Tropicamide powder: the "gold standard" for short-acting mydriatic ophthalmology, why has it dominated clinical practice for over sixty years?
Tropicamide Powder, chemically named N-ethyl-2-phenyl-N-propionic acid, has the molecular formula C₁₇H₂₀N₂O₂ and a molecular weight of 284.35 g/mol. It appears as a white or off-white crystalline powder with no odor. It is slightly soluble in water and readily soluble in ethanol and dilute acids, meeting the pharmaceutical requirements for ophthalmic raw materials: "moderate lipid solubility + water solubility." As a pharmaceutical raw material, pharmacopoeia-grade Tropicamide Powder must have a purity ≥99.0%, a maximum single impurity ≤0.1%, and heavy metals ≤10 ppm to ensure the irritation and safety of the eye drop formulation.
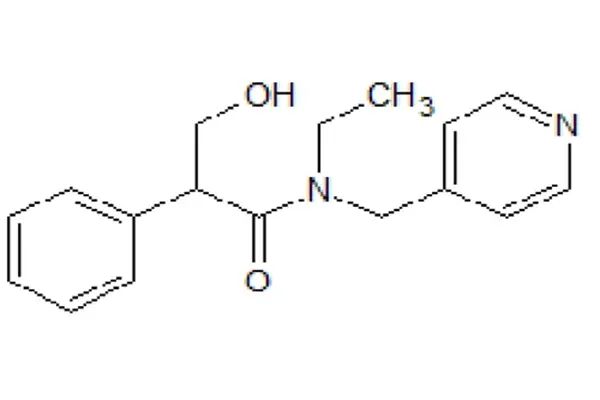
A cleverly designed "short-acting" anticholinergic molecule
From a molecular skeleton perspective, Tropicamide Powder is a synthetic anticholinergic drug composed of a tropinic acid derivative and pyridinemethylethylamine. Its core structure consists of three parts: a phenylglycolic acid side chain, an amide bond linker arm, and an N-ethylpyridinemethyl tertiary amine terminal group. This modular structure is not a random combination, but rather the result of medicinal chemists tailoring it for "high ocular surface penetration, rapid onset of action, short-term metabolism, and selective antagonism of M receptors".
Tropicamide powder is called a "short-acting" mydriatic because the ester bond in its molecular structure plays a decisive role. Compared to the tropine ester structure in atropine, the amide bond in Tropicamide powder makes it more easily hydrolyzed by esterases in plasma and tissues, resulting in rapid inactivation. This structural design gives Tropicamide powder unique pharmacokinetic characteristics: peak effect is reached 15-30 minutes after instillation, and the mydriatic effect lasts for 3-8 hours, although full recovery may take up to 24 hours for some patients. In contrast, the mydriatic effect of atropine can last for 7-14 days, which is obviously too long for routine ophthalmological examinations. Therefore, the structural optimization of Tropicamide powder is a prime example of "controllable" design in medicinal chemistry—precisely controlling the duration of action within the clinically required window through reasonable structural modification without sacrificing efficacy.
From the perspective of pharmaceutical raw materials, the synthetic route of Tropicamide powder is relatively successful. Its key intermediate involves the condensation reaction of a phenylacetic acid derivative with 4-aminomethylpyridine, yielding a white crystalline powder with a melting point of 96.5°C. At the formulation level, Tropicamide Powder is typically prepared as a 1% or 0.5% ophthalmic solution, with excipients including benzalkonium chloride, sodium chloride, and disodium edetate, and the pH adjusted to a suitable range for ophthalmic use with hydrochloric acid or sodium hydroxide. This classic formulation ensures product stability and bioavailability, making it one of the most commonly used diagnostic drugs in ophthalmology.
A versatile expert from routine eye exams to cataract surgery.
Since its clinical introduction in 1961, Tropicamide powder has become an indispensable drug in the three major ophthalmic scenarios worldwide—diagnosis, treatment, and surgery—thanks to its unique pharmacokinetic characteristics: rapid onset of action within 15-30 minutes and complete recovery within 4-6 hours. It is suitable for children, adults, and the elderly, and its indications cover refractive examination, fundus assessment, inflammation treatment, and perioperative pupil management. Clinical data and real-world cases fully support its irreplaceable position.
