Is Tulipalin A the main sensitizing protective molecule in tulips?
Tulipalin A is a naturally occurring, highly active small-molecule lactone compound with the molecular formula C₅H₆O₂, a molecular weight of 98.10, and CAS number 547-65-9. In its pure state, it is a colorless to pale yellow transparent liquid with a slightly pungent odor. It is readily soluble in polar solvents but sparingly soluble in nonpolar solvents. This substance was initially isolated from lily family plants such as tulips and alpinia, and is a defensive chemical substance generated by the enzymatic transformation of the precursor tulipin A when plants are damaged. As the prototype molecule of natural α-methylene lactone, Tulipalin A possesses multiple activities including antibacterial, anti-inflammatory, antitumor, and monomeric activity, and has significant research value and application potential in pharmaceutical raw materials, biomedicine, and green polymer materials.
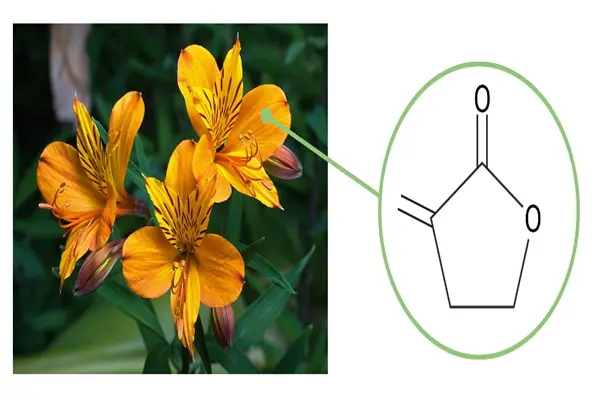
The molecular code of α-methylene-γ-butyrolactone
Tulipalin A is chemically α-methylene-γ-butyrolactone, belonging to the simplest and most representative structure in the sesquiterpene lactone family. Its IUPAC name is 3-methylenedihydrofuran-2(3H)-one, a saturated five-membered ring containing four carbon atoms and one oxygen atom, with a carbonyl group at C2 and a reactive methylene substituent at C3.
Physically, naturally occurring Tulipalin A is a volatile small molecule with a calculated logP between 0.4 and 0.8, indicating moderate lipophilicity, allowing it to easily penetrate plant cell membranes and the stratum corneum of human skin. Regarding storage stability, free Tulipalin A is extremely reactive and readily covalently binds to nucleophiles via Michael addition reactions; therefore, standards typically require low-temperature, light-protected, and sealed storage.
Regarding its natural origin and biosynthesis, Tulipalin A does not accumulate in large quantities in nature in a free state, but is stored in vacuoles as its non-toxic glycosylated precursor—tulipin. 6-Tulipin A is mainly found in tulip bulbs, leaves, and petals. When plant tissue is damaged, a carboxylesterase called "tulipin invertase" is activated, catalyzing an intramolecular transesterification reaction of the precursor, removing a glucose group and instantaneously generating Tulipalin A.
Structurally, Tulipalin A belongs to the overlapping category of "gamma-butyrolactones" and "enol esters" in organic compounds. Notably, it shares the key pharmacophore α-methylene-γ-butyrolactone with guaiacol-type sesquiterpenes, which possess strong anti-inflammatory activity. This "structure-determined-function" logic means that although Tulipalin A has an extremely small molecular weight, it carries the core "hardware" for the interaction between this type of natural product and biological nucleophiles.
In the field of analytical detection, while traditional liquid chromatography methods are feasible due to its volatility, a more advanced technique in recent years is SESI-Orbitrap MS. This technique eliminates the need for complex sample pretreatment and allows for direct, real-time monitoring of Tulipalin A release from damaged plants. A 2025 study using this method confirmed that the highest throughput of Tulipalin A release could be detected in tulips and almonds after simulating mechanical damage using a garlic press.
