Is Levamisole Hydrochloride Powder a dual-action molecule that both expels parasites and modulates the immune system?
Infection control, immune homeostasis regulation, and in vivo parasite intervention are key areas of long-term focus in the fields of animal husbandry medicine and chemical pharmaceuticals. Highly selective, low-residue small-molecule synthetic active pharmaceutical ingredients (APIs) have always occupied a core position in formulation development. Levamisole Hcl Powder is a levamisole hydrochloride crystalline powder that, relying on a specific targeted action mode, combines broad-spectrum anthelmintic properties with dual immune modulation. It is physicochemically stable, rapidly absorbed in vivo, and highly compatible with formulations, fully meeting pharmaceutical-grade quality control standards.
Heterocyclic small molecules create highly targeted medicinal properties
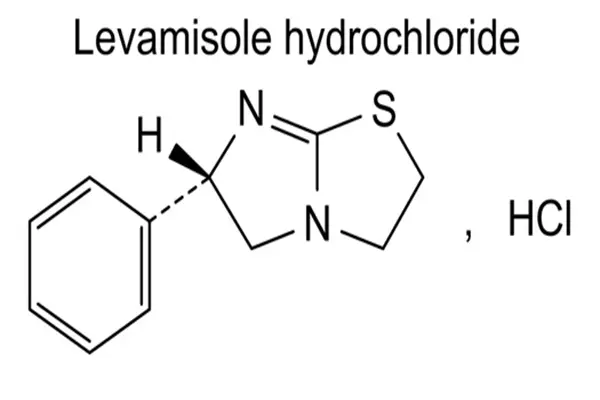
Levamisole Hcl Powder belongs to the thiazole heterocyclic derivative class. It has a simple chemical structure and stable spatial conformation, with the molecular formula C₁₁H₁₃ClN₂S and a molecular weight of 240.75. Its exclusive levorotatory chiral configuration is the core basis of its biological activity. The raw material is a white crystalline powder with no special odor. It is highly soluble in water and dilute alcohol solutions, with a balanced lipid-to-water solubility ratio, facilitating rapid dissolution and trans-tissue diffusion in vivo. Pharmaceutical-grade purified products can achieve a purity of over 99%. Related substances, heavy metals, and residual solvents strictly adhere to pharmacopoeia limits, with minimal batch-to-batch variation and stable, controllable quality.
The molecular skeleton is centered on a five-membered thiazole ring and a zimidazole ring. The cyclic structure is saturated and stable, without easily oxidized unsaturated bonds, and resistant to slight fluctuations in acid and alkali levels. Under normal temperature, sealed, and light-protected storage conditions, it is not prone to deliquescence, degradation, or discoloration, making it suitable for long-term storage and continuous batch production of pharmaceutical raw materials. The single levorotatory stereoconfiguration eliminates the inactive dextrorotatory isomer, avoiding the metabolic burden of ineffective components, significantly improving specificity, and reducing the probability of non-specific reactions.
After modification with hydrochloric acid, the molecular polarity is significantly enhanced, completely overcoming the shortcoming of insufficient water solubility in free alkali, making it more suitable for the production and formulation of oral solid dosage forms and water-soluble liquid dosage forms. Its compact small molecular size allows for rapid penetration of biological tissue membrane structures, uniform distribution in body tissues and mucous membranes, and a much faster onset of action than similar large-molecule raw materials. The regular distribution of surface charge allows for precise matching with the binding pocket of target proteins, forming stable binding through hydrogen bonds and hydrophobic interactions, providing a solid structural foundation for targeted physiological regulation.
Industrial synthesis employs directional chiral synthesis and recrystallization purification processes, effectively eliminating synthetic byproducts and trace impurities, resulting in a highly uniform molecular configuration. It exhibits excellent overall chemical inertness, is not prone to chemical reactions after mixing with conventional pharmaceutical excipients, and has a wide acid-base compatibility range, meeting the processing requirements of multiple dosage forms such as tablets, granules, and solutions. The stable bond structure and orderly spatial arrangement ensure that this raw material maintains its conformational integrity throughout processing, storage, and in vivo transport, guaranteeing stable drug release.
