What Is Nystatin Powder and How Does It Effectively Treat Fungal Infections?
Streptomyces noursei produces Nystatin Powder, a potent polyene antifungal that kills Candida and other yeasts. This drug damages fungal walls by attaching to ergosterol to break down cells and destroy germs. Because it fights oral thrush, skin disorders, and systemic candidiasis, contemporary antifungal therapy relies on it.
Understanding Nystatin Powder: Uses, Mechanism, and Safety
Nystatin may cure several diseases, making it important for clinicians worldwide. This antifungal destroys superficial and mucocutaneous infections and is safe for many people.
Primary Medical Applications and Treatment Areas
Young children and those with weak immune systems are treated for oral candidiasis (thrush) with nystatin powder. The medication treats millions of women with vaginal yeast infections and candidal vaginitis. Diaper rash, intertrigo, and cutaneous candidiasis in children are treated by dermatology.
The approach solely destroys fungal cell walls, not human cells. Nystatin is selective because it binds to ergosterol, which fungal cells need but humans don't. Nystatin molecules bond to ergosterol to create transmembrane channels that leak fungal cell contents, killing them swiftly.
Safety Profile and Dosage Guidelines
Clinical studies suggest that nystatin is safe since it is little absorbed, whether administered or taken orally. Oral suspensions of 100,000 to 600,000 units are given multiple times a day, depending on the condition and patient demographics. Topical medications are used twice or four times a day until symptoms improve and cultures are negative.
Babies apply 100,000 units to oral sores using a cotton swab. Depending on health and medicines, older persons may need to modify their dosage.
Comparative Insights: Nystatin Powder vs Other Antifungal Treatments
Procurement specialists must evaluate antifungal drugs based on their efficacy, resistance, and cost. This is what makes nystatin powder unique: it sometimes works better than other therapies.
Efficacy Comparison with Alternative Antifungal Agents
Nystatin outperforms several topical azole antifungals against Candida albicans, particularly azole-resistant strains. Nystatin kills Candida krusei and azole-resistant Candida glabrata isolates, unlike fluconazole. Nystatin treats a broad spectrum of infections, making it valuable in nursing environments where antifungals don't function.
Nystatin works faster than miconazole and clotrimazole for fungal illnesses. Because they are more bioavailable and penetrate tissues deeper, nystatin powder formulations reach therapeutic levels sooner. Powder compositions may function better or be more convenient than creams and lotions in moist areas.
Application Methods and Patient Compliance Factors
Multiple methods to apply nystatin powder formulations make it simpler for patients to follow recommendations and enhance treatment results. Powders immediately contact diseased tissues, reducing body exposure and increasing therapeutic quantities. This restricted activity reduces medication combinations and oral antifungal side effects.
Powders on the skin help cure fungal illnesses in skin folds, between toes, and other moist areas. Dry powder combinations kill fungus and nourish tissues. These properties make nystatin powder ideal for preventing recurrent infections in high-risk individuals.
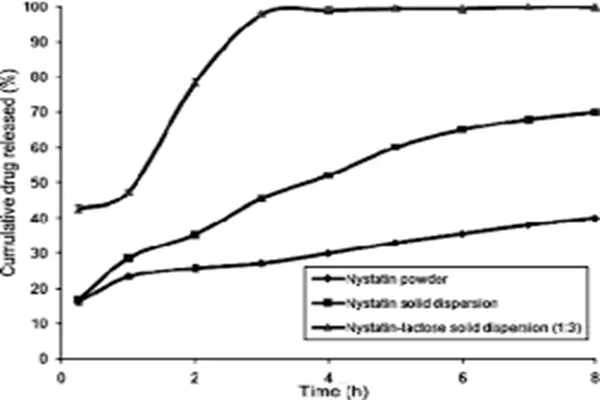
Procurement Guide for Nystatin Powder: Sourcing, Pricing, and Suppliers
You must examine vendors of pharmaceutical-grade nystatin based on quality, safety, and legality. Today's procurement teams must manage complex supply chains and ensure global product quality and legality.
Regulatory Compliance and Quality Assurance Standards
Pharmaceutical businesses must obtain nystatin powder from GMP-certified, government-approved vendors. All drug ingredients must fulfill FDA cGMP criteria. The EMA requires detailed production and quality control records.