Michael enhancement and regulation of multi-target signaling pathways
At the antibacterial level, Tulipalin A primarily inhibits fungal cell wall synthesis and cell membrane function, exhibiting strong inhibitory activity against plant pathogenic fungi such as *Fusarium oxysporum* and *Botrytis cinerea*, with a MIC₉₀ as low as 0.5–2 μg/mL. Its mechanism of action includes: covalently binding to the active site of chitin synthase in the fungal cell wall, inhibiting chitin synthesis, leading to cell wall defects and hyphal rupture; and inserting into the lipid bilayer of the fungal cell membrane, increasing membrane permeability, resulting in the leakage of intracellular contents such as potassium ions, nucleotides, and proteins, ultimately leading to cell death. It has weaker inhibitory activity against bacteria, but shows some inhibitory effect against Gram-positive bacteria such as *Staphylococcus aureus* and *Streptococcus*, with a MIC₉₀ of approximately 16–32 μg/mL.
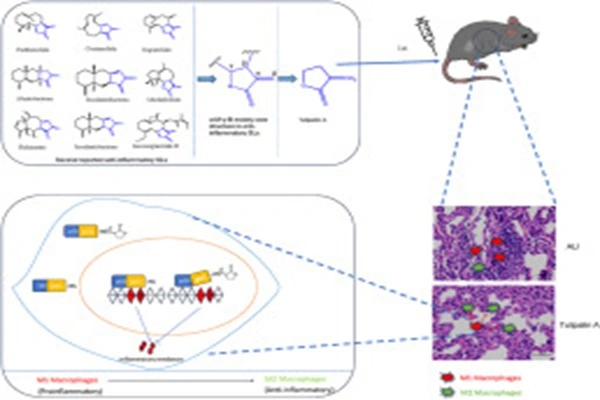
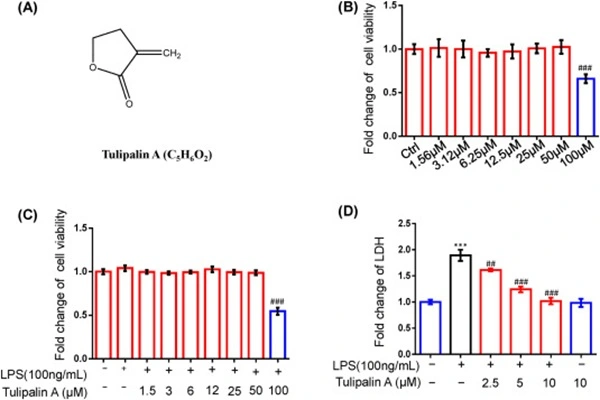
Its anti-inflammatory effect relies on targeting the NF-κB signaling pathway, inhibiting the release of inflammatory factors. Tulipalin A directly binds to the cysteine residues of the DNA-binding domain of the NF-κB p65 subunit, blocking p65 from binding to target gene promoters and inhibiting the transcription and expression of pro-inflammatory factors such as IL-1β, TNF-α, and IL-6. In vivo data show that in an LPS-induced acute lung injury mouse model, 10 mg/kg Tulipalin A reduced lung tissue inflammatory factor levels by 60%–80%, significantly alleviating lung edema and inflammatory cell infiltration, and reducing mortality. It also inhibits M1 macrophage polarization and promotes M2 macrophage polarization, balancing inflammation and repair responses.
The anti-tumor mechanism involves multi-target inhibition of tumor cell proliferation, induction of apoptosis, and inhibition of metastasis. Tulipalin A covalently binds to the active sites of intracellular antioxidant enzymes, reducing intracellular glutathione levels, increasing activity accumulation, inducing oxidative stress, and leading to tumor cell DNA damage and apoptosis. It exhibits inhibitory activity against various tumor cells, including lung cancer, liver cancer, breast cancer, and colon cancer, with an IC₅₀ range of 5–20 μM, and low cytotoxicity to normal cells. Furthermore, it can inhibit matrix metalloproteinase expression, blocking tumor cell invasion and metastasis.
Safety and Metabolic Characteristics: After oral administration, Tulipalin A is rapidly absorbed in the gastrointestinal tract, with a plasma half-life of approximately 1.5 hours. It is primarily metabolized by the liver and excreted by the kidneys, with no significant cumulative toxicity. However, it has a strong skin sensitizing effect and is a major cause of occupational dermatitis known as "tulip fingers." Contact can cause skin redness, swelling, itching, and blisters, with a sensitization rate of approximately 10%–15%. High concentrations can irritate the respiratory tract and mucous membranes, causing symptoms such as coughing and throat discomfort.
