Is Pepsin Enzyme Powder the acidic activated form of pepsinogen?
Imbalances in digestive and absorptive functions and weakened gastrointestinal metabolism have long been a focus of the digestive health pharmaceutical industry. Acidic hydrolytic enzymes, with their physiological homology, mild effects, and compatibility with the gastrointestinal environment, have become core ingredients in stomach-strengthening and digestive aid formulations. Pepsin Enzyme Powder is an aspartic protease powder refined and extracted from animal gastric mucosa. Utilizing its specific activity in the highly acidic environment of the stomach, it selectively hydrolyzes large protein molecules, reducing the digestive burden on the gastrointestinal tract and maintaining its microenvironmental homeostasis. The raw material boasts stable physicochemical properties, controllable enzyme activity, and strong compatibility with excipients, fully complying with pharmaceutical-grade production standards.
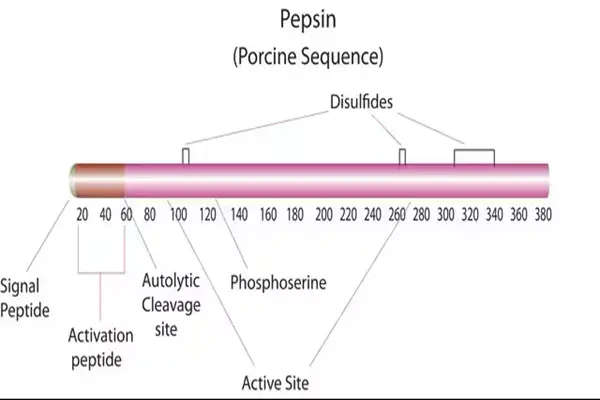
The unique spatial folding and structural characteristics of sex proteases
Pepsin Enzyme Powder belongs to the aspartic protease family. It is naturally synthesized as an inactive prozymogen and spontaneously cleaves and activates under acidic gastric conditions, forming a mature monomeric protein capable of hydrolysis. The overall molecule has a compact, globular, single-chain structure with an average relative molecular mass of approximately 35 kDa. Its regular peptide chain folding is maintained by hydrogen bonds and hydrophobic interactions, creating a spatial conformation adapted to acidic environments. The finished product is a uniform, off-white, fine powder with a loose, non-clumping texture. Pharmaceutical-grade purification processes significantly improve the purity of the active ingredient, with strict control over impurities such as extraneous proteins, pyrogens, and microbial contaminants, ensuring stable and compliant overall quality.
The most crucial structural feature of this enzyme molecule lies in the combination of two aspartic residues at the active site. This specific amino acid arrangement forms an embedded substrate-binding groove, allowing it to specifically bind to large globular proteins and fibrin, thus determining the substrate recognition specificity at the structural level. The overall molecule is acid-resistant but not alkali-resistant. It maintains a stable conformation in the gastric juice environment (pH 1.0–3.5). However, upon entering the neutral or slightly alkaline intestinal environment, its spatial structure naturally expands and becomes inactive, creating a natural limitation on its range of action and preventing indiscriminate decomposition of the body's own tissue proteins.
Pepsin Enzyme Powder, after low-temperature drying and purification, exhibits high peptide chain integrity, stable enzyme activity retention, and a long shelf life when stored at room temperature in a sealed, light-protected environment. It is not prone to spontaneous denaturation or degradation. The uniform distribution of hydrophilic groups on the molecular surface allows for rapid dispersion and dissolution in acidic aqueous systems, making it suitable for the production and formulation of oral solid dosage forms and acidic liquid dosage forms. Compared to microbial proteases, the amino acid sequence of animal-derived natural pepsin is more closely aligned with human digestive physiology, resulting in stronger molecular affinity and more stable efficacy.
Industrial purification processes, such as membrane separation and low-temperature precipitation, remove impurities and harmful residues, retaining highly active functional segments and reducing the proportion of ineffective components. The molecule has no complex cross-linked structure and moderate flexibility, allowing it to maintain its functional form and continue its hydrolytic activity even in the dynamic environment of gastric peristalsis and slight fluctuations in acidity and alkalinity. Its simple and compact protein backbone design enhances its processing adaptability, enabling it to be mixed with various pharmaceutical excipients and molded for large-scale pharmaceutical production.
Acidic environment-directed hydrolysis and digestion homeostasis regulation
The core operating logic of Pepsin Enzyme Powder relies on the highly acidic microenvironment of the stomach to activate and take effect. Through a gentle and targeted peptide bond hydrolysis mode, it breaks down large, difficult-to-digest protein molecules, streamlining the stomach's metabolic rhythm and improving functional imbalances caused by insufficient digestive motility. Unlike broad-spectrum hydrolysants, it has a specific target and limited area of action, specifically addressing the shortcomings in acidic digestion without interfering with the normal metabolic processes of carbohydrates and lipids.
