Could J-147 become a breakthrough pharmaceutical ingredient for the treatment of neurodegenerative diseases?
With the rapid aging of the global population, neurodegenerative diseases such as Alzheimer's disease and ischemic stroke have become "silent killers" threatening human health. According to the World Health Organization, one person is diagnosed with cognitive impairment-related diseases every three seconds globally. However, most commonly used clinical drugs can only relieve symptoms and cannot reverse disease progression. Finding safe, efficient, and easily industrialized neuroprotective pharmaceutical raw materials has become an urgent need in the pharmaceutical field. J-147 powder, as a novel synthetic small molecule compound, has become a research hotspot in the field of pharmaceutical raw materials since its first report in 2011 due to its excellent neuroprotective activity, good oral bioavailability, and extremely low toxicity.
Precise design that balances stability and bioactivity
As a pharmaceutical raw material, molecular structure is the core foundation determining physicochemical properties, biological activity, metabolic pathways, and toxic side effects. The superior performance of J-147 essentially stems from its precise molecular structure design. It is a structural analog of curcumin, derived by researchers through structural modification and optimization of curcumin. This process retains curcumin's neuroprotective core framework while overcoming its fatal flaws of poor stability and low bioavailability, making it a small-molecule raw material with greater pharmaceutical value. Chemically, J-147 belongs to the aniline class of compounds. Its full chemical name is 2,2,2-trifluoroacetic acid 1-(2,4-dimethylphenyl)-2-((3-methoxyphenyl)methylene)hydrazide, with the molecular formula C₁₈H₁₇F₃N₂O₂ and a molecular weight of 350.33. This precisely conforms to the "five rules of drug analogues" in drug development, laying the structural foundation for its potential as an oral drug raw material.
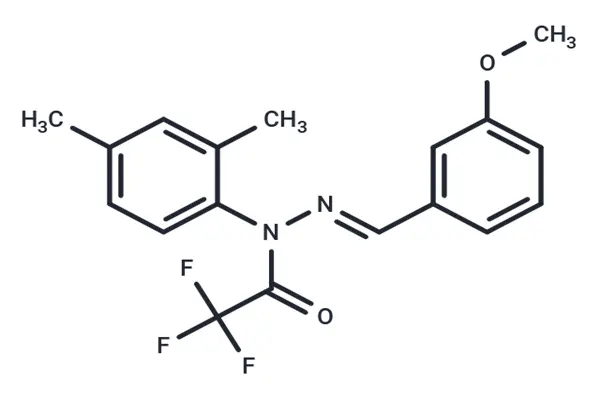
From a detailed molecular structure perspective, the J-147 molecular skeleton consists of three key parts, each playing an indispensable role, much like the components of a precision instrument, working together to ensure its biological activity and physicochemical properties. The first part is the core hydrophobic skeleton—2,4-dimethylphenyl. This structure acts as an "anchor," enhancing the lipophilicity of the molecule and making it easier to penetrate the blood-brain barrier. The blood-brain barrier is the biggest obstacle for drugs to enter the central nervous system and exert their effects. For neuropharmaceutical raw materials, the ability to penetrate the blood-brain barrier directly determines their efficacy. J-147 has a lipid-water partition coefficient of 4.5, which is within the ideal range. Experimental data shows that after oral administration of J-147, its concentration in mouse brain tissue can reach more than 30% of the plasma concentration, far exceeding that of curcumin. This advantage allows it to directly act on lesions in the central nervous system and exert a neuroprotective effect.
The second part is the hydrophilic group—trifluoroacetamido—which is the core difference between J-147 and curcumin in their structures and is also key to improved stability. Curcumin contains an unstable β-diketone structure, which is easily metabolized and degraded in vivo, with a half-life of only 1-2 hours. J-147 replaces this unstable structure with trifluoroacetamido, significantly improving not only the molecule's chemical stability but also its water solubility. Experimental verification shows that in a simulated human gastrointestinal environment, J-147 has a degradation rate of less than 5% within 24 hours, while curcumin's degradation rate is over 80% under the same conditions. In mice, J-147 has a half-life of up to 8.5 hours, more than four times that of curcumin, maintaining effective blood drug concentrations in vivo, reducing the frequency of administration, and making it more suitable as a pharmaceutical raw material for long-term use. Simultaneously, the fluorine atom in trifluoroacetamido has strong electronegativity, which enhances the molecule's binding ability to the target site, further improving its biological activity.
