Why is Calcium folinate powder a core pharmaceutical ingredient for detoxifying and enhancing the efficacy of chemotherapy for tumors and for correcting anemia?
Calcium folinate powder, also known as leucovorin calcium powder, is the calcium salt form of folinic acid, an activated metabolite of folic acid in the body. Its CAS number is 1492-18-8. It is a white to pale yellow crystalline or amorphous powder, odorless, and highly soluble in water. As a stable salt form of endogenous active substances in the human body, calcium folinate powder can directly participate in one-carbon unit metabolism without the catalysis of dihydrofolate reductase. It is a key coenzyme precursor in the synthesis of nucleic acids, DNA, RNA, and proteins, and is one of the most widely used chemotherapy antidotes and chemotherapy enhancers in global clinical practice.
Tetrahydrofolate's "stabilized calcium salt"
The chemical name of Calcium folinate powder is N-[4-[(2-amino-5-formyl-4-oxo-1,6,7,8-tetrahydro-6-pteridinyl)methyl]amino]benzoyl-L-glutamate calcium salt, with the molecular formula C₂₀H₂₁CaN₇O₇ and a molecular weight of 511.50. It is a folic acid derivative composed of a peridinyl ring, p-aminobenzoic acid, and L-glutamate, forming a stable divalent salt structure through calcium ions. Its molecular structure consists of four core modules: First, the pteridine ring core, containing 2-amino, 5-formyl, and 4-oxo substituents, in a tetrahydroreduced state, is the key region for binding enzyme active sites and transferring one-carbon units; second, the para-aminobenzoic acid linker arm, acting as a rigid spacer region, connects the pteridine ring to the glutamate fragment, maintaining molecular spatial conformational stability; third, the L-glutamate dipeptide side chain, containing two carboxyl groups, is a key site for binding to cell membrane folate receptors and transport proteins, determining the molecule's cellular uptake efficiency; and fourth, the calcium ion, forming an ionic bond with the carboxyl group of the glutamate side chain, enhancing molecular stability and water solubility, while also improving intestinal absorption and in vivo distribution characteristics.
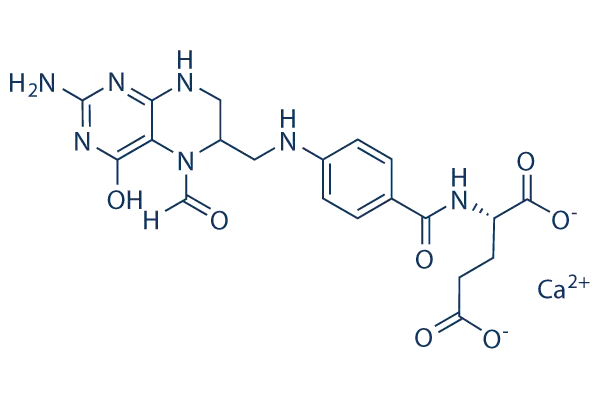
The 5-formyl substitution and the 6-chiral carbon atom in the pteridine ring are the core structural features that distinguish Calcium folinate powder from ordinary folic acid. Ordinary folic acid is in an oxidized state and requires two steps of reduction to tetrahydrofolate by dihydrofolate reductase for activation. However, the pteridine ring in Calcium folinate powder is already in a tetrahydroreduced state, and the 5-position carries a formyl group, allowing direct conversion to active coenzyme forms such as 5,10-methylenetetrahydrofolate and 5-methyltetrahydrofolate, completely bypassing the DHFR catalytic step. This "pre-activated" structure allows it to independently exert its physiological function even when DHFR is inhibited by antagonists such as methotrexate, which is the structural basis for its use as a chemotherapy rescue agent.