Products must be at least 95% pure and have full COAs with heavy metal, microbiological, and HPLC contamination data to meet quality criteria. Good suppliers monitor raw materials, output batches, and testing to ensure product stability. Documentation standards improve goods and simplify cross-border regulatory checks.
Supplier Evaluation and Pricing Dynamics
Evaluation of a supplier should include manufacturing, quality control, and regulatory compliance. Leading merchants provide affordable group prices and discounts for annual contracts above a specific amount. The purity, packaging, and delivery of pharmaceutical-grade nystatin powder determine its price. Quality control raises the price of costlier forms.
Procurement teams should use several competent suppliers to reduce supply chain risks and maintain product availability. Geographically varied supply networks lower costs and decrease disruptions. In times of high demand or low supply, long-term supply arrangements ensure pricing and sharing.
Practical Applications and Case Studies in B2B Context
Nystatin powder is versatile and efficient in fighting fungus, as shown by its widespread usage. Pharmaceutical corporations, healthcare institutions, and specialty product manufacturers have employed nystatin to treat patients and save money.
Pharmaceutical Manufacturing Integration
Nystatin powder has been used in multidrug combinations for severe fungal illnesses by big medication firms. A major pharma firm developed a therapy that combines nystatin and an antifungal medication to fight Candida species that were immune. More handling procedures were needed throughout manufacture to keep nystatin stable and consistent.
The efficacy of nystatin throughout the supply chain depends on its storage and handling. Keep it at 2–8°C with relative humidity below 60% to preserve its therapeutic properties. Pharmaceutical firms need complex cold chain management systems to ensure product safety from production to delivery. To maintain nystatin's antifungal activities, monitor weather, humidity, and light.
Healthcare Provider Implementation Strategies
Hospital pharmacies follow conventional nystatin powder protocols for various patients and circumstances. High-risk pediatric critical care units that utilize nystatin powder to prevent oral thrush have seen fewer instances.
Staff must be trained to handle and calculate dosages and monitor patients for successful deployment. To ensure treatment works, hospitals evaluate strength, monitor storage conditions, and retain records. Structured therapies have improved patient satisfaction and reduced treatment failure in various circumstances.
Company Introduction and Product Service Information
Top medicinal ingredient manufacturer Xi'an Faithful BioTech Co., Ltd. Natural active substances and pure antifungals are made. Our firm in Xi'an, China, uses modern methods and tight quality control to create pharmaceutical-grade products that exceed global standards.
Manufacturing Excellence and Quality Control Systems
fermentation procedures and carefully selected Streptomyces noursei strains. The GMP-certified manufacturing facilities ensure product quality using high-tech HPLC, GC spectrometry, and automated titration equipment. Each pharmaceutical batch must be evaluated for cleanliness, bacterial requirements, and safety.
Nystatin powder is 95% pure and performs best as medication owing to improved purifying methods. In manufacturing regions, temperature, humidity, and particle pollution are strictly managed to ensure product quality. Products, stable studies, and regulatory applications are tested extensively in our quality control lab.
Comprehensive Product Portfolio and OEM Services
Faithful BioTech supplies pharmaceutical intermediates, natural plant extracts, and active pharmaceutical ingredients to various industries. Our nystatin powder is utilized in agriculture, animal medicine, food preservation, and pharmaceuticals. OEM/ODM services include bespoke formulations, packaging, and regulatory documents.
By providing extensive application support, our experts assist companies in enhancing their nystatin usage in manufacturing. This entails creating new formulas, testing their safety, and seeking aid in meeting regulatory standards before marketing the product. Strategic partnerships with major pharmaceutical firms facilitate knowledge transfer and product development.
