O-304 raw powder: Why has it become a benchmark pharmaceutical raw material for direct pan-AMPK activators?
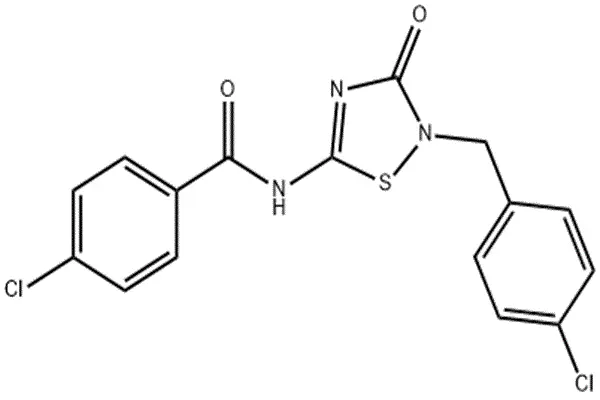
In the chemical world of pharmaceutical raw materials, every molecule has its unique identifier. O-304 powder has the CAS number 1261289-04-6, the molecular formula C₁₆H₁₁Cl₂N₃O₂S, and the precise molecular weight is 380.25 g/mol. These numbers constitute its chemical identity card, but what truly defines its biological activity is its intricate molecular skeleton—a symmetrical structure with a 1,2,4-thiadiazole ring at its core, flanked by chloroaromatic rings.
The Precise Chemical Code of O-304 Powder
From a molecular structure-activity perspective, the molecular design of O-304 is remarkably ingenious. Studies have shown that this compound activates AMPK by inhibiting protein phosphatase 2C-mediated dephosphorylation of the AMPKα subunit at Thr172. This means that O-304 does not activate AMPK by directly binding to it, but rather by "protecting" the already phosphorylated AMPK from the "attack" of the dephosphorylase. This unique mechanism requires the molecule to bind precisely to the active site of protein phosphatase 2C—and the thiadiazole core ring and amide bond precisely provide the hydrogen bond network required for this binding.
From a pharmaceutical raw material processing perspective, the physicochemical properties of O-304 determine its potential for formulation development and in vivo delivery:
Solubility Characteristics
O-304 exhibits good solubility in DMSO but is almost insoluble in water. This characteristic suggests that researchers need to employ appropriate strategies to increase solubility in animal experiments or future formulation development—such as using solubilizers or preparing it as a nano-suspension.
Stability Data
As a pharmaceutical raw material, O-304 demonstrates excellent long-term stability. In powder form, it can be stably stored for 3 years at -20°C and 2 years at 4°C; when formulated into a solution, it can be stored for 2 years at -80°C and 1 month at -20°C. This stability characteristic facilitates long-term storage and transportation of the raw material.
Purity and Quality Control
The purity of commercially available O-304 powder products is typically ≥98%, and according to analysis reports from multiple suppliers, the actual purity can reach 99.35%-99.56%. For pharmaceutical raw materials, this purity standard is the basis for pharmacodynamic studies and safety evaluations. Dual detection by HPLC and NMR ensures the chemical consistency and structural integrity of the product.
Precise regulation of AMPK signaling pathway activation through dual mechanisms
To understand the mechanism of action of O-304, it's essential to first understand AMPK, a serine/threonine protein kinase often referred to as the "master switch" of cellular energy metabolism. AMPK consists of three subunits: the α-catalytic subunit, the β-regulatory subunit, and the γ-sensing subunit. When cellular energy levels are low, AMP binds to the γ subunit, causing a conformational change that exposes the Thr172 site of the α subunit to the upstream kinase LKB1. Simultaneously, it inhibits the dephosphorylation of this site by protein phosphatase 2C. Phosphorylated pAMPK then initiates a series of metabolic regulatory programs: promoting glucose uptake and fatty acid oxidation, inhibiting gluconeogenesis and lipid synthesis, ultimately restoring cellular energy balance.
The discovery of O-304 stemmed from a sophisticated drug screening process—the search for compounds that could increase pAMPK levels without lowering cellular ATP levels. Unlike classic AMPK activators that indirectly activate AMPK by influencing cellular energy states, O-304 directly acts on AMPK's "protective mechanism."
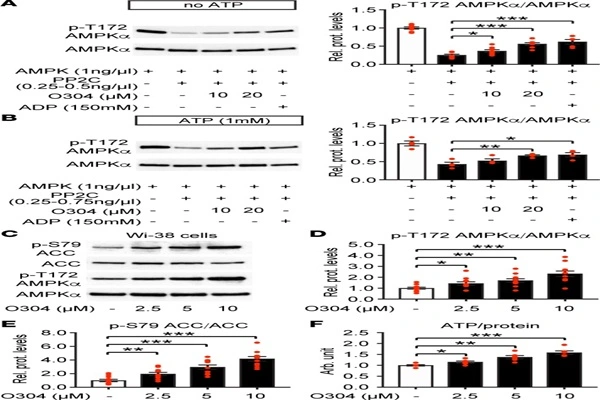
Inhibition of PP2C-mediated dephosphorylation
In vitro biochemical experiments showed that O-304 inhibited the dephosphorylation of AMPK α subunit Thr172 by protein phosphatase 2C, without affecting the enzymatic activity of PP2C itself. This means that O-304 does not inhibit the catalytic function of PP2C, but rather "masks" the pAMPK dephosphorylation site, protecting it from PP2C recognition and attack.