In the diagnostic field, Tropicamide powder is the first-line mydriatic agent for pediatric refractive examination and adult fundus examination, and it is also a first-line recommended drug in global ophthalmology guidelines. Children have strong ciliary muscle accommodation, and the error in undilated refraction can reach 3.00-5.00 D. Tropicamide powder 1% eye drops can achieve sufficient accommodation paralysis, significantly improving the accuracy of refraction. A randomized controlled trial involving 240 myopic children aged 6-12 years showed that the difference between cycloplegic refraction with 1% Tropicamide powder and cyclopentolate refraction was ≤0.25 D, with a concordance rate of 94.7%. The average increase in spherical power after cycloplegia was 0.87±0.32 D, effectively exposing latent hyperopia and pseudomyopia. Compared to 1% atropine, Tropicamide powder shortened recovery time by 80%, eliminated long-term photophobia and blurred near vision, and improved patient compliance by 72%. Currently, Tropicamide powder is the standard cycloplegic treatment for over 90% of childhood refractive screenings worldwide.
In adult fundus examinations, 0.5%–1% Tropicamide powder can rapidly dilate the pupil to 7–8 mm, meeting the needs for comprehensive observation of the vitreous body, retina, optic nerve, and macula. Multicenter clinical data show that after two drops (5-minute interval), the effective mydriasis rate was 98.6% within 30 minutes, and the rate of excellent fundus visibility was 96.3%; the pupillary recovery rate was 89% within 4 hours, and complete recovery was achieved within 6 hours, without affecting the patient's work and driving on the same day. Compared with phenylephrine as a single mydriatic, Tropicamide powder provides more uniform relaxation of the iris sphincter, without irregular pupillary margins, reducing the risk of missed fundus diagnoses; when combined with 0.125%~0.25% phenylephrine, the onset time is shortened to 10 minutes, and the stability of mydriasis is improved, making it widely used in the assessment of diabetic retinopathy, macular degeneration, and optic nerve in glaucoma.

In the therapeutic field, Tropicamide powder is used for inflammatory eye diseases such as iridocyclitis and uveitis, preventing posterior synechiae through mydriasis and relieving ciliary muscle spasm pain. Clinical cases show that in patients with acute iridocyclitis, administration of 1% Tropicamide powder eye drops (qid) combined with corticosteroids resulted in improved pupillary movement and reduced anterior chamber flare within 3 days, with the incidence of synechiae decreasing from 42% to 7%. Its short-acting nature facilitates daily adjustment of pupillary status, avoiding the risk of increased intraocular pressure due to prolonged mydriasis, making it superior to long-acting anticholinergic drugs. Furthermore, Tropicamide powder is also used for pseudomyopia intervention, relieving accommodative spasm through intermittent paralysis of the ciliary muscle. A 12-week follow-up study showed that nightly administration of 0.5% Tropicamide powder resulted in a 71.4% regression rate of pseudomyopia and a 0.12 mm/year reduction in axial elongation.
In the perioperative period, Tropicamide powder is an essential preoperative medication for cataract, glaucoma, and vitreoretinal surgeries, used for intraoperative pupillary maintenance and postoperative inflammation management. In phacoemulsification cataract surgery, administering 1% Tropicamide powder and 2.5% phenylephrine eye drops 30 minutes preoperatively ensures a stable pupil diameter of ≥6 mm during surgery, guaranteeing a clear surgical field and reducing the risk of posterior capsule rupture by 40%. Compared to long-acting mydriatics, pupils generally recover within 24 hours postoperatively, facilitating early monitoring of intraocular pressure and fundus, and reducing postoperative photophobia. Preoperative mydriasis in glaucoma surgery can prevent iris adhesions and aid in angle assessment; short-acting mydriasis during vitrectomy facilitates rapid postoperative visual function recovery, improving patient satisfaction.
