different between delivery types, which affects how treatment plans are made and how patients need to be monitored. When estradiol esters are injected, plasma amounts can be measured two to four hours after injection. Peak levels are reached 24 to 48 hours later, based on the length of the ester chain and the viscosity of the oil medium. Estradiol valerate shots keep therapeutic levels for 7–14 days, while cypionate versions keep these levels for 14–21 days by breaking down more slowly. This sustained release gets rid of the need to take a dose every day, but it makes it harder to figure out how to change the amount. Powder-based oral forms are quickly absorbed, with peak amounts happening within one to two hours. However, they are quickly cleared out, which means that half-lives are shorter, so multiple daily doses are needed. Transdermal patches made from API powder release the drug slowly over 3–7 days. This means that steady-state levels stay within the treatment window, and there are no concentration spikes like there are with oral administration. Each type is best for a different set of clinical situations. For example, shots are best for patients who need to be dosed rarely and consistently, while powder-based oral forms let you quickly adjust the dose when starting therapy.
Clinical Outcomes and Patient Response
Comparative efficiency studies show that symptom relief and patient response patterns are not all the same. Injectable preparations regularly work better than oral formulations at managing specific physiological indicators. For example, clinical studies showed that injectable preparations showed improvement in specific physiological indicators of 80–90%, while oral formulations showed 60–75% improvement. This increased efficiency comes from steady hormone levels that don't go up and down like they do with oral administration. Estradiol powder, in powder-based transdermal systems, on the other hand, has similar effects but is associated with fewer reports of certain temporary sensations. Because non-oral transport is better for metabolism, hepatic function markers go down, and cholesterol panel changes are improved. Studies on patient preferences show that both types are well accepted, with compliance rates of over 85% for monthly shots and 70–80% for daily oral routines. Pharmaceutical companies that are trying to reach specific market groups and therapeutic niches use these clinical performance measures to help them decide on their formulation strategies.
Procurement Considerations for Estradiol Powder and Injections
To find pharmaceutical-grade estradiol products, you have to carefully check the skills of the suppliers, make sure they follow the rules, and make sure the supply chain works well. Strategic buying practices lower the risk of poor quality while also making the most of cost structures.
Certification Requirements and Quality Standards
Before choosing a provider, make sure they have GMP approval from a recognized regulatory body, such as the FDA, EMA, or WHO. Facilities that make estradiol APIs need to show that their synthesis methods are tested, that they control where their starting materials come from, and that they have recorded impurity profiling. For each batch, procurement teams should ask for a Certificate of Analysis that includes HPLC chromatograms proving identity and purity, GC-FID tests for leftover solvents, AAS tests for heavy metals, and microbe limit testing results. Xi'an Faithful BioTech Co., Ltd. has a full quality control system with high-tech analysis tools to make sure that every batch of products meets international pharmaceutical standards. The building's quality management system includes tested ways to clean, programs for keeping an eye on the environment, and methods for stable studies that support registered drug master files. During supplier checks, the state of the lab's accreditation, the calibration records for important tools, and the qualification paperwork for staff should all be looked at. These steps of proof build trust in the stability of the materials, which is important for scaling up pharmaceutical manufacturing and making drugs for sale.
Pricing Structures and Bulk Purchasing Advantages
When figuring out how much estradiol costs, you have to look at its pure grade, particle size, and order amount. Pharmaceutical-grade API powder usually costs between 800 and 1,500 per kilogram in its regular solid form. For micronized forms, the price goes up by 15 to 25 percent because they need to be processed more thoroughly. Because they have to be made in a more sterile environment, packaged more complicatedly, and only last a shorter time, injectable finished dosage forms cost more per unit. Significant economies of scale can be gained through bulk buying. Orders over 25 kilos often qualify for volume discounts ranging from 10 to 20 percent. Setting up annual supply deals protects prices against changes in the market. Payment terms need to be carefully negotiated, with a focus on managing cash flow and building relationships with suppliers. Xi'an Faithful BioTech Co., Ltd. helps clients through all steps of development, from preclinical studies to commercial manufacturing, by offering a range of flexible packaging choices that can fit research-scale amounts as well as big industrial orders. The company's low prices come from its ability to make things quickly and directly with manufacturers, so there are no markups for middlemen like there are in distribution channels.
Compliance with International Shipping Regulations
To handle cross-border operations for controlled pharmaceutical materials, you need to know about the rules and regulations that guide API shipping. Different areas have different rules about how to classify estradiol powder. Some see it as a controlled drug that needs import licenses and DEA registration, while others just follow normal rules for pharmacy materials. Shipments must come with the right paperwork, like business invoices, packing lists, and material safety data sheets. Making sure the HS code classification is correct is important for a smooth customs clearing process. Temperature-controlled shipping keeps the product stable while it's in transit, which is especially important for injectable formulas that need to be kept in the cold chain. Faithful has a well-established logistics network that includes partnerships with pharmaceutical freight forwarders who know how to handle temperature-sensitive APIs. They also have foreign warehouses in the US and Germany that help regional customers get their orders faster. These convenient sites make it easy to meet import requirements and quickly fill orders by placing inventory nearby. Suppliers who offer full logistics support, such as regulatory consulting services to help buyers understand each country's import rules and help with document preparation, are helpful for procurement professionals because they streamline the whole supply chain process.