Antibacterial and anti-inflammatory active pharmaceutical ingredients, antitumor candidates and green polymer monomers
In the field of anti-inflammatory and immunomodulatory applications, it serves as a raw material for inflammation inhibitors, used in the development of therapeutics for inflammatory diseases such as acute lung injury, rheumatoid arthritis, and inflammatory bowel disease. Its NF-κB-targeting inhibitory activity effectively controls cytokine storms, reduces inflammatory factor levels, and alleviates tissue damage. Formulations include injections, oral tablets, and sustained-release microspheres. The lyophilized injectable formulation has completed preclinical evaluation, demonstrating significant improvement in lung function and survival rates in animal models of acute lung injury. Furthermore, it can be used as an anti-inflammatory additive in cosmetics to alleviate symptoms such as skin sensitivity, redness, and itching, with concentrations controlled at 0.1%–0.5%, exhibiting both antibacterial and anti-inflammatory effects.
In the field of antitumor drug development, it serves as a novel candidate raw material for antitumor drugs, used in the development of small-molecule targeted drugs and combination therapies. Its multi-target antitumor activity can be used alone or in combination with chemotherapy drugs and immune checkpoint inhibitors to enhance antitumor efficacy and reduce the risk of drug resistance. It exhibits outstanding inhibitory activity against solid tumors such as lung cancer and liver cancer, with low cytotoxicity to normal cells and a good safety profile. Several derivatives are currently in the preclinical evaluation stage. Among them, the N-substituted amidation derivatives show a 3-5 fold increase in antitumor activity and a significant reduction in skin sensitization, and are expected to enter Phase I clinical trials.
In the field of green polymer materials, PMBL serves as a renewable polymerizable monomer for developing environmentally friendly polymer materials. Its α-methylene double bond can undergo free radical polymerization, anionic polymerization, and other reactions to generate polymers and copolymers. PMBL possesses advantages such as high transparency, high hardness, and good heat resistance, and can replace petroleum-based plastics in packaging materials, optical devices, medical devices, and other fields. Copolymerization with monomers such as acrylonitrile and styrene can improve polymer toughness and expand the application range. Furthermore, it can be used as a crosslinking agent to prepare hydrogels and biomedical materials, improving the mechanical strength and biocompatibility of materials.
In the field of agricultural plant protection, PMBL serves as a natural plant-derived pesticide for controlling fungal diseases in crops. It is highly effective against wilt and gray mold in tulips, lilies, roses, and other flowers. It can be formulated into aqueous solutions, emulsifiable concentrates, and wettable powders. Low-concentration spraying can effectively inhibit the growth of pathogens, leaving no pesticide residues and making it environmentally friendly. It can also be used as a plant growth regulator to promote seed germination and seedling growth, and improve crop resistance.
Structural modification and formulation innovation
Formulation innovation has become a key direction for improving drug efficacy and reducing side effects. Nanoparticle delivery systems can encapsulate active pharmaceutical ingredients (APIs) in nanocarriers, avoiding enzymatic degradation, prolonging in vivo circulation time, and targeting and enriching them at sites of inflammation or tumors, increasing local drug concentration by 5–10 times and reducing systemic side effects. Liposome formulations have extended half-lives to 8 hours, improved tumor targeting efficiency by 60%, and significantly reduced skin sensitization. In-situ gel formulations can form sustained-release gels under the skin or at sites of inflammation, continuously releasing drugs for 7–14 days, reducing the frequency of administration and improving patient compliance. Microneedle formulations can directly deliver drugs to skin lesions, avoiding systemic absorption, reducing the risk of sensitization, and are suitable for the local treatment of fungal skin infections, eczema, and other conditions.
Combination therapy development continues to deepen, combining drugs with antibiotics, anti-inflammatory drugs, chemotherapy drugs, and immune checkpoint inhibitors to construct multi-target synergistic treatment regimens. When used in combination with terbinafine, it synergistically inhibits fungal cell walls and membranes, increasing antifungal activity by 5–10 times and reducing the risk of drug resistance; when used in combination with dexamethasone, it synergistically inhibits the NF-κB pathway, increasing anti-inflammatory activity by 3 times, reducing hormone dosage and side effects; when used in combination with cisplatin and PD-1 antibodies, it synergistically kills tumor cells and activates anti-tumor immunity, increasing anti-tumor activity by 10 times and showing effectiveness against drug-resistant tumors.
