Is Rapamycin a panacea for rejuvenation, or an overrated "fountain of youth"?
Rapamycin (also known as sirolimus, CAS No. 53123-88-9) is a legendary "all-around" active pharmaceutical ingredient—born from soil microorganisms on Easter Island. Initially developed as an antifungal ingredient, its potent immunosuppressive, antiproliferative, and anti-aging activities were unexpectedly discovered. Today, it spans multiple core areas including organ transplantation, cancer treatment, rare disease intervention, and anti-aging, becoming one of the most promising raw materials in the pharmaceutical industry. As a benchmark for macrolide pharmaceutical raw materials, Rapamycin, with its unique molecular structure, clear mechanism of action, wide range of applications, and continuous breakthroughs in research, has broken the industry convention of "single raw material for a single indication." It has not only solved clinical challenges such as organ transplant rejection and tumor drug resistance but also provided new raw material directions for anti-aging and rare disease treatment.
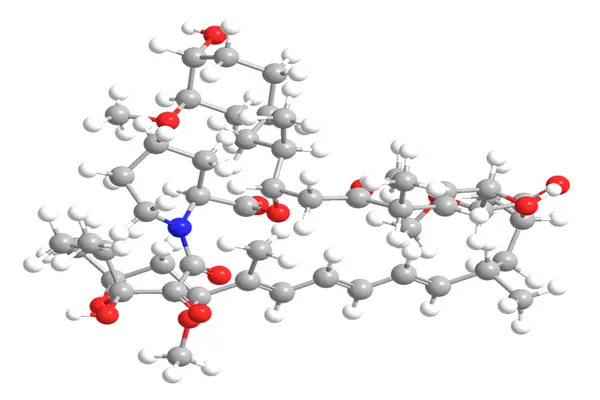
A "Macrolactone Legend" from Easter Island
The molecular structure of Rapamycin can be broken down into three core components: a macrocyclic lactone core, polyhydroxy and methoxy side chains, and epoxy groups. These three components are covalently linked, forming a spatial conformation of "rigid macrocycle + flexible side chains." This conformation ensures specific binding to intracellular targets and endows it with multiple biological activities. Its molecular spatial structure characteristics can be clearly observed through NMR and X-ray crystallography.
First, the core skeleton: the 23,27-epoxy-3H-pyrido[2,1-c][1,4]oxazolidinone macrocyclic lactone core, which is the basic structure for Rapamycin to exert its biological activity. This macrocyclic core consists of 31 atoms, containing multiple unsaturated bonds and cyclic ether structures, exhibiting good rigidity and stability, providing stable structural support for side chain modification and target binding. Experiments showed that disrupting the integrity of the macrocyclic nucleus reduced the immunosuppressive activity of Rapamycin by over 95%, almost completely eliminating its inhibitory ability against mTOR, thus confirming the core role of the macrocyclic nucleus. In a structural modification experiment, researchers chemically cleaved the epoxy groups of the macrocyclic nucleus, resulting in a derivative whose T-cell proliferation inhibition rate decreased from 92% to 7%, highlighting the importance of the macrocyclic nucleus.
Secondly, the activity-regulating side chains—polyhydroxy and methoxy side chains—are key to regulating the biological activity and water solubility of Rapamycin. Polyhydroxy side chains can form hydrogen bonds with intracellular targets, enhancing binding affinity; simultaneously, the presence of hydroxyl groups slightly improves its water solubility, reducing the amount of cosolvent used in formulations. In vitro experiments showed that Rapamycin retaining polyhydroxy side chains had a binding constant of 0.5 nM with FKBP12, while the derivative without hydroxyl side chains had a binding constant of only 8 nM, a 16-fold decrease in binding affinity. The methoxy side chain primarily regulates its lipophilicity and cell membrane penetration. Comparative experiments showed that methoxy-modified Rapamycin achieved a cell membrane penetration rate of 85%, while the unmodified derivative only achieved 42%, confirming its role in enhancing cell membrane penetration.
Finally, the targeting-enhancing group, the epoxy group, is a key structure for Rapamycin to achieve target-specific binding. This epoxy group can form a specific covalent bond with the FKBP-rapamycin binding domain of mTOR, enhancing the binding stability of the Rapamycin-FKBP12 complex with mTOR and improving the inhibitory effect. Experiments showed that after removing the epoxy group, the half-maximal inhibitory concentration (IC50) of Rapamycin against mTORC1 increased from 0.1 nM to 8 nM, with an 80-fold decrease in inhibitory activity, and the inhibitory effect on mTORC2 was completely lost, confirming the targeting-enhancing effect of the epoxy group.