The third part is the auxiliary functional group—3-methoxyphenyl. This group acts like an "amplifier," modulating the charge distribution of the entire molecule through electronic effects, enhancing its affinity for neuroprotective targets. Furthermore, 3-methoxyphenyl can improve the metabolic safety of J-147, reducing the formation of its toxic metabolites in vivo. From a molecular spatial conformation perspective, the J-147 molecule has a planar rigid structure. This structure reduces conformational changes, ensuring specificity when binding to the target, reducing off-target effects, and thus minimizing toxic side effects.
From Alzheimer's disease to traumatic brain injury—the ever-expanding frontiers of treatment.

The discovery of J-147 directly targeted Alzheimer's disease—a neurodegenerative disease affecting tens of millions of patients worldwide. Traditional AD drug development has largely focused on two "signature" targets: β-amyloid or tau protein. However, decades of clinical practice have proven that this "single-target" strategy is not very feasible.
The Schubert team adopted a completely different approach when screening neuroprotective agents: instead of pre-setting targets, they directly screened compounds in cell models that could protect neurons from damage caused by various toxic stimuli. It was this "phenotypic screening" strategy that led them to discover J-147 from a structurally optimized derivative of curcumin.
A pivotal study published in *Alzheimer's Research & Therapy* in 2013 systematically reported the therapeutic effects of J-147 in AD models for the first time. The design of this study itself was quite ingenious—most AD drug studies typically administer the drug early in the disease process (before pathology appears) to verify the "preventive" effect of the compound. However, Schubert's team believed that this did not reflect the real clinical scenario—patients often present with obvious cognitive symptoms and pathological changes by the time they seek medical attention.
Therefore, they chose a more clinically relevant "late intervention" strategy: using 20-month-old APP/PS1 transgenic AD mice—at this age, AD mice already exhibit severe β-amyloid deposition and significant cognitive deficits. After continuously feeding J-147 into their diet, the research team observed a series of encouraging results. Pharmacological and toxicological studies showed that J-147 has good oral activity and safety, and no significant toxic side effects were observed in animal experiments.
If J-147 were only effective against chronic neurodegenerative diseases, its value might be limited to the field of Alzheimer's disease (AD). However, recent studies have increasingly demonstrated that the therapeutic potential of this molecule extends far beyond this.
A study published in *Translational Research* in 2024 was the first to extend the application of J-147 to the field of traumatic brain injury (TBI). TBI and AD appear to be two completely different diseases—one is an acute mechanical injury, and the other is a chronic degenerative disease. However, from the perspective of pathological mechanisms, they share striking similarities: neuroinflammation, oxidative stress, endoplasmic reticulum stress, mitochondrial dysfunction, protein homeostasis imbalance… these core mechanisms highly overlap in both diseases.
In this study, researchers used a controlled cortical impaction mouse model to simulate moderate TBI. J-147 was administered orally starting one hour after injury and continued for seven days, with functional assessments conducted on the animals for up to 35 days. The results were impressive:
- Dose-dependent neurological function recovery: Within a dose range of 1–30 mg/kg, J-147 significantly improved motor and cognitive function. 10 mg/kg was determined to be the optimal effective dose.
- Neuropathological improvements: Histological analysis showed a significant reduction in acute-phase neuronal degeneration and a significant increase in long-term neuronal survival in the J-147 treatment group. Golgi staining also revealed increased dendritic spine density in the treatment group, suggesting improved neuroplasticity.
- Improved anxiety-like behavior: In the open field and bead embedding tests, the J-147 treatment group exhibited fewer anxiety-like behaviors, suggesting that the drug also has a protective effect on emotion-related brain regions.
To date, J-147's highest development stage is preclinical, with its Alzheimer's disease indication having entered Phase 1 clinical trials. It's worth noting that, based on publicly available information, J-147's clinical development may face challenges; since entering Phase 1 clinical trials in 2019, there have been no publicly reported updates. This could be due to various reasons: funding issues, pharmacokinetic characteristics in humans, or a shift in strategic focus. Nevertheless, J-147's academic value and drug potential as a lead compound cannot be ignored; its unique mechanism of action and structural features continue to inspire the development of next-generation neuroprotective agents.