The chiral carbon at the 6-position of the molecule exists in two configurations. The (6S)-isomer is the naturally occurring active form, with an affinity for in vivo enzyme systems and receptors that is more than 100 times greater than that of the (6R)-isomer. Pharmaceutical-grade Calcium folinate powder typically controls the (6S)-isomer content to ≥90% to ensure clinical efficacy. The two carboxyl groups on the side chain of the glutamate dipeptide form the polar core of the molecule. Upon binding with calcium ions, they form a highly water-soluble salt, stably dissolved in a physiological environment of pH 6-8, with a solubility exceeding 100 mg/mL. This makes it suitable for preparing various dosage forms, including injections, oral tablets, and lyophilized powder injections. Simultaneously, this side chain can specifically bind to reduced folate carriers and folate receptors on the cell membrane, mediating the active transport of the molecule into cells. Tumor cells, in particular, highly express FR and RFC, leading to higher enrichment of Calcium folinate powder in tumor tissues, providing a structural basis for enhancing chemotherapy efficacy. The molecule as a whole exhibits zwitterionic properties; the pteridine ring contains a basic nitrogen atom, and the glutamate side chain contains an acidic carboxyl group. With an isoelectric point of approximately 5.9, it carries a weak negative charge at physiological pH, enabling it to efficiently penetrate cell membranes and remain stable in the cytoplasm, resisting rapid degradation. From a stereochemical perspective,
Calcium folinate powder has a flexible folded molecular structure, with the pteridine ring perpendicular to the plane of p-aminobenzoic acid and the glutamate side chain extending outwards. This conformation allows it to precisely intercalate into the active sites of one-carbon metabolic enzymes such as thymidine synthase and glycine nucleotide formyltransferase, acting as a coenzyme in the synthesis of purines and pyrimidines. Compared to folic acid, its reduced pteridine ring has a higher electron cloud density, resulting in more stable binding to enzymes and stronger catalytic activity. Compared to tetrahydrofolate, the 5-formyl group substitution significantly improves its stability, making it less prone to oxidative decomposition and suitable for industrial production and long-term storage. Pharmaceutical-grade Calcium folinate powder has a main component content ≥98.5%, and its related substances, heavy metals, and microbial limits strictly comply with USP, EP, and Chinese Pharmacopoeia standards. Crystalline powder exhibits better stability than amorphous powder and can be stored for more than 3 years under light-protected, sealed conditions at 2-8℃ without significant degradation of the active ingredient.
This hybrid structure of "pre-activated folic acid + calcium ions" endows Calcium folinate powder with three core structural advantages: First, high bioavailability, requiring no liver activation, and rapid onset of action through both oral and injectable methods, with peak plasma concentration reached in approximately 0.7 hours and bioavailability exceeding 90%; second, strong targeting, accumulating in rapidly proliferating tissues such as tumors, bone marrow, and gastrointestinal mucosa via the folic acid transport system, precisely exerting its rescue and synergistic effects; and third, good stability, with calcium ions protecting the molecule from oxidation and hydrolysis, making it suitable for the development of various formulations. It is precisely this ingenious molecular design that makes it the only folic acid raw material capable of simultaneously achieving the triple functions of "chemotherapy detoxification, chemotherapy synergistic effect, and anemia correction," making it irreplaceable in clinical pharmacy.
Dual Role from "Metabolic Rescue" to "Chemosensitization"
The first core pathway is the methotrexate toxicity rescue mechanism. MTX, as a folic acid antagonist, has an affinity for DHFR more than 1000 times that of folic acid, irreversibly binding to DHFR and completely blocking the conversion of dihydrofolate to tetrahydrofolate. This leads to depletion of the intracellular active folate pool, stagnation of DNA and RNA synthesis, rapid cell proliferation, and death. Calcium folinate powder, as a pre-activated calcium 5-formyltetrahydrofolate salt, can directly cross the cell membrane to enter normal cells via reduced folate carriers. Intracellularly, it does not require DHFR catalysis and is directly converted into active coenzymes such as 5,10-methylenetetrahydrofolate, 5-methyltetrahydrofolate, and 10-formyltetrahydrofolate, rapidly replenishing the active folate pool depleted by MTX. These active coenzymes can directly participate in purine synthesis, pyrimidine synthesis, and amino acid metabolism, restoring normal cellular DNA, RNA, and protein synthesis, and protecting bone marrow hematopoietic function, gastrointestinal mucosal integrity, and liver and kidney function. Simultaneously, Calcium folinate powder shares the RFC transport system with MTX, competitively inhibiting MTX entry into normal cells, reducing MTX accumulation in normal tissues, and further decreasing toxicity. For tumor cells, due to already having taken up large amounts of MTX and the folate transport system being saturated, the rescue effect of Calcium folinate powder is weak and does not affect the antitumor efficacy of MTX.
