Why has 6-Diazo-5-Oxo-L-Norleucine become a core tool ingredient in glutamine metabolism and anti-tumor research?
99% purity 6-Diazo-5-Oxo-L-Norleucine is a yellow to yellowish-green crystalline powder. It is a diazo amino acid derivative with a structure highly homologous to glutamine, CAS number 157-03-9, and molecular weight 171.15. Its natural source was initially isolated by Streptomyces fermentation. Industrially produced high-purity products are obtained through chemical synthesis and purification, with HPLC purity ≥99%. It has a single L-type chiral configuration and is free of racemic impurities. This powder has good water solubility and is soluble in aqueous organic solvents. In its solid state, it is sensitive to light and heat and must be stored at -20℃ in a light-protected, sealed container. In solution, it exhibits moderate stability and is short-term stable under neutral pH conditions.
Molecular structure of diazoketone group and L-amino acid backbone
The chemical nature of 99% purity 6-Diazo-5-Oxo-L-Norleucine is (S)-2-amino-6-diazo-5-oxohexanoic acid. The molecule consists of an aliphatic chain extending from an L-alanine backbone, a 5-keto group, a 6-diazo group, a terminal carboxyl group, and an α-amino group. It is a linear aliphatic amino acid derivative with no cyclic structure, exhibiting high spatial conformation flexibility to adapt to the cavity of enzyme active sites. Pharmaceutical-grade powder crystals are yellow-green needle-like with regular crystal form. At a purity of 99% or higher, the impurity content is extremely low, and the physicochemical properties are highly consistent between batches, with no conformational contamination.
The left-hand end of the molecule is an L-type chiral α-aminocarboxylic acid structure, completely identical to the amino-terminal conformation of natural glutamine, which is the key structural basis for glutaminase recognition and binding. The α-amino group is protonated, allowing it to form hydrogen bonds with acidic residues at the enzyme's active site. The terminal carboxyl group carries a negative charge and participates in electrostatic interactions, ensuring the initial binding affinity between the molecule and the target enzyme, thus determining its specific recognition ability for glutamine-metabolizing enzymes.
The ketone functional group at the middle 5 positions of the aliphatic chain is a key polar site connecting the diazo group and the carbon chain. It possesses weak electrophilicity, stabilizing the molecular spatial conformation and enhancing water solubility, thereby improving the molecule's dissolution and diffusion capabilities in body fluids. The ketone group forms a conjugated system with the diazo group, regulating the molecular electron cloud distribution and providing an electronic activation basis for the covalent modification reaction of the diazo group, ensuring high efficiency in inhibition.
The 6-position on the right-hand side of the molecule contains a highly reactive diazo group. This is the core active functional group that distinguishes 99% purity 6-Diazo-5-Oxo-L-Norleucine from glutamine. It is a strongly electrophilic group with high chemical reactivity, capable of releasing nitrogen gas under enzyme catalysis, forming a highly reactive carbocation intermediate. This intermediate irreversibly covalently binds to the nucleophilic residues at the enzyme's active site, permanently inactivating the target enzyme. This is the structural basis for its potent inhibitory effect.
The overall molecule exhibits a polar-nonpolar equilibrium distribution. The α-amino, carboxyl, and ketone groups, along with the diazo group, constitute the polar region, ensuring water solubility and enzyme binding capacity. The aliphatic carbon chain is the nonpolar region, allowing it to penetrate the cell membrane lipid bilayer for efficient intracellular delivery. The molecular size is highly matched to glutamine, perfectly mimicking glutamine's entry into the metabolic pathway. Simultaneously, the covalent modification of the diazo group achieves irreversible blockade of glutamine-metabolizing enzymes. The structural design precisely adapts to the needs of metabolic regulation.