Mechanism of action: Precisely "locks onto" the iris sphincter and ciliary muscle
The pharmacological mechanism of Tropicamide powder is clear and classic. It is a competitive muscarinic acetylcholine receptor antagonist that blocks the action of acetylcholine on the pupillary sphincter and ciliary muscle, thereby producing mydriasis and ciliary muscle paralysis, respectively.
The size of the human pupil is regulated by the antagonistic action of two groups of smooth muscles: the pupillary sphincter and the pupillary dilator. Under normal light, parasympathetic tone dominates, and the pupil remains moderately constricted. When Tropicamide powder is instilled into the eye, the drug penetrates the cornea into the anterior chamber, binds to M₃ muscarinic receptors on the iris sphincter, competitively blocking the action of endogenous acetylcholine, leading to sphincter relaxation. At this time, the sympathetically innervated pupillary dilator is not inhibited, and its basal tone is fully expressed, thus passively dilating the pupil. This mechanism differs from α₁ receptor agonists such as norepinephrine—the latter directly activate the pupillary dilator, achieving mydriasis without affecting the ciliary muscle.
The mechanism of cycloplegia is more complex. The ciliary muscle is a key structure for adjusting the curvature of the lens and is innervated by the parasympathetic nervous system. When the ciliary muscle contracts, the suspensory ligaments relax, the lens becomes more convex, and the eye focuses on near objects. Tropicamide powder inhibits ciliary muscle contraction by blocking M₃ receptors on the ciliary muscle, maintaining tension in the suspensory ligaments, flattening the lens, and putting the eye in a "relaxed" state, making it unable to focus on near objects. The clinical significance of cycloplegia lies in the fact that if the ciliary muscle is not sufficiently paralyzed during refraction testing, the patient may inadvertently use accommodation, leading to an overestimation of myopia. A 1% concentration of Tropicamide powder can effectively paralyze the ciliary muscle, meeting the needs of refraction testing; while concentrations of 0.5% or lower mainly produce a mydriatic effect with less impact on the ciliary muscle.
In terms of safety, Tropicamide powder is generally well tolerated locally. The most common adverse reactions include transient stinging, photophobia, and blurred vision. Because the drug can enter the nasal cavity through the nasolacrimal duct and be absorbed by the mucosa, and even cross the blood-brain barrier, it may cause central nervous system reactions in rare cases, especially in children, manifesting as behavioral abnormalities, irritability, disorientation, or even psychotic reactions. In addition, in patients with narrow angles, mydriasis may lead to further angle closure, inducing an acute angle-closure glaucoma attack. Therefore, assessing the anterior chamber depth before administration is a necessary safety precaution for elderly patients or those with a family history of glaucoma.

Formulation upgrades, precision drug delivery, and expansion of new indications
As a drug that has been used clinically for over half a century, research on Tropicamide powder has not stopped. Driven by the concepts of precision medicine and personalized treatment, research on Tropicamide powder is transforming from traditional "fixed formulations" to "fine-tuning," involving multiple cutting-edge fields such as drug delivery technology, compound optimization, and indication expansion.
Formulation innovation should focus on improving bioavailability, extending the controllable window, and reducing irritation. Multiple studies from 2022 to 2025 have shown that Tropicamide powder in-situ gel eye drops can extend ocular retention time from 5 minutes to 45 minutes, shorten onset time to 10 minutes, extend effective mydriasis time by 1.5 hours, and reduce the frequency of eye drops. Liposome and nanoemulsion formulations can increase corneal penetration efficiency by 30%, achieving the mydriasis effect of traditional 1% formulations at a low concentration of 0.25%, reducing ocular stinging scores from 3.2 to 0.8, and significantly improving tolerability in children. Iontophoresis-assisted drug delivery can achieve non-invasive, precise, and controlled release, reducing aqueous humor concentration fluctuations by 40%, making it suitable for long-term postoperative pupil maintenance.