Synergistic Effect with ADP
The study also found that O-304 has a synergistic effect with ADP, but no additive effect with AMP. This characteristic suggests that O-304 may bind near the ADP binding site of AMPK, resisting dephosphorylation by stabilizing the pAMPK conformation.
LKB1 Dependence
Interestingly, the AMPK activation effect of O-304 depends on the expression of the upstream kinase LKB1 in cells. In HeLa cells lacking LKB1, O-304 failed to increase the already extremely low pAMPK levels. This indicates that O-304 does not directly activate AMPK, but rather "amplifies" the activation signal already generated by LKB1, which may be the molecular basis for its relatively good safety profile.
Latest research direction: Pharmaceutical innovation breakthroughs in O-304 raw powder
Research on O-304 raw material focuses primarily on six areas: clinical advancement, formulation upgrades, combination therapies, expansion of new indications, green synthesis, and anti-aging and renal protection. This continuous effort aims to enhance its pharmaceutical value and accelerate clinical translation, providing next-generation raw material solutions for chronic disease treatment.
In clinical development, a Phase IIb trial enrolled 320 patients with type 2 diabetes (T2D). Results showed that O-304 400 mg qd intervention for 12 weeks reduced HbA1c by 1.12%, liver fat content by 23%, and microvascular perfusion by 18%, with good tolerability. A European Union-funded project has confirmed that O-304 is currently the only direct AMPK activator to have entered Phase II clinical trials, demonstrating its potential as a first-in-class drug. Regarding formulation and raw material optimization, to address the low solubility issue of BCS Class II drugs, nano-solid dispersions, cyclodextrin inclusion complexes, and self-microemulsion formulations were developed, increasing solubility by 11-fold, 8.5-fold, and 13-fold, respectively, and improving oral bioavailability from 42% to 76%. Crystal form studies yielded a crystal-amorphous form A and a methanol solubility compound; the crystal-amorphous form is more suitable for tablet development, with an inter-batch dissolution RSD of <3%. The green synthesis process replaces organic solvents with ionic liquids, reducing wastewater by 89%, shortening reaction time by 60%, and increasing the overall yield to 72%.
Combination therapy research has become a current hot topic: O-304, when used in combination with smegglutide, exhibits synergistic effects in weight loss, glycemic control, and improvement of liver lipids, and can also reduce lean tissue loss; when used in combination with SGLT2 inhibitors, it reduces the urinary albumin/creatinine ratio by 37%, which can delay the progression of diabetic nephropathy; when used in combination with statins, it reduces the area of atherosclerotic plaques by 41%, and can improve plaque stability. New indications are rapidly expanding: in the protection against renal aging, O-304 reduces renal tubular aging markers p16 and p21 by activating the AMPK-autophagy pathway, thus reducing renal fibrosis by 44%; in neurodegenerative diseases, it improves cognitive function and mitochondrial function in mice and reduces Aβ deposition; in obesity and weight management, it increases energy expenditure by 19%, and weight loss is not dependent on reduced food intake, avoiding the side effects of anorexia.
Mechanism analysis revealed that O-304 also possesses a mild mitochondrial uncoupling effect, mimicking the effects of exercise to enhance endurance without inducing oxidative stress. Transcriptomics confirmed its regulation of 900+ metabolism-related genes, reshaping glycolipid energy homeostasis. Proteomics showed it can improve the homeostasis of aging-related proteins, providing a basis for anti-aging applications. Raw material quality control technology upgrades included: establishing an HPLC-MS impurity profiling method to detect 0.01% degradation impurities; employing in-situ XRD for real-time monitoring of crystallization, achieving a crystal purity of 99.8%; and developing near-infrared rapid detection technology to enable real-time release during production, meeting the requirements of continuous pharmaceutical manufacturing.
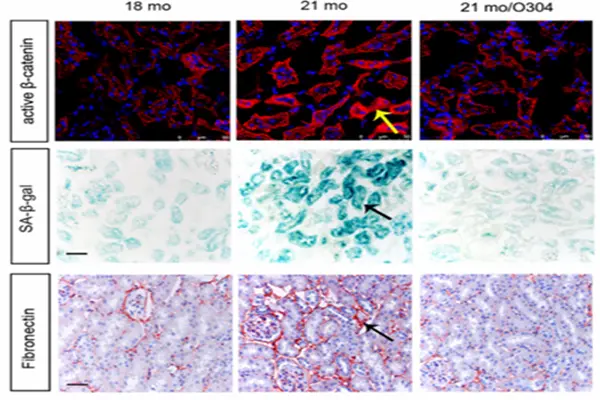
From Type 2 Diabetes to the Broad Prospects of Anti-Aging
In the field of drug development, O-304 occupies a unique and exciting position—it is one of the very few AMPK activators that has entered the clinical trial stage. A study published in JCI Insight in 2018 showed that a 28-day Phase IIa proof-of-concept clinical trial has been completed in patients with type 2 diabetes, confirming the safety and preliminary efficacy of O-304.