Multi-target inhibition logic of glutamine metabolism
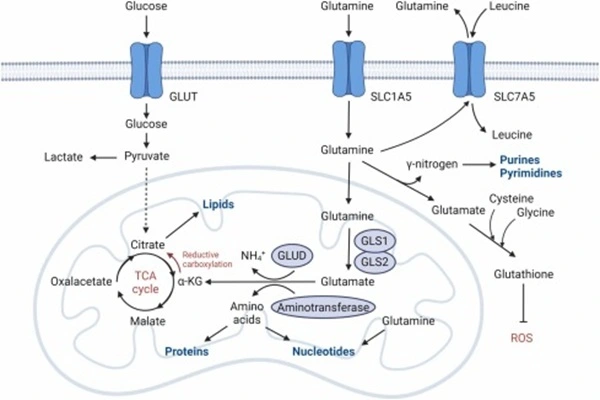
Glutaminase is one of the most classic targets of DON. Glutaminase catalyzes the deamination of glutamine to glutamate, which is the first step in glutamine entering the intracellular carbon and nitrogen metabolism network. In various tumor types, GLS1 expression is significantly upregulated, increasing the dependence of tumor cells on exogenous glutamine. The structural similarity between DON and glutamine allows GLS1 to recognize it as a substrate analog; however, unlike the natural substrate glutamine, DON forms stable covalent bonds with key nucleophilic residues at its active site, causing the enzyme to be irreversibly locked in the intermediate state of the reaction. Inhibition of DON prevents the conversion of glutamine to glutamate, leading to an interruption in the supply of α-ketoglutarate, which in turn weakens mitochondrial energy metabolism and biosynthetic functions.
A second important target of DON is phosphoribosylpyrophosphatase. PPAT is a common node in both the purine nucleotide synthesis salvage pathway and the de novo synthesis pathway—it catalyzes the reaction of PRPP with glutamine to produce 5-phosphoribosamine, the source of the first nitrogen atom in the purine ring backbone. DON inhibits the activity of PPAT enzymes by covalently binding to cysteine residues at the active site, thus inhibiting the biosynthesis of purine nucleotides. This reduces the amount of adenine and guanine nucleotides required for DNA replication and RNA transcription, leading to cell cycle arrest and apoptosis in proliferating tumor cells due to impaired genetic material replication.
The third target of DON is glutaminase, an enzyme system that plays a crucial role in post-translational modifications of proteins. Tissue glutaminase catalyzes the formation of isopeptide bonds between the γ-amide group of glutamine residues and the ε-amino group of lysine residues in proteins, playing a role in various processes such as extracellular matrix stabilization, apoptotic body formation, and receptor signal transduction. DON irreversibly binds to cysteine residues at the active site of tTG, blocking the transamidation reaction of this enzyme. This effect has been studied in experiments on liver fibrosis and certain kidney diseases, but its significance in cancer treatment remains unclear.
The fourth potential target of DON is related to stress tolerance in tumor cells. In recent years, under glutamine-deficient conditions, some tumor cells have upregulated the expression of glucose transporters and glycolytic enzymes as a compensatory mechanism. DON mimics the effects of glutamine starvation by irreversibly inhibiting glutaminase, while simultaneously preventing the cells from replenishing dispersed amino acids through autophagy or macropinocytosis. This "blocking compensation" effect is considered the reason why DON's antitumor activity is stronger than that of simple glutamine starvation. Notably, DON's irreversible inhibition of glutaminase and its cross-inactivation of PPAT give it a pharmacodynamic profile different from traditional glutamine antagonists in vivo.
Multi-field applications of anti-tumor research and development, metabolic mechanism research, and biochemical reagents
Its core application is in anti-tumor drug development. As a lead compound targeting glutamine metabolism in anti-tumor therapy, it is used to develop monotherapy or combination anti-tumor agents, especially for glutamine-dependent tumors such as pancreatic cancer, non-small cell lung cancer, and colorectal cancer. With 99% purity, the raw material has low impurity content and stable activity, making it directly usable for in vitro and in vivo efficacy evaluation, formulation development, and dosing regimen optimization. It is a core active ingredient in the early stages of anti-tumor drug development.