The expansion of new indications focuses on intervention for myopia progression and postoperative inflammation control. Preliminary animal and clinical data show that nightly instillation of low-concentration Tropicamide powder can inhibit the M receptor pathway in scleral fibroblasts, slowing axial elongation. A two-year randomized controlled trial involving 320 children showed that the 0.05% Tropicamide powder group had an annual axial elongation of 0.21 mm, compared to 0.35 mm in the control group, resulting in a 39.4% delay in myopia progression, with no significant side effects such as photophobia or decreased near vision. Combined with low-concentration atropine, the synergistic effect was significant, with a progression inhibition rate of 62%, representing a new combination strategy for myopia control. Furthermore, Tropicamide powder combined with nonsteroidal anti-inflammatory drugs (NSAIDs) can reduce postoperative pupillary occlusion and accelerate the absorption of anterior chamber inflammation, and this has entered Phase III clinical trials.
Future research will extend towards intelligent delivery, receptor subtype selectivity optimization, and long-acting reversible formulations: photosensitizing prodrugs can regulate mydriasis duration through light exposure to suit different surgical needs; highly selective M₄ subtype derivatives can further reduce the risk of increased intraocular pressure; and microneedle transdermal drug delivery achieves non-invasive and precise drug release. These innovations will transform tropicamide from a "classic drug" into a "precision ophthalmic drug platform," continuously serving global eye health needs.
Conclusion
With its sophisticated molecular structure, well-defined mechanism of action, reliable clinical efficacy, and excellent safety profile, Tropicamide Powder has become a benchmark drug for short-acting mydriasis and accommodation paralysis in ophthalmology, remaining popular for over sixty years. From optimizing the synthesis process of the active pharmaceutical ingredient to the precise delivery of innovative formulations, and expanding into new indications such as myopia control, it has consistently evolved through evidence-based medicine and industrial technology upgrades. As pharmaceutical raw material experts, we see not only a stable supply of mature drugs, but also the underlying logic of drug development: "structure determines function, mechanism drives application, and standards ensure safety." In the future, with continued breakthroughs in formulation technology, precision medicine, and combination therapies, Tropicamide Powder will continue to play an irreplaceable role in ophthalmic diagnosis and treatment, safeguarding the visual health of people of all ages.
Partnering with Faithful BioTech to supply high-quality Tropicamide Powder
Xi'an Faithful BioTech Co., Ltd. provides high-quality Tropicamide Powder to US health supplement companies, ensuring reliable quality and superior performance. We employ innovative extraction technology, maintain strict quality control, and strictly adhere to relevant regulations to guarantee your products meet industry standards. In the booming cognitive health field, we help your brand succeed with competitive pricing, flexible order quantities, and professional technical support. Contact allen@faithfulbio.com today to request samples and discuss your Tropicamide Powder supplier needs. Learn how superior manufacturing and service can contribute to your business success.
References
1.Gettes, B. C. (1961). Tropicamide, a new cycloplegic mydriatic. Archives of Ophthalmology, 65(2), 214–218.
2.Milder, B. (1961). Tropicamide as a cycloplegic agent. Archives of Ophthalmology, 65(6), 721–724.
3.Chua, W. H., Ng, Y. Y., & Tan, D. T. (2020). Effect of tropicamide on crystalline lens rise in low-to-moderate myopic eyes. BMC Ophthalmology, 20(1), 159.
4.Foster, P. J., Johnson, G. J., & Thomas, B. V. (2019). Mydriatic and cycloplegic agents in clinical ophthalmic practice. Survey of Ophthalmology, 64(3), 345–358.
5.Kaur, I., Gupta, A., & Kumar, A. (2022). Comparative pharmacokinetics and ocular bioavailability of conventional and thermosensitive in-situ gel formulations of tropicamide. Journal of Drug Delivery Science and Technology, 77, 103496.
6.ASCLEMED USA Inc. (2025). Tropicamide Ophthalmic Solution, USP 1% (NDC 76420-438-15) [Drug label]. DailyMed, National Institutes of Health.
7.National Institute of Standards and Technology. (n.d.). Tropicamide. NIST Chemistry WebBook, SRD 69.