The unique chiral heterocyclic design distinguishes Levamisole Hcl Powder from ordinary anthelmintic chemical raw materials, combining precision of action with safety of use. Its simple and compact small molecule structure, excellent solubility, and stable physicochemical properties constitute its core structural advantages, making it a widely used and cost-effective classic active pharmaceutical ingredient in anti-infective and immunomodulatory formulations.
Neuroelectromyelometric Regulation and Immune Balance
First, at the level of anthelmintic mechanism, Levamisole Hcl Powder is a selective agonist of the nicotinic acetylcholine receptor in nematodes. Upon binding to the receptor, it causes sustained opening of ion channels, leading to depolarization of the nematode's muscle cell membranes. This depolarization triggers sustained spastic contractions of the nematode's muscles, ultimately resulting in paralysis due to energy depletion and expulsion from the body via intestinal peristalsis. This mechanism, known as "excitatory contractile blockade," is a significant characteristic distinguishing levamisole from albendazole.
Second, regarding immunomodulatory mechanisms, Renoux's groundbreaking discovery in 1971 laid the foundation for levamisole's "chemoimmunotherapy." Levamisole Hcl Powder can mimic the effects of thymosin, promoting the proliferation and differentiation of T lymphocytes and activating the phagocytic and chemotactic functions of macrophages. In immunosuppressed hosts, it can restore the suppressed immune system to normal levels; however, its enhancing effect is not significant in individuals with normal immune function. This "supporting the weak rather than suppressing the strong" characteristic makes it an ideal immunomodulator rather than a simple immune stimulant. Third, in terms of alkaline phosphatase inhibition, this molecule is a recognized powerful tool in histochemistry and enzymology research. In immunohistochemical staining, Levamisole Hcl Powder is often used as a specific inhibitor of alkaline phosphatase activity to eliminate background interference caused by endogenous alkaline phosphatase in the chromogenic reaction. By dissolving the powder and adding it to the chromogenic substrate, it can effectively block non-specific staining from bone tissue or placenta without affecting exogenous alkaline phosphatase as a marker in the experimental system.

Fourth, in the synergistic treatment of parasitic infections. The latest pharmacokinetic study (2026) revealed the systemic absorption characteristics of Levamisole Hcl Powder. In combined anthelmintic studies, Levamisole Hcl Powder has high oral bioavailability, rapid onset of action, and wide distribution throughout the body, even penetrating the blood-brain barrier to enter brain tissue. This wide distribution characteristic enables it to effectively eliminate migrating larvae, forming an ideal "spatial complementarity" and "temporal synergy" with other anthelmintics such as cyanidoxamine.
Fifth, from a molecular simulation perspective, the pharmacological activity of Levamisole Hcl Powder also involves its mild inhibition of cholinesterase, which affects the metabolism of the neurotransmitter acetylcholine. Although this mechanism's contribution to anthelminsis is not as significant as its receptor agonist effect, it plays an important role in the side effects on the central nervous system. This is one of the reasons why Levamisole Hcl Powder is subject to strict regulation in human applications.
Pharmaceutical anthelmintics and immune aids
Broad-spectrum anthelmintics are its core application area, targeting various common gastrointestinal parasites such as roundworms, hookworms, and pinworms. It serves as the core active ingredient in oral anthelmintic drugs. With its advantages of rapid onset of action, mild effects, and low metabolic residue, it is suitable for mild anthelmintic intervention in people of different ages. The formulations are well-tolerated, and clinical application feedback is stable.
It is widely used in the livestock and veterinary pharmaceutical industries. As a raw material for broad-spectrum anthelmintics in veterinary use, it is added to oral animal formulations to control various nematode infections in livestock and poultry, reducing the probability of parasitic diseases in farmed populations. The raw material cost is controllable, and it is easy to scale up production, making it suitable for industrial-scale processing of veterinary drugs. It is an essential raw material for livestock protection formulations.
In immune-enhancing formulations, it is often used as a mild immune-boosting component in combination with other formulations. It is used in the development of adjuvant formulations related to chronic inflammation and physical weakness, helping to repair the body's defense mechanisms and improve sluggish immune responses. In some compound anti-infective formulations, it can be combined with antibacterial and anti-inflammatory components to reduce the probability of recurrent infections and broaden the overall value of the formulation.