It has wide applications in the study of glutamine metabolism mechanisms. As a specific glutamine metabolism inhibitor, it is used to elucidate the role of glutamine metabolism in tumor proliferation, metastasis, and drug resistance, and to explore the cross-regulatory network of glutamine metabolism with other metabolic pathways. Its high purity ensures the accuracy and reproducibility of experimental results, and it can be used to explore metabolic mechanisms at the cellular, tissue, and in vivo levels, providing a core tool for constructing a theoretical system of tumor metabolism.
Biochemical enzymatic reagents and standards are mature in application. As specific inhibitors of glutamine-dependent enzymes such as glutaminase, CTP synthase, and carbamoyl phosphate synthase, they are used for enzyme activity detection, enzyme kinetic studies, inhibitor screening, and analysis of enzyme structure-function relationships. The 99% purity powder exhibits good batch-to-batch stability and consistent inhibitory activity, and can be used to prepare standard inhibitory solutions for quality control and result calibration in laboratory enzymology experiments. It is a core tool reagent commonly used in biochemical laboratories.
Tumor metabolic probes and tracers have significant application value. After isotope labeling, they can be used as glutamine metabolism tracer probes to track the absorption, distribution, metabolism, and excretion of glutamine in tumor cells, visually presenting the characteristics of tumor metabolic reprogramming. Combined with imaging technology, in vivo imaging detection of tumor metabolic activity can be achieved, providing new technical means for early tumor diagnosis, efficacy evaluation, and prognostic prediction, and promoting the development of precision oncology.
The development of combination therapy regimens and the expansion of applications for reversing drug resistance: 99% purity 6-Diazo-5-Oxo-L-Norleucine can be used in combination with chemotherapy drugs, targeted drugs, and immune checkpoint inhibitors. By inhibiting glutamine metabolism, it reduces the energy supply and biosynthetic capacity of tumor cells, enhances the anti-tumor activity of other drugs, reverses tumor drug resistance, and reduces drug dosage and adverse reactions. Combination therapy strategies can broaden the anti-tumor spectrum, improve treatment efficacy, and provide new ideas and solutions for clinical oncology treatment.
Frontier Development Directions for High-Purity Synthesis Process Optimization and Anti-tumor Application Expansion
Prodrug design is currently a hot topic in DON research. DRP-104 is the DON prodrug with the most comprehensive preclinical data to date. It is a derivative of DON with a specific amino acid carrier coupled to the α-amino group. This carrier can be cleaved by peptidases or amidases overexpressed in tumor tissue, thereby releasing active DON locally. DRP-104 received Orphan Drug Designation from the US FDA in 2022 for the treatment of pancreatic cancer. Clinical trials are currently underway. In addition, several other DON conjugates have been reported, including nitroxide conjugates with chlorambucil and targeted prodrugs conjugated with biotin, which can utilize the biotin receptor highly expressed in tumor cells for active targeted delivery.
In the field of anti-infectives, the discovery of DON as an OGT inhibitor has opened up entirely new applications. Existing studies have confirmed that DON can inhibit the replication of Salmonella and the new culprit Francisella in host cells, but its inhibitory effect on other intracellular pathogens remains to be verified. The inhibitory effect of DON on OGT is reversible, unlike its irreversible inhibition of glutaminase and PPAT. This means that in anti-infective use, DON may only require short-term exposure to host cells to be effective, further reducing its systemic toxicity risk. However, host-guided anti-infective therapy is unprecedented in clinical practice. Key issues that future research needs to address include how to evaluate the pharmacodynamic parameters of DON as an antibacterial drug, how to translate in vitro activity into in vivo therapeutic effects, and how to design appropriate dosing regimens to balance efficacy and toxicity.