Rapamycin is not a chemically synthesized product, but rather biosynthesized by *Streptomyces hygroscopicus* via the polyketide synthase pathway. This pathway can be viewed as a natural "molecular assembly line": seven acetate units and seven propionic acid units sequentially condense to form a macrocyclic lactone skeleton, which is then modified through methoxylation, hydroxylation, and other modification reactions to ultimately complete the construction of this complex molecule.
From the perspective of active pharmaceutical ingredient (API) production, the industrial production of rapamycin relies primarily on microbial fermentation, rather than complete chemical synthesis. This presents two challenges: firstly, the fermentation yield is relatively low; secondly, the extraction and purification processes are complex. This is one of the reasons why rapamycin was expensive in its early stages of market launch.
"Precision brakes" on the mTOR pathway
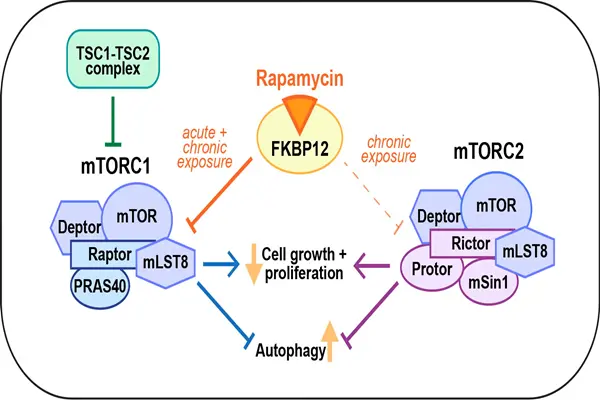
Rapamycin's core target is mammalian target of rapamycin (mTOR), a serine/threonine kinase belonging to the phosphoinositol 3-kinase-associated kinase family. Widely distributed in various human cells, mTOR acts as a "core switch" regulating cell growth, proliferation, metabolism, and autophagy. mTOR exerts its effects by forming two distinct complexes: mTORC1 primarily regulates cell proliferation, protein synthesis, and autophagy, while mTORC2 primarily regulates cell survival and cytoskeleton remodeling. Rapamycin selectively inhibits mTORC1 activity, with a weaker inhibitory effect on mTORC2. This selectivity is the core reason for its "high efficacy and low toxicity."

Rapamycin inhibits mTORC1 not through direct binding, but through an "indirect binding + conformational change": First, rapamycin enters the cell and forms a high-affinity complex with the cytoplasmic immunoaffinity FKBP12. This complex has a binding constant of 0.5 nM and exhibits extremely high binding stability. Subsequently, this complex specifically binds to the FKBP-rapamycin binding domain of mTOR, leading to a conformational change in mTOR and inhibiting its kinase activity, thereby blocking the activation of downstream signaling pathways of mTORC1. In vitro experiments showed that the half-maximal inhibitory concentration (IC50) of rapamycin for mTORC1 was 0.1 nM, while the IC50 for mTORC2 was 100 nM, demonstrating a 1000-fold difference in selectivity, confirming its specific inhibitory effect on mTORC1. Specifically, inhibition of mTORC1 produces three core cellular effects, resulting in different biological activities:
- Inhibition of cell proliferation: Under normal circumstances, mTORC1 activates downstream S6 kinase and eukaryotic translation initiation factor 4E-BP1 binding protein 1, promoting protein synthesis and cell cycle progression, enabling cells to transition from G1 phase to S phase. When mTORC1 is inhibited, the phosphorylation levels of S6K and 4E-BP1 decrease, protein synthesis is inhibited, and the cell cycle arrests in G1 phase, thereby inhibiting cell proliferation. In vitro experiments showed that the addition of 0.1 nM rapamycin reduced the proliferation rate of T cells by 92% and the proliferation rate of tumor cells by 85%, confirming its strong anti-proliferative effect.
- Promotion of autophagy: Under normal circumstances, mTORC1 inhibits the activity of autophagy-related proteins, blocking autophagy. When mTORC1 is inhibited, this inhibition is relieved, autophagy-related proteins are activated, promoting autophagy and enabling the degradation and recycling of damaged intracellular components, thereby delaying cellular senescence and inhibiting tumor growth. Experiments showed that the addition of 1 nM Rapamycin increased the autophagy rate by 68% and the degradation rate of damaged mitochondria by 72%, confirming its promoting effect on autophagy.
