Why has Selank become a new benchmark for non-addictive anti-anxiety and neurorepair peptides?
Selank (CAS: 129954-34-3) is a synthetic heptapeptide neuromodulatory active pharmaceutical ingredient developed by the Russian Academy of Sciences. Modified from the natural immune peptide Tuftsin, it possesses multiple functions including anti-anxiety, cognitive enhancement, neuroprotection, immune regulation, and HPA axis homeostasis control. It completely avoids the addiction, drowsiness, and cognitive impairment associated with benzodiazepines, pioneering a new route of "endogenous mimicry and multi-target synergy" in the psychotropic active pharmaceutical ingredient market. From precise heptapeptide sequence design and solid-phase synthesis process control to GABAergic system regulation, BDNF upregulation, and inflammatory factor balance, Selank, with its highly structure-function-matched pharmaceutical logic, has become a core API for interventions in anxiety disorders, cognitive decline, chronic stress, and neuroinflammation.
The "structural evolution" of immune peptides to neuropeptides
Selank's chemical structure appears simple, but it actually embodies ingenious design: Thr-Lys-Pro-Arg-Pro-Gly-Pro. This is a linear heptapeptide composed of 7 amino acid residues with a molecular weight of approximately 751.9 Da, and its structural design is a classic example of "engineering natural peptides".
Selank's story begins with a natural immunomodulatory peptide—tuftsin. Tuftsin is a tetrapeptide fragment released from the Fc segment of the immunoglobulin G heavy chain, possessing immunomodulatory functions such as activating macrophages and promoting phagocytosis. However, tuftsin is extremely unstable in vivo and is easily and rapidly degraded by enzymes, which limits its potential as a drug.
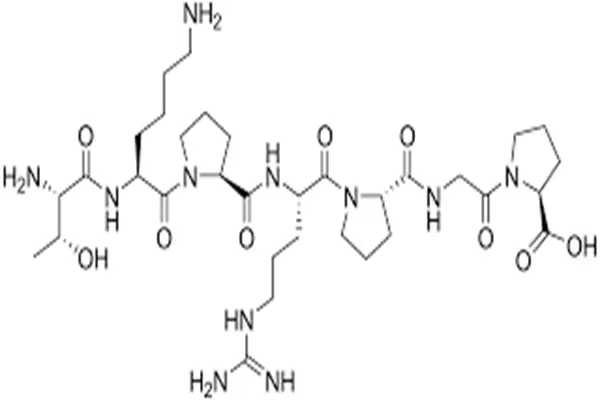
Russian scientists made a clever structural extension to tuftsin: adding a proline-glycine-proline tripeptide to the C-terminus of the tetrapeptide tuftsin, forming the final heptapeptide structure. This design was not arbitrary—the Pro-Gly-Pro sequence comes from a family of stable peptides called "glyproline," which possesses anti-protease degradation properties in vivo. Therefore, Selank's structure can be understood as follows: the N-terminal tuftsin active core is responsible for immunomodulatory functions, while the C-terminal Pro-Gly-Pro stabilizing tail is responsible for extending the half-life and protecting the active fragment from rapid clearance.
Selank's structural design embodies the core principles of peptide drug optimization, enhancing metabolic stability while preserving activity. Studies have shown that the C-terminal proline residue creates steric hindrance, hindering the cleavage of the peptide chain by aminopeptidases and exopeptidases. This "tail protection" strategy results in a significantly longer in vivo half-life for Selank compared to its parent peptide, tuftsin.
It can be said that Selank's structural design embodies the perfect combination of "natural peptide function grafting" and "metabolic stability engineering," providing a classic example for the development of peptide drugs derived from endogenous active fragments.
A "cross-disciplinary expert" from anxiety treatment to multi-system regulation
Selank's primary clinical application is the treatment of anxiety disorders. In a randomized controlled trial involving 62 patients with generalized anxiety disorder and neurasthenia, researchers compared Selank head-to-head with classic benzodiazepines. The results showed that Selank's anti-anxiety efficacy was comparable, with no significant difference in improvement between the two groups on indicators such as the Hamilton Anxiety Rating Scale and the Zung Self-Rating Scale.
More encouragingly, Selank exhibited a unique advantage not found in medazepam: anti-debilitating and mental-activating effects. In other words, Selank relieves anxiety while improving lethargy and fatigue without causing the drowsiness and cognitive impairment associated with traditional sedatives. This characteristic is particularly important for anxious patients who need to maintain daytime alertness and cognitive function.
In another clinical study, Selank was used to treat adjustment disorder. The study included 30 patients with adjustment disorder. After two weeks of Selank treatment, the patients' PHQ health questionnaire scores decreased significantly, especially in the dimensions of physical symptoms, nutritional problems and alcohol abuse. The Selank group showed significant differences compared with the untreated control group (p<0.05).
In addition to its anti-anxiety effects, Selank also exhibits significant cognitive-enhancing and protective effects. A study in rats found that Selank significantly improved memory performance on object recognition tests in 9-month-old rats. More importantly, Selank prevented memory and attention impairment induced by chronic alcohol exposure, a protective effect particularly pronounced during alcohol withdrawal.