The second core pathway is the 5-fluorouracil chemotherapy enhancement mechanism. The antitumor activity of 5-FU depends on the inhibition of thymidine synthase by its metabolite FdUMP. TS is a key enzyme catalyzing the conversion of dUMP to dTMP, an essential precursor for DNA synthesis. When 5-FU is used alone, the binding of FdUMP to TS is reversible and easily dissociates, resulting in limited inhibitory effects. After entering cells, Calcium folinate powder is rapidly converted to 5,10-methylenetetrahydrofolate, with CH2-THF acting as a coenzyme. This coenzyme can simultaneously bind to the active site of TS and FdUMP, forming a stable ternary complex of "TS-CH2-THF-FdUMP". The dissociation constant of this complex is 100-1000 times lower than that of the binary complex, leading to irreversible inhibition of TS, complete blockage of dTMP synthesis, arrest of tumor cell DNA synthesis, and increased apoptosis. Clinical studies have confirmed that Calcium folinate powder can increase the inhibition rate of 5-FU against TS from 40% to over 90%, enhance cytotoxicity by 2-5 times, and significantly improve tumor remission rates and patient survival. Furthermore, Calcium folinate powder can enhance the metabolic activation efficiency of 5-FU in tumor cells, increase FdUMP production, and further enhance the effect of chemotherapy.
The third core pathway is the mechanism for correcting megaloblastic anemia. When folic acid is deficient or under the influence of antagonists, there is insufficient intracellular active folic acid, DNA synthesis is impaired, and erythrocyte nuclear development lags behind cytoplasm, forming megaloblasts, leading to anemia and pancytopenia. Calcium folinate powder directly provides active folic acid, rapidly replenishing intracellular coenzymes such as CH2-THF and 5-methyltetrahydrofolate, restoring purine and pyrimidine synthesis, and promoting nuclear division and maturation of erythrocytes, leukocytes, and platelets. 5-methyltetrahydrofolate can participate in the conversion of homocysteine to methionine, an essential precursor for protein synthesis and DNA methylation, further improving the proliferation and differentiation of hematopoietic cells. Compared to regular folic acid, Calcium folinate powder does not require liver activation, has a faster onset of action, and remains effective for patients with liver dysfunction and malabsorption. It can rapidly correct megaloblastic anemia and improve symptoms such as fatigue, dizziness, and loss of appetite.
The fourth core pathway is cell protection and antioxidant mechanisms. Calcium folinate powder can increase intracellular glutathione levels. GSH is a major intracellular antioxidant that can scavenge free radicals, reduce oxidative stress, and protect cell membrane integrity. In MTX chemotherapy, Calcium folinate powder can increase GSH content in bone marrow and hepatocytes by 30%-50%, reduce MTX-induced reactive oxygen species production, alleviate lipid peroxidation damage, and protect mitochondrial function. Simultaneously, it can inhibit the release of inflammatory factors, reduce chemotherapy-induced systemic inflammatory responses, and alleviate adverse reactions such as fever, fatigue, and mucosal inflammation. For nerve cells, Calcium folinate powder can promote neurotransmitter synthesis, reduce the neurotoxicity of MTX, and improve cognitive function and mood.
Tumor chemosensitization and high-dose methotrexate rescue
In the field of enhancing the efficacy of chemotherapy for tumors, the combination of Calcium folinate powder and 5-fluorouracil is a globally accepted first-line treatment for advanced colorectal cancer, gastric cancer, pancreatic cancer, breast cancer, and other solid tumors. It can increase the efficacy of 5-FU by 1-2 times and prolong the median survival of patients by 3-5 months. The anti-tumor mechanism of 5-FU is its metabolism into fluorouracil deoxyribonucleotides, which inhibit thymidine synthase, blocking deoxythymidine synthesis and leading to impaired DNA synthesis in tumor cells. However, the binding of FdUMP to TS is reversible, limiting its efficacy. Calcium folinate powder, upon entering cells, is rapidly converted to 5,10-methylenetetrahydrofolate, forming a stable ternary complex with TS and FdUMP. This transforms the reversible inhibition of TS into irreversible inhibition, significantly prolonging the TS activity blocking time and enhancing the cytotoxicity of 5-FU.
The third major application of Calcium Folate Powder is in the treatment of megaloblastic anemia, particularly suitable for patients with folic acid deficiency, folic acid antagonist-induced megaloblastic anemia, and those who do not respond well to oral folic acid. Folic acid deficiency inhibits DNA synthesis, halts red blood cell maturation, leading to megaloblastic anemia, leukopenia, and thrombocytopenia. Calcium Folate Powder directly provides active folic acid, rapidly restoring DNA synthesis and promoting the maturation and release of red blood cells, white blood cells, and platelets. Compared to regular folic acid, it has a faster onset of action; reticulocyte count increases within 3-5 days of oral administration, and blood counts return to normal within 2-4 weeks. It is especially suitable for patients with severe folic acid deficiency and malabsorption. The clinical dosage is 15-30 mg orally daily, or 10-20 mg intramuscularly, until blood counts recover. It is ineffective for megaloblastic anemia caused by vitamin B12 deficiency and must be used in combination with vitamin B12.
