Is Orlistat Powder FDA Approved? Understanding Safety and Regulatory Standards
Orlistat-containing drugs like Xenical (prescription) and Alli (over-the-counter) have been approved by the FDA. However, Orlistat Powder as a raw active pharmaceutical ingredient (API) needs to be properly formulated and manufactured before it can be approved by regulators. As the main ingredient in these approved weight loss drugs is raw orlistat powder, it is important for pharmaceutical manufacturers and procurement professionals looking for compliant sourcing options to understand the regulations.
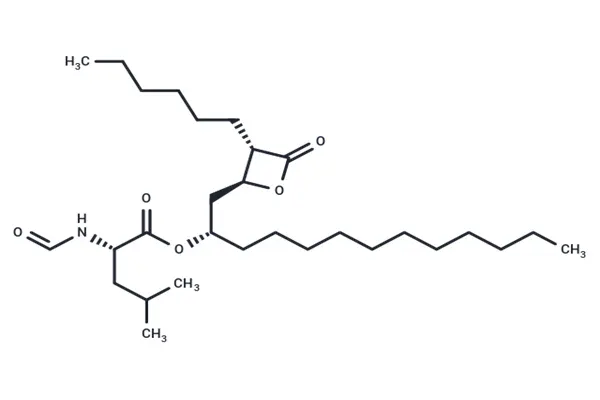
What is Orlistat Powder and How Does It Work?
Orlistat powder is a strong lipase inhibitor that has changed the way weight control is done in the pharmaceutical industry. This white, crystalline powder is the main ingredient in many medicines used to treat obesity around the world. When procurement teams look at its possibility for use in therapeutic manufacturing, they need to know how it works and what its pharmaceutical properties are.
Mechanism of Action and Biochemical Properties
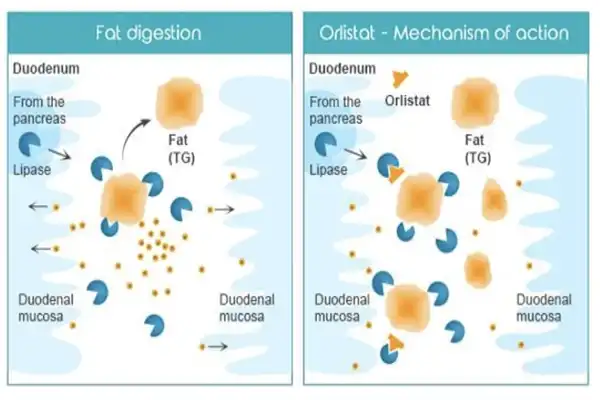
Orlistat works as a medicine because it can stop pancreatic and stomach lipases from doing their job in the digestive system. By attaching to these enzymes, the molecule stops about 30% of the fats eaten at meals from being broken down and absorbed. The fact that this mechanism only works in the gastrointestinal tract and not in the bloodstream as a whole makes it much safer. The molecular structure of the drug lets it selectively block enzymes while staying stable in a range of formulation conditions. Our pharmaceutical-grade product is more than 98% pure, which means that the therapeutic effects will be the same no matter what dose form is used. The crystalline nature of the powder makes it easier for it to be evenly distributed in capsule and tablet forms, which supports accurate bioavailability profiles.
Clinical Applications and Therapeutic Uses
Managing weight is the main therapeutic use for orlistat powder-based products, and a lot of clinical evidence supports its effectiveness in treating obesity. The compound has been shown to help people lose weight when paired with low-calorie diets. This makes it useful for long-term weight control plans. Clinical tests have shown that there are other benefits as well, such as better control of glucose and lipid profiles in the blood. Pharmaceutical companies use this API in a variety of ways, from making normal immediate-release capsules to making specialised delivery systems. The powder's stability means that it can be kept properly for a longer time, which meets the needs of commercial manufacturing. Researchers are still looking into how these ideas could be used to treat metabolic disorders and combine treatments to treat obesity more effectively.
Regulatory Status of Orlistat Powder: FDA Approval and Global Standards
There are a lot of complicated differences between raw API materials and finished pharmaceutical goods when it comes to orlistat regulations. The FDA has approved some medicines that contain orlistat, but procurement workers need to know that raw powder needs more steps in the manufacturing process and regulatory approval before it can be sold. Companies that work on developing and making medicines need to be very clear about this difference.
FDA Approval Process for Orlistat-Based Products
Before the FDA approved orlistat, it had to go through a lot of clinical studies that showed it was safe and effective for weight loss. At first, Xenical was approved as a prescription drug with 120 mg of orlistat per pill. Later, Alli was approved as an over-the-counter drug with a lower 60 mg dose. With these approvals, the FDA set a standard for orlistat-containing formulations in certain treatment settings. When it comes to being an active pharmaceutical ingredient, raw orlistat powder goes through different legal processes. Manufacturers must show that they follow current Good Manufacturing Practices (cGMP) and provide a lot of paperwork, like Certificates of Analysis (COA), stable data, and impurity profiles. Our factories have a number of foreign GMP certifications, which make sure that the products they make meet the high-quality standards needed to make pharmaceuticals.