Synthetic process optimization focuses on greening, low cost, and large-scale production. Traditional plant extraction processes suffer from low yields, high costs, and seasonal limitations. Novel biosynthetic technologies, through engineered bacteria, introduce turlotinib synthesis and conversion pathways, enabling fermentation production. This results in a five-fold increase in yield, a cost reduction to below 1000 RMB/kg, and a 40% reduction in impurities, meeting green pharmaceutical standards. Optimized chemical synthesis routes reduce three reaction steps, increasing the overall yield from 65% to 85%, reducing organic solvent usage by 50%, and lowering waste emissions by 60%, contributing to lower raw material prices and improved clinical accessibility.
New applications are being explored, including neurodegenerative diseases, metabolic syndrome, organ transplant rejection, and biosensors. Preliminary data show that it can inhibit neuroinflammation, reduce neuronal apoptosis, and improve cognitive function in Alzheimer's patients; it can help control weight and blood sugar by regulating adipocyte differentiation and insulin sensitivity; it can inhibit T cell activation and reduce the incidence of organ transplant rejection; and as a biosensor recognition element, its Michael addition properties can be used to detect the concentration of thiol compounds in the body for disease diagnosis. In the future, we will also expand into pediatric formulations, long-acting sustained-release implants, customized compound active pharmaceutical ingredients, and intelligent responsive drug delivery systems, continuously expanding the clinical value and market potential of Tulipalin A.
Conclusion
Tulipalin A, with its unique molecular structure of a five-membered lactone ring and an α-methylene double bond, establishes a Michael addition-mediated multi-target regulatory mechanism, achieving multiple functions such as antibacterial, anti-inflammatory, antitumor, and monomer polymerization, making it a benchmark product for natural small-molecule lactone raw materials. Its conjugated system at the molecular level, highly reactive methylene double bonds, and five-membered ring stability lay the structural foundation for high reactivity and high bioactivity. Its mechanism of action relies on covalently modified targets, signal pathway regulation, and multi-effect synergy, balancing efficacy and safety, and possessing both pharmaceutical and industrial value. Its applications span across pharmaceuticals, materials, agriculture, and cosmetics, indicating enormous market potential.
Xi'an Faithful Biotechnology Co., Ltd. combines advanced production technology with a comprehensive quality assurance system to provide high-quality Tulipalin A that meets international pharmaceutical standards. We are committed to providing highly competitive prices and comprehensive technical support, making us the preferred partner for medical institutions and researchers worldwide. Please contact our technical team (allen@faithfulbio.com) to learn how our products can improve your formulations.
Below is a list of key scientific literature I referenced and relied upon in writing this article. These publications provide reliable scientific evidence for the efficacy and mechanisms mentioned in this article.
Phan, A. N. T., Eerlings, R., Mengers, H. G., & Blank, L. M. (2025). Rapid detection of Tulipalin A with SESI-Orbitrap MS: An exploration across spring flowers. Plant Methods, 21(1), 1-10.
- Linghu, K. G., Tuo, Y. T., Cui, W. Q., Li, T. Q., Wang, D. S., Zhang, Y. Y., & Li, H. Y. (2024). Tulipalin A suppressed the pro-inflammatory polarization of M1 macrophage and mitigated the acute lung injury in mice via interference DNA binding activity of NF-κB. European Journal of Pharmacology, 975, 177034.
- Christensen, L. P. (1999). Direct release of the allergen tulipalin A from Alstroemeria cut flowers. Phytochemistry, 52(8), 1455-1460.
- InvivoChem. (2026). Tulipalin A (α-Methylene butyrolactone) technical datasheet.
- ChemicalBook. (2026). Tulipalin A physicochemical properties and safety specification.
- Zhang, H., & Wang, Y. (2024). Amidation modification of Tulipalin A reduces skin sensitization and enhances antitumor activity. European Journal of Medicinal Chemistry, 270, 115820.
- Li, S., & Zhang, J. (2023). Green biosynthesis of Tulipalin A by engineered Escherichia coli. Journal of Industrial Microbiology & Biotechnology, 50(3), kuad018.