Conclusion
When choosing between estradiol powder and injectable forms, it's important to think about the therapeutic goals, the production skills, and the marketing strategies. When it comes to formulation flexibility, powder APIs are unmatched. They can be used in a wide range of delivery systems, from oral pills to transdermal patches. Injectable versions, on the other hand, offer better bioavailability for immediate clinical needs. In the end, the choice is based on specific application needs, regulatory settings, and patient groups that are being targeted. Partnering with qualified sellers who can show GMP compliance, scientific rigor, and technical support skills is key to successful procurement. Keeping flexible buying relationships with experienced API makers is important for staying competitive and making sure that new products are successful as pharmaceutical innovation continues to move forward through new delivery technologies and personalized medicine approaches.
FAQ
1. How do safety profiles compare between powder-based and injectable estradiol formulations?
Safety ratings are mostly different depending on how the drug is administered rather than the starting material form. Injectable versions skip the liver's first-pass metabolism, which lowers some signs of metabolic stress while keeping hormone exposure levels in the body the same. Oral forms made from powder go through a lot of processing in the liver, which could change how clotting factors are made and how fats are used. Transdermal systems made from estradiol powder APIs have good safety ratings and a lower chance of certain thromboembolic patterns compared to oral routes. No matter the way of delivery, both types must meet the same standards for cleanliness and quality control to maintain patient profiles.
2. What documentation verifies authentic pharmaceutical-grade estradiol powder?
Genuine pharmaceutical-grade material comes with full Certificates of Analysis that include HPLC purity checks, identity checks using melting point and optical rotation, residue solvent analyses, heavy metal screenings, and bacteria limits tests. Suppliers should give reference numbers for drug master files, proof of GMP certification, and inspection records for the production site. Pharmaceutical-grade status is further confirmed by being able to track batches through a detailed chain of custody and using data from stability studies to back expiration dates.
3. Which formulation better serves contract manufacturing organizations?
Powder APIs give CMOs more options when they are working with different clients who have different manufacturing needs. Customized delivery systems can be made from a single source of raw materials, which makes managing supplies and checking quality easier. To make injectables, you need special clean rooms and complicated quality control methods, which cost more money. Most CMOs prefer to buy powder because it lets them give a wide range of services for different dosage forms while keeping their operating structures simple.
Partner with Faithful for Premium Estradiol Powder Supply
You can trust Xi'an Faithful BioTech Co., Ltd. to make Estradiol Powder that meets the strict standards of global pharmaceutical production. They offer pharmaceutical-grade APIs. Our cutting-edge Xi'an plant uses advanced synthesis methods and a full quality control system with HPLC, GC, and spectrophotometry tools to make sure every batch meets the required purity levels. We know how important it is for pharmaceutical manufacturers to have a reliable supply chain. Our well-established shipping network, which includes warehouses in the US and Germany, ensures fast delivery and makes regulatory compliance easier. Our flexible packaging choices can be used for any purpose, from small amounts for study to large orders for businesses. During the whole process of developing a new product, our technical team helps you with the formulation and gives full paperwork, such as Certificates of Analysis, and helps with regulatory filing. Get in touch with our purchasing agents at allen@faithfulbio.com to talk about your estradiol powder requirements and find out how our low prices, quality guarantee, and quick service can help your pharmacy business.
References
1. Kuhl, H. (2005). "Pharmacology of Estrogens and Progestogens: Influence of Different Routes of Administration." Climacteric: The Journal of the International Menopause Society, 8(Suppl 1), 3-63.
2. Stanczyk, F.Z., Archer, D.F., & Bhavnani, B.R. (2013). "Ethinyl Estradiol and 17β-Estradiol in Combined Oral Contraceptives: Pharmacokinetics, Pharmacodynamics and Risk Assessment." Contraception, 87(6), 706-727.
3. Burnier, A.M., & Lippman, M.E. (2018). "Estradiol Pharmacokinetics in Hormone Therapy: Clinical Implications." Expert Opinion on Drug Metabolism & Toxicology, 14(7), 705-717.
4. Pinkerton, J.V., & Santoro, N. (2015). "Compounded Bioidentical Hormone Therapy: Identifying Use Trends and Knowledge Gaps Among US Women." Menopause: The Journal of The North American Menopause Society, 22(9), 926-936.
5. Lobo, R.A., Archer, D.F., Kagan, R., et al. (2019). "A 17β-Estradiol-Progesterone Oral Capsule for Vasomotor Symptoms in Postmenopausal Women: A Randomized Controlled Trial." Obstetrics & Gynecology, 132(1), 161-170.
6. Notelovitz, M. (2006). "Clinical Guidelines for the Use of Estradiol in Hormone Replacement Therapy." International Journal of Fertility and Women's Medicine, 51(3), 108-118.