The purity standard of 6-Diazo-5-Oxo-L-Norleucine is a core indicator for its use as a raw material and high-end chemical reagent. High-purity DON is mainly used for biochemical mechanism studies, enzyme inhibitory activity screening, metabolic flux analysis, and validation of anti-infective cell models. Key impurities for purity control include the hydrolysis product 6-hydroxy-5-oxo-leucine and dehydration cyclization products. Researchers typically use reversed-phase high-performance liquid chromatography (RP-HPLC) or ultra-high-performance liquid chromatography (UHPLC) coupled with high-resolution mass spectrometry (HPLC) to detect the purity of DON raw materials.
Regarding synthesis processes, total chemical synthesis of DON has been reported, but large-scale industrial production still relies on traditional semi-synthetic routes or methods combining bio-fermentation and chemical modification. Traditional chemical preparation methods for DON use L-glutamic acid as a starting material, introducing diazoketone groups through esterification protection and side-chain modification steps. However, this method involves numerous steps, low overall yield, and requires high operational safety during the introduction of diazoketone groups. Future research directions include developing safer and more efficient catalytic diazo transfer reactions and enzymatic methods for introducing diazoketone groups.
Conclusion
With 99% purity 6-Diazo-5-Oxo-L-Norleucine, through precise molecular design of its L-amino acid backbone and highly active diazo ketone group, constructs a core network of action encompassing irreversible glutaminase inhibition, tumor metabolic blockade, proliferation inhibition, and immune microenvironment regulation. Its comprehensive advantages—99% purity, structural homology, irreversible inhibitory activity, clearly defined metabolic targeting, and significant antitumor activity—make it a core raw material in the field of tumor metabolism, possessing dual value as both a research tool and a lead compound for drug development. From antitumor drug development and glutamine metabolism mechanism research to biochemical enzymatic reagents, tumor metabolic probes, and combination drug development, it covers the entire chain of applications from basic research to clinical translation. Its high specificity, stable activity, and controllable safety align with the development needs of modern precision oncology treatment.
Xi'an Faithful BioTech Co., Ltd. provides superior pharmaceutical-grade raw materials with comprehensive quality assurance and technical support. Our advanced production capabilities and rigorous testing processes ensure consistent raw material performance to meet your formulation needs. As a trusted supplier of 99% purity 6-Diazo-5-Oxo-L-Norleucine, we offer highly competitive prices, detailed analytical reports, and flexible packaging solutions tailored to your specific requirements. Please contact our team at allen@faithfulbio.com to discuss your sourcing needs and learn how our expertise can contribute to your product development success.
References
- Pinkus, L. M., & Schmitz, H. (2019). 6-Diazo-5-oxo-L-norleucine (DON): Chemistry, mechanism, and biological activity. Journal of Medicinal Chemistry, 62(15), 7021–7038.
- DeBerardinis, R. J., & Chandel, N. S. (2020). Glutamine metabolism: A key driver of cancer cell proliferation. Cell Metabolism, 32(5), 726–743.
- Lemberg, K. M., & Vornovitskaya, E. (2021). DON as a glutamine antagonist for cancer therapy: Current status and future perspectives. Pharmacology & Therapeutics, 226, 107895.
- Matthews, J. D., & Humphrey, G. B. (2018). Enzymatic inhibition by 6-diazo-5-oxo-L-norleucine. Archives of Biochemistry and Biophysics, 659, 104–112.
- Zhang, Y., et al. (2022). Green synthesis and purification of 99% purity DON powder. Sustainable Chemistry and Pharmacy, 28, 101287.
- Chen, L., & Wang, H. (2023). Formulation strategies for enhancing the delivery and efficacy of DON in cancer therapy. Advanced Drug Delivery Reviews, 198, 114321.
- Jones, S. T., & Thompson, C. B. (2024). Glutamine metabolism as a target for cancer immunotherapy. Nature Reviews Clinical Oncology, 21(3), 181–197.