After entering the acidic environment of the stomach, pepsinogen rapidly loses its inhibitory peptides and transforms into highly active pepsin. It precisely identifies the hydrophobic peptide bonds and aromatic amino acid linkage sites of large protein molecules in food. By breaking down key linkages in long-chain proteins, it gradually breaks down intact macromolecules into peptides, oligopeptides, and small molecule fragments, significantly reducing the metabolic load on subsequent small intestinal digestion, improving overall nutrient breakdown and absorption efficiency, and alleviating the gastrointestinal burden caused by protein accumulation.
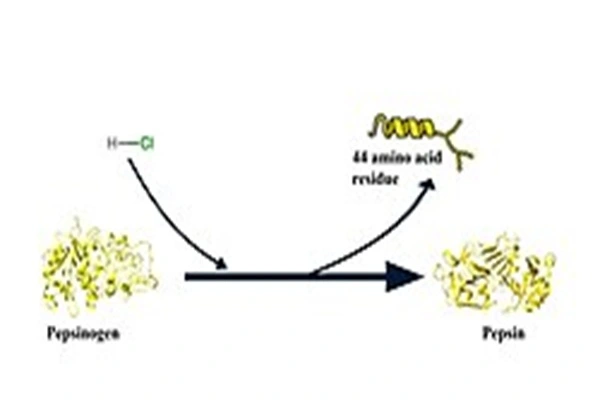
For those with insufficient endogenous digestive enzyme secretion, exogenous supplementation with this powder can directly fill the gap in acidic digestive enzymes. As we age, experience gastric mucosal damage, or suffer from long-term irregular eating habits, the body's own synthesis of pepsin declines. Incompletely broken-down proteins tend to remain and ferment in the stomach, leading to symptoms such as bloating, belching, and decreased appetite. This ingredient can provide long-lasting support for pepsin metabolism, gradually restoring a regular digestive rhythm.
Simultaneously, this ingredient gently regulates the gastric metabolic microenvironment, reducing irritating metabolites from undigested protein accumulation, alleviating chronic infiltration pressure on the gastric mucosa, and indirectly maintaining the integrity of the gastric mucosal barrier. Its effectiveness is autonomously regulated by the pH of the environment; the higher the gastric acid concentration, the stronger the activity, while the effect is milder when gastric acid is low. It exhibits excellent physiological adaptability, perfectly aligning with the body's natural digestive regulation.
The entire regulatory mechanism is gentle and reversible, without strong stimulation or excessive metabolic consumption. It automatically inactivates upon entering the neutral intestinal environment, without interfering with the normal intestinal protease system. This environmentally adaptive mode of action allows Pepsin Enzyme Powder to achieve targeted onset and time-limited action, balancing therapeutic value with physiological safety, and meeting the pharmaceutical application needs for long-term, gentle digestive intervention.
A versatile tool in food digestion and biopharmaceuticals
The most mature and widely used applications of Pepsin Enzyme Powder are in the food industry and biopharmaceutical field, its versatility stemming from its unique acidic proteolytic ability.
- First, in in vitro digestion models, Pepsin Enzyme Powder is a globally recognized standard enzyme for "gastric digestion." According to the experimental protocol published by Sigma-Aldrich, pepsin is dissolved in hydrochloric acid or 0.1M glycine-hydrochloric acid buffer at pH 1.0-2.0, the food sample to be digested is added, and the mixture is incubated in a 37°C water bath for 1-4 hours. The reaction is then terminated by adjusting the pH to 7.0 with sodium bicarbonate or sodium hydroxide. This simulation system is widely used to assess food bioavailability, degrade pesticide residues, and study the potential of bioactive peptides released during digestion.
- Second, in diagnostic reagent production, Pepsin Enzyme Powder is one of the core enzymes used in pathological and immunohistochemical staining. It is used to digest antigens in fixed, paraffin-embedded tissue sections, exposing epitopes by hydrolyzing cross-linked proteins, enhancing the signal of immunohistochemical staining, and aiding pathologists in differential diagnosis. In fecal occult blood immunochemical detection kits, Pepsin Enzyme Powder is also used to lyse hemoglobin in fecal samples, exposing hidden antigenic epitopes for capture and detection by specific antibodies.
- Third, in the biopharmaceutical field, the most classic use of Pepsin Enzyme Powder is in the preparation of F(ab')₂ antibody fragments. Pepsin cleaves IgG at the C-terminus of the disulfide bond in the hinge region, removing the entire Fc fragment and retaining two Fab fragments linked by disulfide bonds. This cleavage product has a molecular weight of approximately 100 kDa, retaining antigen-binding ability but losing Fc-mediated effector function, making it uniquely valuable in immunoassay and targeted drug delivery. This "de-Fcification" is an irreplaceable application of Pepsin Enzyme Powder in the field of immunological reagents.