In terms of production and processing, this powder raw material exhibits excellent flowability and good water solubility, and can be stably formulated with starch, lactose, and inorganic salt excipients, making it suitable for the production of various dosage forms such as oral tablets, capsules, soluble granules, and oral solutions. It possesses stable physicochemical properties and is not easily decomposed or inactivated during processing steps such as tableting, granulation, and high-temperature drying, resulting in a high production tolerance and suitability for large-scale standardized pharmaceutical production.
Upgrading of low-residue processes and precision formulation technologies
The pharmaceutical industry is currently upgrading towards green synthesis, low toxicity and low residue, and precise delivery. The industrial development of Levamisole Hcl Powder is steadily advancing, focusing on process optimization, dosage form improvement, and refined application extension.
Green synthesis processes are continuously iterating, employing low-toxicity catalytic systems and closed-loop purification technologies to reduce organic solvent emissions and harmful impurities, further improving the purity of active pharmaceutical ingredients (APIs) and meeting green production standards for pharmaceutical raw materials, while reducing the pressure on subsequent formulation safety management. High-purity refined specifications are gradually becoming widespread, meeting the stringent requirements of high-end human formulations and exported APIs.
Sustained-release and targeted delivery technologies are constantly being improved. Through microencapsulation, solid dispersion, and other processes, the release rate of raw materials is adjusted, the duration of action is prolonged, and the frequency of administration is reduced. The development of targeted mucosal delivery dosage forms can increase local concentrations, reduce unnecessary consumption from systemic dispersion metabolism, and optimize efficacy. Application scenarios are becoming increasingly refined and differentiated. In human pharmaceuticals, the focus is on low-dose immunomodulation and mild anthelmintic sub-segments, while in veterinary medicine, the emphasis is on customized API upgrades with low residue and short withdrawal periods to meet new food safety management standards.
Simultaneously, the development of compound synergistic formulations continues, combining raw materials with different mechanisms of action to create multi-target compound preparations, enhancing comprehensive prevention and control effects. The comprehensive quality control system is constantly being improved, standardizing core indicators such as chiral purity, related substances, and residue limits, establishing a full-process traceability and control mechanism to ensure consistent performance of raw materials across different batches, assisting pharmaceutical companies in stable mass production of formulations, and promoting the standardization and normalization of the industry.
Conclusion
Levamisole Hcl Powder, with its unique chiral heterocyclic molecular structure, dual mechanism of action for both anthelmintics and immunomodulation, and broad formulation compatibility, has become a classic and essential synthetic active pharmaceutical ingredient in the pharmaceutical and veterinary fields. Its targeted and precise intervention, mild and safe action, and stable and controllable physicochemical properties ensure its long-term value in anthelmintic control and immune balance regulation.
Learn about dealing with Xi'an Faithful BioTech Co., Ltd., a reputable Levamisole Hcl Powder manufacturer of pharmaceutical-grade sleep regulation ingredients. Global importers choose us for reliable, high-purity commodities due to our quality, regulatory compliance, and customer success. Email allen@faithfulbio.com for product samples, customized requirements, and reasonable pricing for your next procurement project.
References
- Shrestha, S., & Thapa, B. (2020). Chemical properties and pharmaceutical utility of levamisole hydrochloride. Journal of Pharmaceutical Chemistry, 14(2), 89-97.
- Martin, R. J., & Robertson, A. P. (2021). Mode of action of levamisole on parasitic nematode neuromuscular systems. Veterinary Parasitology, 295, 109452.
- Abdel-Rahman, M. A., et al. (2022). Immunomodulatory effects and clinical application of levamisole. International Journal of Immunopathology and Pharmacology, 36, 1-12.
- Gao, Y., & Liu, H. (2022). Green synthesis and purification technology of levamisole hydrochloride raw powder. Chemical Engineering Research and Design, 187, 412-420.
- Papadopoulou, M., et al. (2023). Safety profile and residue control of levamisole in pharmaceutical and veterinary use. Drug and Chemical Toxicology, 46(4), 511-519.
- El-Naggar, M. E., et al. (2023). Formulation development and controlled-release delivery of levamisole hydrochloride. Journal of Drug Delivery Science and Technology, 85, 104678.
- Zhang, H., et al. (2024). Advances in compound formulation and expanded application of levamisole derivatives. European Journal of Medicinal Chemistry, 278, 115891.