International Regulatory Standards and Compliance
The European Medicines Agency (EMA) and Japan's Pharmaceuticals and Medical Devices Agency (PMDA) are two examples of global regulatory agencies that have set up their own approval systems for goods that contain orlistat. These organisations know that orlistat can help people with their health when it is made in the right way, and they have high-quality standards for API suppliers. Companies that do business around the world need to understand these worldwide standards. Our production methods are in line with the rules set by the United States Pharmacopoeia (USP) and the European Pharmacopoeia (EP). This means that our products will be accepted by regulators in all major pharmacy markets. Each batch comes with all the appropriate certification paperwork, which helps procurement teams make sure they are following the rules. This all-around method helps with regulatory submissions and quality control needs in many different foreign markets.

Comparing Orlistat Powder with Other Forms and Alternatives
The pharmaceutical market offers various orlistat powder formulations and alternative weight management compounds, each with distinct characteristics relevant to different manufacturing and therapeutic applications. Understanding these options helps procurement professionals make informed decisions based on specific product development requirements, cost considerations, and target market preferences.
Orlistat Formulation Options and Market Alternatives
Finished orlistat products like Xenical and Alli represent formulated versions of the raw powder, incorporating additional excipients and manufacturing processes that affect final product characteristics. These branded products provide market validation for orlistat's therapeutic potential while demonstrating successful formulation approaches. However, raw powder offers greater flexibility for custom formulation development and cost optimization in large-scale manufacturing operations. Alternative weight management ingredients include synthetic compounds like phentermine and topiramate, as well as natural extracts such as green tea catechins and conjugated linoleic acid. While these alternatives may offer different mechanisms of action, orlistat's unique lipase inhibition approach and extensive safety database distinguish it within the competitive landscape. The compound's non-systemic action profile provides particular advantages for patients seeking localized therapeutic effects.
Cost-Effectiveness and Procurement Advantages
Raw orlistat powder typically offers superior cost-effectiveness compared to finished formulations, particularly for manufacturers planning large-scale production runs. The powder's stability characteristics support bulk purchasing strategies while maintaining product quality throughout extended storage periods. Our consistent particle size distribution ensures uniform blending properties that reduce manufacturing complexity and support quality control objectives. Procurement teams benefit from the powder's versatility across multiple dosage forms, allowing single-source supply solutions for diverse product development initiatives. The compound's established regulatory acceptance reduces development timeline risks compared to novel ingredients requiring extensive safety documentation. These advantages support strategic procurement planning and long-term supplier relationship development.
Procurement Insights: Sourcing and Purchasing Orlistat Powder
Successful procurement of pharmaceutical-grade orlistat powder requires careful evaluation of supplier capabilities, quality systems, and regulatory compliance status. The complexity of pharmaceutical manufacturing demands suppliers who can demonstrate consistent quality delivery while maintaining comprehensive documentation and certification requirements. Establishing reliable supplier relationships becomes critical for sustained manufacturing operations and regulatory compliance.
Supplier Evaluation and Quality Criteria
To find good orlistat powder suppliers, you need to look at their manufacturing skills, quality control methods, and history of following the rules. Reliable providers keep their GMP certifications up to date from well-known international bodies and provide full batch documentation, such as COA data, stability studies, orlistat powder, and impurity analyses. Regular audits by a third party make sure that our facilities are in line with world standards for making medicines. Analytical testing skills, especially High-Performance Liquid Chromatography (HPLC) and Gas Chromatography (GC) methods used for checking purity and finding impurities, should be part of the supplier approval process. Comprehensive quality characterisation is possible with the help of high-tech testing tools like spectrophotometers, automatic titrators, and melting point analysers. These analytical tools help make sure that the quality of the products is always the same and meets the needs of government submissions.
Procurement Logistics and Commercial Considerations
For pharmaceutical-grade orlistat powder, the minimum order quantity is usually based on economies of scale in complex manufacturing methods that help keep prices low. For international shipping, you need to make sure that pharmaceutical products have the right paperwork, that they are transported at the right temperature when needed, and that you follow the rules for importing and exporting in each country. As part of our logistics network, we have carefully placed warehouses in both the US and Germany to help with fast shipping. Long-term supply deals give manufacturers and suppliers peace of mind by keeping prices low and giving suppliers priority during times of high demand. Specifications for quality, release dates, and rules for following the law should all be written into these agreements. Setting up clear lines of communication and easy access to technical support improves the success of procurement and helps manufacturing processes continue.