- Fourth, in the food processing field, Pepsin Enzyme Powder is used as a protein-hydrolyzing enzyme to improve the nutritional and functional properties of products. It is also used in the production of hypoallergenic infant formula by partially hydrolyzing allergenic epitopes in milk proteins, thus reducing their immunogenicity. In beer clarification processes, pepsin is used to remove protein-polyphenol complexes that can form turbidity, improving product stability and transparency.
- Fifth, in scientific research, Pepsin Enzyme Powder is widely used in collagen extraction and tissue engineering research. It has been used to increase the proportion of extractable soluble collagen and reduce the immunogenicity of collagen derived from bovine dermal tissue. In cell biology experiments, Pepsin Enzyme Powder is also used to process cell samples, and in veterinary pathology studies to digest dried cervical samples for subsequent analysis.
Enzyme activity optimization and formulation delivery technologies continue to be upgraded
As digestive medicines evolve towards more natural, gentle, precise, and long-lasting effects, Pepsin Enzyme Powder's industrial upgrade is steadily progressing, focusing on purification process improvement, stability modification, dosage form innovation, and refined application expansion.
Green low-temperature extraction processes are continuously optimized, replacing traditional high-temperature processing methods. This maximizes the preservation of the protein's natural spatial conformation and enzyme activity, reduces impurities and allergenic residues, and improves the purity and safety of the active pharmaceutical ingredient (API), meeting the stringent raw material standards of high-end pediatric formulations and geriatric specialty drugs.
Enzyme stability improvement has become a key research focus. Through gentle modification methods such as microencapsulation and carrier adsorption, activity loss caused by acid-base and temperature fluctuations during storage and formulation processing is reduced, extending the shelf life of finished formulations, broadening the acid-base compatibility range of formulations, and facilitating the stable development of compound multi-enzyme preparations.
Targeted release formulation technology is constantly iterating. Utilizing acid-resistant coating and targeted microencapsulation technologies, enzymes are protected to smoothly pass through areas of fluctuating gastric acid, achieving precise site-specific action, further improving effective utilization, reducing activity attenuation during processing, and optimizing the actual effectiveness of formulations.
The application boundaries continue to expand, from traditional stomach-strengthening and digestion-aiding products to sub-sectors such as post-gastric and intestinal repair formulas, low-irritant medical conditioning preparations, and pet digestive specialty raw materials. Relying on the core advantages of natural homology, low irritation, and high tolerance, it opens up diversified and compliant application tracks and promotes the standardization and upgrading of the digestive enzyme pharmaceutical raw material industry.
Conclusion
Pepsin Enzyme Powder, with its unique molecular structure of natural acidic protease, its targeted hydrolysis mechanism in the stomach, and its broad formulation compatibility, has become an essential raw material in the field of digestive regulation pharmaceuticals. It precisely addresses the shortcomings of acidic digestion in the human body, gently breaks down large protein molecules, and stabilizes gastrointestinal metabolic balance, offering multiple advantages such as clear efficacy, high tolerability, and outstanding cost-effectiveness. With the optimization of purification processes and the improvement of novel delivery technologies, this enzyme powder raw material will continue to provide stable and compliant high-quality raw material support for stomach-strengthening and digestive aid formulations, medical nutritional supplements, and biopharmaceutical processing.
Xi'an Faithful BioTech Co., Ltd. offers high-quality Pepsin Enzyme Powder, comprehensive technical support, and highly competitive wholesale prices. Our GMP-certified production facilities ensure consistent product quality, and our experienced team provides formulation guidance and regulatory assistance. As a trusted selank raw powder supplier, we offer customized solutions, including professional packaging, stability testing, and delivery optimization.
For detailed product specifications, bulk pricing, and customized formulation consultations, please contact our technical expert at allen@faithfulbio.com.
References
- Kageyama, T. (2019). Structure and catalytic mechanism of pepsin family aspartic proteases. Journal of Biochemistry, 166(3), 245-253.
- Singh, A., & Kumar, V. (2020). Pharmaceutical application and stability evaluation of pepsin enzyme powder. Process Biochemistry, 92, 102-108.
- Hassan, M. I., Abdelhamid, A., & Mahmoud, S. (2021). Acidic protease functional characteristics in gastrointestinal digestion regulation. Nutrients, 13(7), 2214.
- Lee, S. H., & Park, J. H. (2022). Extraction purification and quality control of medicinal grade pepsin. Journal of Pharmaceutical Sciences, 111(5), 1489-1495.
- Garcia, R., Mendez, J., & Ruiz, S. (2022). Compatibility of pepsin with common pharmaceutical excipients. Drug Development and Industrial Pharmacy, 48(8), 1201-1209.
- El-Sayed, A. (2023). Modern green extraction technology for animal-derived digestive enzymes. Sustainable Chemistry and Pharmacy, 33, 100967.
- Russo, F., & Vitale, G. (2023). Advancements in modified pepsin and targeted digestive enzyme formulations. European Journal of Medicinal Chemistry, 262, 115680.