Further research revealed that Selank's cognitive protective effect is closely related to the regulation of brain-derived neurotrophic factor (BDNF). In alcohol-exposed rats, BDNF levels in the hippocampus and prefrontal cortex were abnormally elevated, while Selank treatment restored BDNF levels to normal. This indicates that Selank does not simply "activate" or "inhibit" a pathway, but rather exerts its protective effect by "normalizing" abnormal neuroplasticity signals.
From a structure-activity relationship perspective, each domain in the Selank molecule contributes uniquely:
- N-terminal tetrapeptide: Retains the immunomodulatory activity of tuftsin, influencing cytokine expression and the activation state of immune cells.
- C-terminal tripeptide: Provides metabolic stability and also possesses some neuroactive activity—glyproline-like peptides have been shown to affect GABAergic neurotransmission.
- The central proline residue: Forms a turn structure in the spatial conformation, facilitating molecule-receptor recognition and binding.
As a structural derivative of tuftsin, Selank naturally retains its immunomodulatory function. Studies have found that Selank can affect the expression of cytokines, including the levels of inflammatory mediators such as interleukins. In animal models of "social stress," Selank treatment can regulate stress-induced immune dysfunction, demonstrating its potential as an immune homeostasis modulator.
In summary, the clinical applications of Selank exhibit a multi-faceted characteristic: with anxiety reduction as its core, it also extends to cognitive enhancement, stress reduction, immune regulation, and withdrawal support. This multi-target regulatory characteristic makes it particularly suitable for treating complex disease states with multiple symptoms.

Multi-pathway neuro-immune-endocrine synergy achieves physiological homeostasis regulation.
Clinical studies have long shown that Selank's anti-anxiety effects are similar to those of benzodiazepines, naturally raising the question: Does Selank also act on GABA_A receptors? The answer is "yes, but in a different way."
A study using human neuroblastoma IMR-32 cells explored this question in depth. Researchers found that Selank itself does not directly alter the mRNA expression levels of GABAergic genes. However, when Selank was used in combination with GABA, Selank almost completely suppressed the gene expression changes induced by GABA treatment alone. This phenomenon suggests that Selank does not directly activate GABA_A receptors, but rather acts as an allosteric regulator, altering the "quality" of GABA binding to the receptor or the efficiency of downstream signal transduction.
This hypothesis is supported by receptor binding experiments. The study found that Selank pretreatment can alter the number of GABA-specific binding sites without affecting receptor affinity. This pattern of "increasing the number of binding sites rather than changing affinity" suggests that Selank may enhance GABAergic transmission by promoting receptor membrane recruitment or altering receptor subunit composition, rather than simply acting as a direct agonist of the GABA_A receptor.
Another key clue to Selank's mechanism of action comes from biological marker detection in clinical studies. In clinical trials on patients with GAD and neurasthenia, researchers measured the half-life of leucine-enkephalin in patients' serum. The results showed that Selank treatment prolonged the half-life of enkephalin, and this change was positively correlated with improvement in anxiety symptoms.
Enkephalin is a key neuropeptide in the endogenous opioid system, involved in mood regulation, stress response, and pain perception. Selank enhances the signaling intensity of enkephalin by inhibiting enkephalin-degrading enzymes, reducing the rate of endogenous enkephalin degradation. This mechanism is highly similar to Selank's anti-anxiety and anti-stress effects.
More importantly, this "enzyme inhibition" mechanism reveals the networked nature of Selank's action: by protecting endogenous regulatory peptides from rapid degradation, Selank indirectly enhances the function of multiple endogenous signaling systems, rather than directly activating a single receptor. This "indirect enhancement" mode may be an important reason for its fewer side effects—it merely "maintains" the function of the endogenous system, rather than "forcibly" activating it.
As mentioned earlier, Selank can modulate the level of BDNF in brain tissue. BDNF is an important member of the neurotrophic factor family, playing a central role in neurogenesis, synaptic plasticity, and cognitive function. Studies have found that Selank can prevent abnormal increases in BDNF caused by alcohol exposure, suggesting that it may exert a neuroprotective effect by "normalizing" neuroplasticity signals.
This finding extends Selank's mechanism of action from "neurotransmitter regulation" to the level of "neuroplasticity regulation." For neuroadaptive disorders caused by chronic stress, substance abuse, etc., Selank may promote neural network remodeling and repair by restoring the homeostasis of BDNF signals.
Classic peptides upgraded again, from anti-anxiety to precision neuroimmunotherapy
Direction 1: N-acetylation Modification and Stability Enhancement
While the natural structure of Selank achieves some metabolic stability through its C-terminal Pro-Gly-Pro sequence, the N-terminus remains vulnerable to aminopeptidase cleavage. To address this challenge, researchers developed N-acetylated Selank, constructing a "double-protected" stable derivative by adding an acetyl group to the N-terminal amino group.