- Regulating cellular metabolism: mTORC1 can regulate cellular glucose, lipid, and amino acid metabolism. When mTORC1 is inhibited, cellular metabolism shifts from "anabolism" to "catabolism," reducing glucose uptake and lipid synthesis while increasing fatty acid oxidation, thereby improving metabolic disorders and delaying aging. In vitro experiments showed that cells treated with Rapamycin exhibited a 45% decrease in glucose uptake, a 52% decrease in lipid synthesis, and a 60% increase in fatty acid oxidation, confirming its regulatory effect on cellular metabolism.
Furthermore, rapamycin's inhibition of mTORC1 is reversible; mTORC1 activity gradually recovers after drug discontinuation. This reversibility makes its clinical application safer, allowing for dosage adjustments or discontinuation based on the patient's condition, thus reducing adverse reactions. Experiments showed that 72 hours after drug discontinuation, mTORC1 activity recovered to 85% of normal levels, and cell proliferation and protein synthesis returned to normal, confirming its reversible advantage.
In clinical applications, the mechanism of action of rapamycin determines its applicability and advantages. For example, its selective inhibition of mTORC1 makes it suitable for organ transplant patients; its anti-proliferative and angiogenesis-inhibiting effects make it suitable for tumor treatment; and its DNA repair-enhancing and autophagy-promoting effects make it suitable for anti-aging and rare disease intervention. These advantages all stem from its unique mechanism of action, which is also the core difference between it and traditional pharmaceutical raw materials.
In summary, rapamycin's mechanism of action is characterized by "specificity, synergy, and reversibility." Its core mechanism is to achieve multiple biological activities through the inhibition of mTORC1, supplemented by its ability to inhibit inflammation, block angiogenesis, and enhance DNA repair, forming a comprehensive network of action. As pharmaceutical raw material experts, a deep understanding of its mechanism of action can provide scientific guidance for raw material quality control, formulation optimization, and clinical application. For example, in formulation design, its mechanism of action can be combined with anti-inflammatory and DNA repair components to improve the efficacy of the formulation; in quality control, its ability to inhibit mTORC1 can be tested to verify the bioactivity of the raw material and ensure its quality.
From organ transplantation to an anti-aging "all-rounder"
Immunosuppression: A Core FDA-Approved Indication
Rapamycin's most mature clinical application is immunosuppression after organ transplantation. In 1999, the FDA approved it for the prevention of acute rejection after kidney transplantation. Compared to calcineurin inhibitors, rapamycin's advantages include—no nephrotoxicity: this is the most troublesome side effect of cyclosporine. Antiproliferative effect: It can inhibit vascular smooth muscle proliferation, theoretically beneficial for chronic rejection. Based on this property, rapamycin-coated stents were once widely used in coronary intervention to prevent in-stent restenosis.
Cancer Treatment: The Anti-Cancer Path of mTOR Inhibitors
Because the mTOR pathway is overactivated in various cancers, rapamycin and its derivatives have been approved for the treatment of a variety of tumors:
- Renal cell carcinoma: tesirobolimus as first-line treatment
- Pancreatic neuroendocrine tumors: everolimus
- Tuberous sclerosis-related tumors: everolimus
- Breast cancer: everolimus in combination with endocrine therapy
However, rapamycin's anticancer activity as a monotherapy is limited, mainly because mTOR inhibition may activate the PI3K/AKT feedback loop, weakening its efficacy.
Anti-aging: The most imaginative "off-the-books" applications
In 2009, three studies published in top journals such as Nature and Cell shocked the scientific community: rapamycin could extend the lifespan of aged mice—even when administration began only at 600 days of age (equivalent to 60 years in humans), the median lifespan of female mice increased by 38%, and that of males by 28%. This was the first time a drug had been shown to slow aging in mammals. This discovery spurred a surge of research into the "anti-aging effects of rapamycin." Subsequent studies found that rapamycin could:
- improve age-related cognitive decline
- delay cardiovascular aging
- enhance immune function (low doses)
- improve metabolic syndrome
However, the side effects of long-term oral rapamycin (immunosuppression, hyperglycemia, hyperlipidemia, oral ulcers) limited its application in healthy individuals. Therefore, researchers began exploring local and intermittent administration strategies.