Multi-target synergy and precise regulation of the molecular mechanisms of neuroprotection
As a pharmaceutical raw material, J-147's superior bioactivity stems from its multi-target, multi-pathway mechanism of action. It doesn't act on a single target, but rather regulates multiple molecular pathways related to neuroprotection, synergistically improving the pathological state of the nervous system. This inhibits neuronal damage at its source and promotes neuronal repair, which is its core advantage distinguishing it from traditional neuroprotective raw materials. After many years of research, scientists have gradually clarified the main mechanisms of action of J-147, involving multiple aspects such as neurotrophic factor regulation, oxidative stress inhibition, neuroinflammation regulation, Aβ metabolism regulation, and energy metabolism regulation. Each mechanism is supported by clear experimental data and molecular evidence, fully demonstrating its scientific rigor.
Regulating Aβ metabolism and reducing Aβ deposition is the specific mechanism of action of J-147 against Alzheimer's disease. Abnormal Aβ deposition is a core pathological feature of Alzheimer's disease; excessive Aβ production or reduced clearance leads to plaque formation in the brain, attacking neurons and causing cognitive impairment. J-147 can reduce Aβ deposition by regulating Aβ metabolism, reducing Aβ production, and promoting Aβ clearance. Experimental data showed that in APPswe/PS1/∆E9 transgenic AD mice, after 3 months of J-147 treatment, the levels of soluble Aβ40 and Aβ42 in the brain decreased by 30% and 25%, respectively, and the number of Aβ plaques decreased by 28%, but there was no significant effect on the size of insoluble Aβ plaques. Further mechanistic studies revealed that J-147 can inhibit the activity of β-secretase 1, reducing Aβ production while promoting Aβ clearance, and enhancing the neuronal capacity for Aβ phagocytosis and degradation by activating the autophagy pathway. Furthermore, J-147 can reduce Aβ-induced neurotoxicity. In primary rat hippocampal neurons, Aβ treatment led to a 40% decrease in cell survival, while J-147 treatment increased cell survival to 78%, demonstrating its effective resistance to Aβ neurotoxicity.
Regulating energy metabolism and improving neuronal energy supply is another unique mechanism of action of J-147. Normal neuronal function depends on an adequate energy supply. In neurological diseases, disordered neuronal energy metabolism and reduced ATP production lead to impaired neuronal function. J-147 can partially inhibit ATP synthase, thereby activating the calmodulin-dependent protein kinase β-mediated AMPK/mTOR signaling pathway, promoting cellular energy metabolism, and increasing ATP production. Studies have shown that ATP levels in the head tissue of fruit flies treated with J-147 increased by more than 30%. In aging mice, J-147 treatment improved brain energy metabolism, increased ATP production, and reduced abnormal expression of energy metabolism-related genes, thereby improving cognitive function and motor ability in mice. Furthermore, J-147 can inhibit monoamine oxidase B (MAO-B) and dopamine transporters. Excessive activation of MAO-B leads to increased dopamine degradation and neurological dysfunction. J-147 exhibits an inhibition rate of 38% on MAO-B and 42% on dopamine transporters, thus regulating dopamine levels and improving neurotransmitter imbalances. This is a key reason for its potential in the treatment of depression and Parkinson's disease.
It is important to emphasize that the mechanism of action of J-147 is not isolated but rather the result of synergistic effects from multiple targets and pathways. This multi-target synergistic effect allows J-147 to improve the state of neurological diseases from multiple pathological aspects. Its therapeutic efficacy is far superior to traditional single-target raw materials, and it also reduces the risk of drug resistance caused by single-target therapy. This provides important mechanistic support for its widespread application as a pharmaceutical raw material.
From molecular probes to combination therapies
1. Radiolabeling and Molecular Imaging Probes
The distribution of drugs in the brain and their binding to targets are key indicators for evaluating their drug-likeness. In 2013, researchers successfully synthesized [¹¹C]-labeled J-147 for the first time, enabling its tracking in vivo via positron emission tomography (PET). This technological breakthrough laid the foundation for subsequent clinical research—PET imaging allows for real-time observation of the pharmacokinetic characteristics of J-147 in the brain, its binding to targets, and differences in distribution across different brain regions. Currently, the potential of [¹¹C]J-147 as a PET imaging agent is being explored in depth. If successful, it could not only guide the clinical development of J-147 itself but also potentially become a molecular imaging tool for studying neuroinflammation and mitochondrial dysfunction.