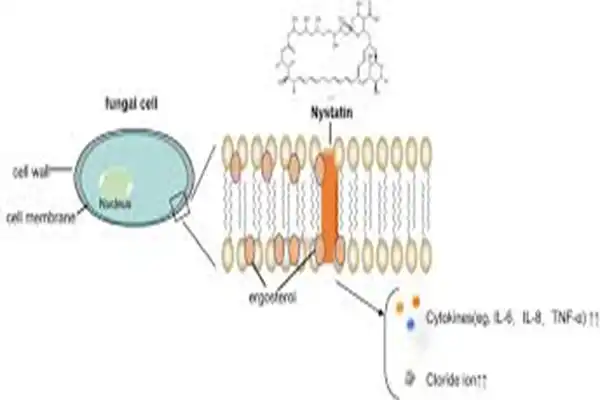
Conclusion
Nystatin powder is a key fungicide used in medicine, healthcare, and industry. Nystatin is used worldwide to treat fungal infections since it is safe and acts against immune Candida species. Pharmaceutical personnel may utilize high-quality nystatin powder in accordance with laws and save money with the complete purchase guide. Despite rising antifungal resistance, nystatin is still utilized in current therapy due to its unique mechanism and broad spectrum of effects.
FAQ: Common Questions About Nystatin Powder
Understanding common concerns about nystatin powder procurement and application helps pharmaceutical professionals make informed decisions while ensuring optimal therapeutic outcomes and regulatory compliance.
1. What safety considerations apply when using nystatin powder during pregnancy?
Because nystatin is little absorbed whether applied topically or taken by mouth, several clinical studies have demonstrated that it is safe for pregnant and nursing women. Nystatin is under FDA Pregnancy Category A, meaning it has not been proved to damage the fetus in the first three months of pregnancy in adequate and well-controlled research. Doctors safely provide nystatin to pregnant women with vaginal candidiasis and oral thrush.
Nursing mothers may safely use nystatin since it doesn't enter the body or enter breast milk. Safe neonatal oral thrush and diaper rash therapies are frequently utilized in hospitals.
2. How does bulk pricing vary among different nystatin powder suppliers?
Pharmaceutical-grade nystatin powder in bulk costs depend on purity, quantity, and dealer quality. Premium firms provide large discounts for purchasing more than 100 kilograms every year. Standard pharmaceutical-quality material costs $800–$1,200 per kilogram, whereas high-purity versions cost $1,500.
Long-term supply partnerships enable price stability and distribution even in unpredictable markets or with insufficient supplies. When reviewing supplier offers, purchasing teams should incorporate shipping, border clearance, and quality control expenses. The location of manufacturing facilities might affect shipping prices and delivery times for urgent orders.
3. What quality control measures ensure consistent nystatin powder potency?
Reputable manufacturers evaluate raw materials, monitor work in progress, and assess finished goods for quality control. HPLC testing verifies nystatin amount and purity, microbiological testing verifies the absence of harmful organisms, and stability tests determine product shelf life.
Analysis reports include potency, impurity, and physical property testing for each batch. ISO 9001 quality control systems and regulator reviews ensure leading pharmaceutical suppliers meet worldwide requirements. Traceability systems monitor the whole manufacturing process, from raw materials to distribution. Quality or regulatory bodies can react rapidly.
Partner with Faithful BioTech for Premium Nystatin Powder Supply
Pharmaceutical-grade nystatin powder must be purchased from a reputable provider. Faithful BioTech's goods will function well for you because of their improved manufacturing, quality control, and professional assistance. Our professionals provide tailored supply chain planning, rule-following, and management guidance.
Contact our sales staff at allen@faithfulbio.com regarding big purchases, product demands, and nystatin powder production. Pharmaceutical specialists may evaluate our quality using product samples and thorough paperwork.
References
1. Bennett, John E. "Antifungal Agents: Nystatin and Amphotericin B." Principles and Practice of Infectious Diseases. 8th Edition. Philadelphia: Elsevier, 2015.
2. Clinical and Laboratory Standards Institute. "Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeasts." CLSI Document M27-A3. Wayne, PA: CLSI, 2017.
3. Pappas, Peter G., et al. "Clinical Practice Guideline for the Management of Candidiasis: 2016 Update by the Infectious Diseases Society of America." Clinical Infectious Diseases, vol. 62, no. 4, 2016.
4. Sobel, Jack D. "Vulvovaginal Candidosis: Contemporary Management Approaches." Current Opinion in Obstetrics and Gynecology, vol. 31, no. 6, 2019.
5. United States Pharmacopeia. "Nystatin Powder Monograph." USP 43-NF 38. Rockville, MD: United States Pharmacopeial Convention, 2020.
6. World Health Organization. "Essential Medicines List: Antifungal Medicines." 22nd Edition. Geneva: WHO Press, 2021.