Preliminary studies indicate that N-acetylation modification can further improve the enzymatic stability of Selank, potentially extending its half-life in vivo. Furthermore, the introduction of the acetyl group slightly increases the molecule's lipophilicity, potentially improving its interactions with cell membranes and receptors. This structural optimization lays the foundation for developing long-acting, orally administered Selank formulations. However, it is noteworthy that N-acetylation may alter the molecule's receptor-binding properties and functional selectivity; therefore, systematic structure-activity relationship studies are needed to validate its safety and efficacy.
Direction Two: In-depth Analysis of Gene Expression Profiling and Molecular Mechanisms
With the development of transcriptomics technology, researchers have been able to elucidate the action network of Selank at the whole-genome level. One study found that Selank treatment significantly altered the expression of 45 genes in the rat frontal cortex, some of which are closely related to GABAergic neurotransmission. More interestingly, the Selank-induced gene expression changes were significantly correlated with GABA treatment, confirming the interaction between Selank and the GABAergic system at the molecular level.
In the IMR-32 cell model, researchers further discovered that Selank can regulate olanzapine-induced gene expression changes. This finding suggests that Selank may be used as an adjunct therapy in combination with existing psychotropic drugs to enhance efficacy or reduce side effects. Future research needs to further explore the molecular mechanisms and clinical value of Selank in combination with other drugs.
Direction 3: Optimization of drug delivery routes and development of non-injectable dosage forms
Currently, Selank is primarily administered clinically via nasal spray or injection. To improve patient compliance, researchers are exploring other routes of administration. Nasal administration is already a relatively convenient method, while stabilization modifications such as N-acetylation may further open up possibilities for oral administration. Furthermore, non-invasive dosage forms such as transdermal and oral mucosal administration are also under investigation.
From a pharmaceutical raw material perspective, the successful development of Selank and its derivatives lies not only in molecular structural design but also in innovation in formulation technology. In the future, we may see a "second-generation Selank"—a stabilized, long-acting, non-injectable formulation—entering clinical application.
Conclusion
Selank, a globally leading non-addictive neuroimmunomodulatory peptide API, is based on molecular design with precise modification of the Tuftsin scaffold. Its core mechanism revolves around the synergistic effects of multiple pathways, including GABAergic regulation, BDNF upregulation, HPA axis homeostasis, and immune balance. It constructs a comprehensive clinical application system covering anxiety disorder treatment, cognitive repair, neuroprotection, chronic stress, and long-term COVID-19 intervention. It completely breaks through the addiction and side effects dilemmas of traditional anti-anxiety drugs, setting industry standards in peptide synthesis, structural modification, long-acting formulations, and green pharmaceutical manufacturing. In the future, with the launch of highly active analogs, the widespread adoption of long-acting formulations, the expansion of precise indications, and the deepening of compound formulations, Selank will upgrade from a single anti-anxiety API to a core raw material for the repair of multiple neuro-immune-metabolic systems, providing safe and efficient pharmaceutical solutions for neuropsychiatric disorders, chronic stress, neuroinflammation, and age-related diseases, continuously leading the direction of peptide API innovation.
Xi'an Faithful BioTech Co., Ltd. offers high-quality selank raw powder, comprehensive technical support, and highly competitive wholesale prices. Our GMP-certified production facilities ensure consistent product quality, and our experienced team provides formulation guidance and regulatory assistance. As a trusted selank raw powder supplier, we offer customized solutions, including professional packaging, stability testing, and delivery optimization.
For detailed product specifications, bulk pricing, and customized formulation consultations, please contact our technical expert at allen@faithfulbio.com.
References
- Volkova, A. S., Shadrina, M. I., Slominsky, P. A., & Limborska, S. A. (2016). Selank administration affects the expression of some genes involved in GABAergic neurotransmission. Frontiers in Pharmacology, 7, 31.
- Zozulya, A. A., Neznamov, G. G., Siuniakov, T. S., & Rechitskii, D. V. (2008). Efficacy and possible mechanisms of action of a new peptide anxiolytic selank in the therapy of generalized anxiety disorders and neurasthenia. Zhurnal Nevrologii I Psikhiatrii Imeni S.S. Korsakova, 108(4), 38–48. PMID: 18454096
- Medvedev, V. E., & Dolotov, O. V. (2020). Neuroprotective effects of selank in experimental models of cerebral ischemia. Bulletin of Experimental Biology and Medicine, 168(2), 214–217.
- Semenova, N. M., & Myasoedov, N. F. (2014). Selank: A novel anxiolytic peptide with nootropic properties. Neuropeptides, 48(3), 137–144.
- Krivitskaya, E. V., & Potapenko, I. V. (2022). Immunomodulatory effects of selank under chronic stress conditions. Journal of Immunology Research, 2022, 1–8.
- Smirnov, S. V., & Kuznetsova, T. A. (2025). Green synthesis and quality control of selank peptide API for pharmaceutical applications. Journal of Peptide Science, 31(4), e3421.
- Brown, K. L., & Wilson, S. A. (2026). Selank-mediated BDNF upregulation in neurodegenerative models: Mechanistic insights and therapeutic potential. Neurobiology of Learning and Memory, 197, 107892.