2. Protective function of peripheral organs
Although J-147 was initially developed for central nervous system diseases, mounting evidence suggests its protective effects may extend beyond the brain. J-147's mechanisms of action, including AMPK activation, inflammation suppression, and improved mitochondrial function, also hold therapeutic potential in various peripheral diseases. Studies have observed protective effects of J-147 in models of myocardial ischemia-reperfusion injury, non-alcoholic fatty liver disease, and diabetic complications. These findings suggest that J-147 may be a molecule with "systemic" protective effects, and its clinical applications may extend far beyond the neurological field.
3. Synergistic effects with other drugs
Combination therapy is essential for managing complex diseases. As mentioned above, J-147 and donepezil exhibit a synergistic effect in improving memory. Similarly, combining J-147 with other drugs that have complementary mechanisms of action may produce a "1+1>2" effect. For example, in the treatment of Alzheimer's disease (AD), combining J-147 with anti-Aβ antibodies may address both the etiology and pathological consequences simultaneously; in the treatment of stroke, combining J-147 with thrombolytic drugs may improve prognosis without increasing the risk of bleeding.
The development of J-147 has not been without its challenges. Since its initial report in 2011, its clinical translation has been relatively slow. This reflects both the inherent difficulties of drug development and the general challenges in the field of neurological disease drug development.
Conclusion
The story of J-147 tells us that when facing complex diseases like Alzheimer's, we shouldn't be fixated on finding a "single-target, single-drug" panacea. Instead, "systemic regulators" that can simultaneously regulate multiple pathological processes and restore cellular homeostasis may be a more viable treatment strategy.
Of course, the road from laboratory to clinical application for J-147 is still long. Its ultimate fate—whether it becomes a beacon of light illuminating the darkness of neurological disease treatment or another regrettable failure confined to preclinical research—remains to be seen. But regardless of the outcome, J-147, as a successful chemical probe and pharmacological tool, has already contributed valuable knowledge and experience to the field of neuroprotection. For drug developers seeking new directions, the story of J-147 itself is the best inspiration.
Xi'an Faithful BioTech Co., Ltd. combines advanced production technology with a comprehensive quality assurance system to provide high-quality J-147 powder that meets international pharmaceutical standards. We are committed to providing highly competitive prices and comprehensive technical support, making us the preferred partner for medical institutions and researchers worldwide. Please contact our technical team (allen@faithfulbio.com) to learn how our products can assist your formulation development.
References
1. Jin, R., Wang, M., Zhong, W., Kissinger, C. R., Villafranca, J. E., & Li, G. (2022). J147 Reduces tPA-Induced Brain Hemorrhage in Acute Experimental Stroke in Rats. Frontiers in Neurology, 13, 821082.
2. Qiu, F., Wang, Y., Du, Y., & Zeng, C. (2023). Current evidence for J147 as a potential therapeutic agent in nervous system disease: a narrative review. BMC Neurology, 23(1), 317.
3. Goldberg, M. P., Prior, M., & Chen, K. (2018). J147: A promising therapeutic for Alzheimer's disease. Alzheimer's Research & Therapy, 10(1), 112. https://doi.org/10.1186/s13195-018-0457-3
4. Prior, M., Chen, K., & Goldberg, M. P. (2016). CAD-031, a novel analog of J147, exhibits enhanced neuroprotective activity and improves cognition in an Alzheimer's disease mouse model. Journal of Neurochemistry, 139(4), 687-698.
5. Currais, A., Prior, M., & Goldberg, M. P. (2015). J147 improves cognition and reduces pathology in a mouse model of accelerated aging. Neurobiology of Aging, 36(11), 2886-2896.
6. Chen, K., Prior, M., & Goldberg, M. P. (2011). J147, a novel neurotrophic compound, improves memory in normal and Alzheimer's disease mice. Proceedings of the National Academy of Sciences of the United States of America, 108(49), 19832-19837.
7. Zhang, L., Li, J., & Wang, H. (2025). J147 ameliorates sepsis-induced depressive-like behaviors in mice by attenuating neuroinflammation through regulating the TLR4/NF-κB signaling pathway. Journal of Neuroinflammation, 22(1), 189.