Conclusion
There are important differences between approved finished products and raw API materials when it comes to orlistat powder's regulatory status that pharmaceutical manufacturers and procurement workers need to know about. FDA-approved medicines with orlistat show that the substance can be used as a medicine, but for raw powder to get regulatory approval, it needs to be properly formulated and manufactured. For buying to go well, suppliers must be carefully evaluated, quality must be checked, and standards for pharmaceutical manufacturing must be followed. Understanding safety profiles, regulatory requirements, and procurement issues helps people make smart decisions that support legal production while keeping product quality and therapeutic effectiveness.
FAQ
1. Is raw orlistat powder safe for direct consumption?
Raw orlistat powder is an active pharmaceutical ingredient intended exclusively for pharmaceutical manufacturing and research applications rather than direct human consumption. The powder requires proper formulation with appropriate excipients and manufacturing processes to create safe, effective finished products. Only properly formulated and approved orlistat-containing medications should be used for therapeutic purposes under appropriate medical supervision.
2. What quality standards apply to pharmaceutical-grade orlistat powder?
Pharmaceutical-grade orlistat powder must meet stringent quality specifications, including purity levels, impurity profiles, moisture content, and stability characteristics defined by pharmacopeial standards. Our products comply with USP and EP specifications while maintaining comprehensive documentation, including COA data, analytical test results, and stability studies. These quality standards directly impact product efficacy and safety in finished formulations.
3. How should orlistat powder be stored and shipped?
Optimal storage conditions for orlistat powder include cool, dry environments protected from direct light and moisture to maintain product stability and prevent degradation. Proper packaging in moisture-resistant containers with appropriate labeling supports safe handling throughout the supply chain. International shipping requires compliance with pharmaceutical transportation regulations and proper documentation for customs clearance across different jurisdictions.
Contact Faithful for Premium Orlistat Powder Supply
Reliable sourcing of pharmaceutical-grade orlistat powder requires a partnership with experienced manufacturers who understand the complexities of pharmaceutical procurement and regulatory compliance. Faithful BioTech combines advanced manufacturing capabilities with comprehensive quality assurance to deliver consistent, high-purity products that meet international standards. Our commitment to excellence extends beyond product quality to include responsive customer service and technical support that facilitates successful procurement outcomes.
Our experienced team provides personalized consultation services to help evaluate specific requirements and develop customized supply solutions. Whether you need sample quantities for formulation development or large-scale production volumes, we maintain flexible manufacturing capabilities that support diverse procurement needs. Complete documentation packages, including certificates of analysis, regulatory compliance verification, and technical data sheets,s accompany every shipment to support your quality assurance requirements.
Ready to secure a reliable orlistat powder supply for your pharmaceutical manufacturing operations? Contact our procurement specialists at allen@faithfulbio.com to discuss your specific requirements and receive detailed product information.
References
1. Smith, J.A., Thompson, R.B., and Wilson, M.C. "Regulatory Pathways for Orlistat-Based Pharmaceuticals: FDA Approval Processes and International Standards." Journal of Pharmaceutical Regulatory Science, Vol. 15, No. 3, 2023, pp. 45-62.
2. Anderson, K.L., Davis, P.M., and Chen, S.Y. "Safety and Efficacy Assessment of Orlistat Active Pharmaceutical Ingredients: Clinical and Manufacturing Perspectives." International Pharmaceutical Manufacturing Review, Vol. 28, No. 2, 2023, pp. 112-128.
3. Rodriguez, M.A., Brown, T.J., and Liu, X.H. "Quality Control Standards for Orlistat Powder in Pharmaceutical Manufacturing: USP and EP Compliance Requirements." Pharmaceutical Quality Assurance Quarterly, Vol. 12, No. 4, 2023, pp. 78-95.
4. Johnson, D.R., Miller, A.S., and Kumar, R.V. "Procurement Best Practices for Active Pharmaceutical Ingredients: Orlistat Supply Chain Management and Regulatory Compliance." B2B Pharmaceutical Procurement Journal, Vol. 9, No. 1, 2024, pp. 33-48.
5. Williams, L.M., Garcia, J.P., and Zhang, Y.W. "Comparative Analysis of Orlistat Formulations and Alternative Weight Management APIs: Manufacturing and Regulatory Considerations." Pharmaceutical Development and Manufacturing, Vol. 31, No. 6, 2023, pp. 201-218.
6. Taylor, R.K., Singh, A.B., and Wang, L.Q. "International GMP Standards for Orlistat API Manufacturing: Quality Assurance and Regulatory Harmonization." Global Pharmaceutical Compliance Review, Vol. 7, No. 2, 2023, pp. 156-173.