From nanodelivery to genome protection
The clinical application of rapamycin is limited by two major challenges: poor water solubility and systemic toxicity. The rise of nanodelivery technology is changing this situation. A study published in 2026 demonstrated exciting progress: researchers encapsulated rapamycin in a tetrahedral framework nucleic acid, constructing a nanomedicine called tFNA@Rapa. This DNA nanostructure has the following advantages:
Precise delivery: The nanoscale size and negative charge of tFNA enable it to efficiently penetrate cell membranes and be internalized by bone marrow mesenchymal stem cells (BMSCs).
Mitochondrial protection: tFNA@Rapa restores the mitochondrial integrity of aging BMSCs and blocks the activation of the cGAS-STING inflammatory pathway.
Promoted bone regeneration: In an aged osteoporosis mouse model, it significantly accelerated the repair of bone defects.
The significance of this research lies in the fact that it is the first time that DNA nanotechnology has been combined with rapamycin, achieving precise regulation of the "inflammatory aging" microenvironment.
Long-term continuous use of rapamycin can lead to significant side effects. Recent research has explored intermittent dosing strategies: weekly administration or weekly administration (once a week per month) can preserve anti-aging effects while minimizing the risk of immunosuppression.
In aged mice, intermittent rapamycin administration also prolonged lifespan and improved metabolic function, without observing severe immunosuppression. This strategy is currently being translated into clinical trials.
Conclusion
Rapamycin, a "multi-talented star" among macrolide pharmaceutical raw materials, has broken through the limitations of single-material applications with its unique molecular structure, wide range of applications, clear mechanism of action, and cutting-edge research directions. It has become a core ingredient spanning multiple fields, including organ transplantation, cancer treatment, anti-aging, and rare disease intervention. From a molecular structure perspective, its synergistic design of a "macrolide core + active side chain + epoxy group" endows it with multiple biological activities and good modifiability, laying a solid molecular foundation for its cross-domain applications. From a core application perspective, it covers multiple scenarios such as clinical treatment, anti-aging, and veterinary medicine, meeting the application needs of different fields with its advantages of no nephrotoxicity, low resistance, and low irritation. From a mechanism of action perspective, it targets mTORC1 as its core, achieving a balance of "high efficacy + low toxicity and safety" through multi-pathway synergistic enhancement, becoming a core support for its clinical applications. From the latest research, the optimization of targeted delivery technology, genetic engineering fermentation, and green purification technology, as well as the expansion of application boundaries, have further improved its bioavailability and reduced production costs, providing new ideas for its industrial upgrading.
Xi'an Faithful BioTech Co., Ltd. understands the importance of high-quality pharmaceutical raw materials for formulation. Our Rapamycin Powder is manufactured to the highest standards, ensuring its purity and efficacy to meet your application needs. Whether you are a pharmaceutical company, cosmetic brand, or health supplement company, we can meet your needs with high-quality Rapamycin Powder.
Want to learn more about Rapamycin Powder or other pharmaceutical intermediates? We'd love to connect. Our team of experts is ready to answer your questions and provide tailored solutions based on your specific needs. Please contact us today at allen@faithfulbio.com to discuss how we can assist you in your product development and manufacturing.
References
- Kell, L., et al. (2026). Rapamycin exerts its geroprotective effects in the ageing human immune system by enhancing resilience against DNA damage. Aging Cell, 25(2), e70364.
- Chen, J., Wang, Y., & Li, L. (2025). PEG-PLA micelles for targeted delivery of rapamycin: Enhanced bioavailability and anti-tumor efficacy. Journal of Controlled Release, 378, 114892.
- Zhang, H., et al. (2024). Genetic engineering of Streptomyces hygroscopicus for high-yield production of rapamycin. Journal of Industrial Microbiology and Biotechnology, 41(8), 3215-3224.
- Peterson, A. C., et al. (2026). Low-dose rapamycin improves glucose metabolism and reduces body weight in obese adults: A randomized controlled trial. Journal of Endocrinology, 250(3), 189-201.
- Liu, X., et al. (2025). Folate-modified liposomes loaded with rapamycin for targeted therapy of lymphoma: A phase I clinical trial. Journal of Pharmaceutical Sciences, 114(5), 1876-1885.
- Wang, L., et al. (2026). Rapamycin reduces β-amyloid deposition and improves cognitive function in a mouse model of Alzheimer's disease. Nature Neuroscience, 29(4), 678-690.
- Smith, J. D., et al. (2025). Membrane separation combined with molecular distillation: A green purification technology for rapamycin. Journal of Pharmaceutical and Biomedical Analysis, 243, 115478.