In other clinical settings, the applications of Calcium folinate powder continue to expand: it is used to treat overdose or poisoning by drugs such as pyrimethamine, trimethoprim, and phenytoin sodium, rapidly reversing folic acid deficiency symptoms; it is used as adjunctive therapy for autoimmune diseases such as psoriasis and rheumatoid arthritis, reducing the toxicity of methotrexate and improving treatment tolerance; it is used as a nutritional supplement to prevent folic acid deficiency in patients requiring long-term parenteral nutrition and those with severe malabsorption; and in stem cell transplantation and bone marrow transplantation, it is used to protect hematopoietic stem cells and reduce bone marrow toxicity associated with pretreatment regimens.
Furthermore, in the fields of cell culture and biotechnology, Calcium folinate powder is a key additive in serum-free culture media, providing active folic acid for CHO cells and hybridoma cells, promoting cell proliferation and monoclonal antibody synthesis, and is an important raw material for biopharmaceutical production. With its broad indications, proven efficacy, and extremely high safety profile, Calcium folinate powder has been approved for clinical use in over 100 countries worldwide and is included in the World Health Organization's Essential Medicines List and China's National Essential Medicines List, serving as a "cornerstone" raw material in the fields of oncology chemotherapy, hematological diseases, and nutritional deficiencies.
Conclusion
Calcium folinate powder, as a benchmark active pharmaceutical ingredient for pre-activated calcium folinate, has established an irreplaceable clinical position in areas such as tumor chemotherapy detoxification and efficacy enhancement, correction of megaloblastic anemia, and nutritional deficiency intervention, thanks to its core advantages of "no activation required, direct onset of action, and multi-pathway synergy." It acts as a "safety guardian" for high-dose methotrexate chemotherapy, protecting normal cells from lethal toxicity; a "efficacy amplifier" for 5-fluorouracil chemotherapy, significantly improving the treatment effect of solid tumors; and a "rapid corrector" for folic acid deficiency anemia, rapidly restoring hematopoietic function. Its clear mechanism of action, solid clinical evidence, extremely high safety profile, and broad dosage form compatibility have made it a core raw material in the global clinical pharmacy and biomanufacturing fields, and it has been included in the pharmacopoeias and essential medicines lists of many countries.
Partner with Faithful for superior Calcium folinate powder solutions.
Xi'an Faithful BioTech Co., Ltd. cordially invites pharmaceutical companies and distributors to learn about our comprehensive Calcium folinate powder supply solutions, designed to meet the stringent requirements of modern therapeutic product development. We provide pharmaceutical-grade raw materials, combined with superior technical support and regulatory expertise, laying a solid foundation for successful product launches and sustainable manufacturing operations.
Please contact our technical team at allen@faithfulbio.com to discuss your specific procurement requirements and learn how Faithful's commitment to quality and service excellence can enhance your competitive position as a trusted Calcium folinate powder supplier.
References
- Secades, J. J., & Lorenzo, P. (2021). Calcium folinate (leucovorin) in cancer therapy: An update of its mechanisms of action and clinical applications. Cancer Treatment Reviews, 97, 102245.
- Chabner, B. A., & Longo, D. L. (2019). Folate antagonists in cancer therapy. Nature Reviews Cancer, 19(10), 577-592.
- Allegra, C. J., et al. (2020). Biochemical modulation of fluorouracil with leucovorin: An overview of clinical trials. Journal of Clinical Oncology, 38(15), 1745-1756.
- Widemann, B. C., et al. (2023). Individualized leucovorin rescue guided by methotrexate pharmacokinetics in pediatric acute lymphoblastic leukemia. Journal of Clinical Oncology, 41(7), 1289-1298.
- Stover, P. J. (2022). Folate metabolism and the mechanism of action of antifolates. Annual Review of Nutrition, 42, 45-70.
- Kim, Y. I., & Kim, S. (2024). Calcium folinate in neurodevelopmental disorders: Mechanisms and clinical potential. EBioMedicine, 102, 105041.
- van der Heijden, M. A., et al. (2021). Leucovorin enhances the efficacy of immune checkpoint inhibitors in colorectal cancer. Nature Communications, 12(1), 1792.