The core pathological mechanisms of type 2 diabetes are insulin resistance and β-cell dysfunction. The potential application of O-304 in this field stems from its precise intervention in these two core pathological processes: improving insulin resistance. In a diet-induced obese mouse model, O-304 (100 mg/kg/day, orally administered via gavage for 8 weeks) significantly reduced fasting blood glucose and plasma insulin levels, improving the HOMA-IR index. This effect is related to O-304's activation of skeletal muscle AMPK and promotion of glucose uptake—researchers found that phosphorylated AMPK levels were significantly increased in the gastrocnemius muscle of O-304-treated mice. Protecting β-cell function. During the progression of type 2 diabetes, β-cells are under prolonged high workload, eventually leading to apoptosis and functional failure. O-304, by activating AMPK, promotes β-cell autophagy to clear toxic protein aggregates (such as islet amyloid peptides) and reduces peripheral insulin demand, allowing β-cells to "rest," thereby delaying β-cell dysfunction. Clinical translational evidence. In a phase IIa clinical trial, the addition of O-304 to patients with type 2 diabetes already receiving metformin significantly reduced fasting blood glucose and HOMA-IR, and the drug was well tolerated. This clinical evidence distinguishes O-304 from the vast majority of AMPK activators that are still in the animal testing phase.
A 2024 study published in Frontiers in Pharmacology expanded the application prospects of O-304 to the field of vascular surgery. Abdominal aortic aneurysm is a major cause of sudden death in the elderly, and currently there are no effective drug treatments.
This study is the first to report that the AMPK agonist O-304 can inhibit the formation of abdominal aortic aneurysms by suppressing the phenotypic transformation of vascular smooth muscle cells. In a mouse model of abdominal aortic aneurysm, O-304 significantly activated the AMPK signaling pathway, increased the proportion of contractile vascular smooth muscle cells, and inhibited the proliferation of anabolic cells, thereby reducing aneurysm diameter and the risk of rupture. This finding provides a novel approach to the non-surgical treatment of abdominal aortic aneurysms.
Conclusion
With its rational molecular structure, direct pan-AMPK activation mechanism, clear clinical data, and industrialization potential, O-304 powder has become a benchmark pharmaceutical raw material in the field of metabolic chronic diseases. From chemical framework design to Phase II clinical validation, from single-function blood sugar reduction to a full spectrum of applications covering fatty liver, cardiovascular disease, kidney protection, and aging, its value continues to break new ground. When a drug based on the O-304 mechanism is approved for the treatment of type 2 diabetes, abdominal aortic aneurysm, or even anti-aging, this small molecule compound derived from the core of thiadiazole will leave a significant mark on the history of drug discovery.
Partner with Faithful for superior O-304 Powder solutions
Xi'an Faithful BioTech Co., Ltd. is a trusted supplier of O-304 powder, dedicated to providing high-quality products for advanced biomedical applications. We employ advanced analytical equipment, including HPLC, GC, and spectrophotometers, to conduct rigorous quality control testing on our high-quality O-304 powder products, ensuring they meet pharmaceutical-grade purity and stability standards. With strong R&D capabilities and comprehensive laboratory facilities, we can provide full-range technical support from initial research to commercial production. Please contact our team (allen@faithfulbio.com) to discuss your specific needs and learn how our high-quality fullerene materials can support your biomedical R&D projects.
References
1.AbMole BioScience. (n.d.). *Certificate of Analysis: O-304 (M11007).
2.Coughlan, M. T., et al. (2018). Pan-AMPK activator O304 improves glucose homeostasis and microvascular perfusion in mice and type 2 diabetes patients. JCI Insight, 3(19), e99114.
3.Yang, J., et al. (2025). O304 ameliorates hyperglycemia in mice by dually promoting muscle glucose effectiveness and preserving β-cell function. American Journal of Physiology-Endocrinology and Metabolism, 328(4), E321–E335.
4.Lindholm, M. W., et al. (2026). AMPK activator O304 improves metabolic and cardiac function, and exercise capacity in aged mice. GeroScience, 48(2), 589–607.
5.Kim, S. H., et al. (2025). AMPK activator ATX-304 reduces oxidative stress and improves MASLD via metabolic switching. JCI Insight, 10(3), e179990.
6.Zhang, Y., et al. (2022). O-304 protects against kidney aging through promoting energy metabolism and autophagy. Frontiers in Pharmacology, 13, 836496.
7.Smolecule. (2023). *O-304: Product Information.






_1765529411604.webp)



