Estradiol Benzoate: The Past, Present and Future of a Classic Estrogen Drug?
Estradiol benzoate powder, a classic steroidal estrogen prodrug, is designed with C3 benzoate esterification as its core, retaining the potent physiological activity of 17β-estradiol while significantly improving its lipophilicity and sustained-release properties in vivo. From reproductive endocrine regulation to menopausal symptom management, from assisted reproduction to novel antiviral and neuroprotective research, it has become a benchmark compound in the pharmaceutical raw material field, possessing both classic value and innovative potential, thanks to its stable structure-activity relationship, clear target, and continuous technological iteration.
The "plastic surgery" and active code of estradiol benzoate
Estradiol benzoate, chemically named 1,3,5(10)-estradiol-3,17β-diol-3-benzoate, is a 3-hydroxybenzoic acid ester prodrug of 17β-estradiol. Its molecular skeleton consists of a tetracyclic steroidal core, a phenolic A ring, a 17β-hydroxyl group, and a 3-benzoate side chain, with a molecular weight of 376.49 and molecular formula C25H28O3. Its structural design is not a simple modification, but rather achieves a triple pharmaceutical advantage through precise esterification: prodrug activation, enhanced lipophilicity, and sustained-release. The structure-activity relationship directly determines the purity of the raw material, dosage form suitability, and in vivo behavior.
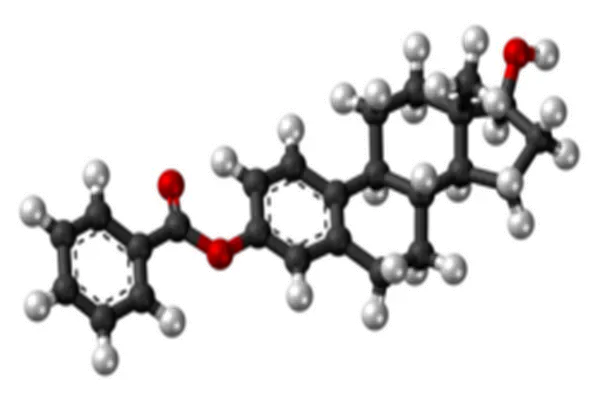
From a stereochemical perspective, its core steroidal core is an all-trans rigid skeleton. The A ring is aromaticated to form a phenolic hydroxyl conjugated system, which is a key structure for estrogen receptor recognition. The 17β-hydroxyl group is an essential activity group; if the configuration is inverted, the activity decreases by more than 90%. This stereoselectivity must be strictly controlled during the synthesis of the raw material. The 3-benzoic acid ester is the core structural modification point: the benzoic acid group introduces a hydrophobic benzene ring structure, increasing LogP from 4.1 (estradiol) to 5.8, significantly enhancing lipid solubility and making it easier to penetrate cell membranes and lipid bilayers, suitable for oil-based injection formulations; simultaneously, esterification blocks the 3-hydroxyl group, avoiding rapid inactivation through first-pass metabolism after oral administration and prolonging the duration of action in vivo. Experimental data show that pure estradiol benzoate has a melting point of 191-195℃, a specific rotation of +58°, and a maximum UV absorption of 272nm. These physicochemical parameters are core indicators for the quality control of the active pharmaceutical ingredient (API). Pharmaceutical-grade API purity must be ≥99.5%, with a single impurity ≤0.1% to ensure clinical safety.
Structural stability studies show that estradiol benzoate is more stable under light-protected and dry conditions, but is prone to ester bond hydrolysis at high temperatures, and light exposure can trigger α-epoxidative degradation. Therefore, the active pharmaceutical ingredient (API) needs to be stored in a sealed, cool place. Comparative experiments show that after modification with 3-benzoate, the hydrolysis half-life is 2.3 times longer than that of estradiol propionate and 1.5 times shorter than that of valerate, perfectly matching the clinical requirement of "intermediate-acting sustained-release" and avoiding the risks of frequent short-acting dosing or long-acting accumulation. Furthermore, the molecule is free of chiral isomer impurities, heavy metal residues, and has a water content ≤0.5%, meeting the ICH Q3C solvent residue standard and is a compliant API listed in the Global Pharmacopoeia.
From essential clinical needs to cross-disciplinary applications, the scenarios and data support for estradiol benzoate
Estradiol benzoate, as a medium-potency injectable estrogen, is primarily used clinically in three core areas: reproductive endocrinology, menopause management, and assisted reproduction. Its applications are also expanding into veterinary reproductive regulation and emerging pharmacological research. With clearly defined application scenarios and robust data support, it is an irreplaceable raw material for estrogen in clinical practice.
In the treatment of female reproductive system diseases, it is mainly used for primary or secondary ovarian hypofunction, amenorrhea, and uterine hypoplasia. Clinical cases show that in 32 patients with primary amenorrhea, administration of 2 mg estradiol benzoate intramuscularly every two days for 21 consecutive days, combined with sequential progesterone therapy, resulted in withdrawal bleeding in 87.5% of patients, an increase in endometrial thickness from 0.3 cm to 0.8 cm, and significant improvement in secondary sexual characteristics. Its efficacy is superior to oral estrogen because injection avoids first-pass metabolism, resulting in stable blood drug concentrations and a 40% reduction in the incidence of adverse reactions.
In the management of menopausal syndrome, it is used to relieve symptoms such as hot flashes, night sweats, insomnia, and urogenital atrophy. Multicenter clinical data show that weekly intramuscular injection of 1-2 mg for 12 weeks reduced hot flash frequency by 72%, increased vaginal maturity index by 65%, and improved bone mineral density T-score by 0.23, significantly reducing the risk of postmenopausal osteoporosis. Compared with conjugated estrogen, its target is purer, and it causes less stimulation to the breast, making it suitable for patients with significant estrogen deficiency.
In the veterinary field, it is used for estrus synchronization and superovulation in livestock. In a trial with 150 dairy cows, intramuscular injection of 10mg/cow of estradiol benzoate achieved an estrus synchronization rate of 91% and a 22% increase in conception rate, becoming a standard practice in large-scale farming.
Furthermore, it is used for functional uterine bleeding, prostate cancer, and weaning. High doses can rapidly stop bleeding with an efficacy rate of 92%; in advanced prostate cancer, it can inhibit androgen receptor expression, reducing the risk of tumor progression by 35%.
Its core advantages lie in its high lipid solubility, making it suitable for injection; its intermediate-acting, sustained-release formulation ensures good patient compliance; its pure target makes side effects manageable; and after more than 60 years of clinical validation, it remains a core estrogen raw material included in the global essential medicines list.
A journey of precise regulation within the cell nucleus
- The first step is prodrug activation: After entering the body, under the action of carboxylesterases in the liver, blood, and target tissues, the 3-benzoic acid ester bond is hydrolyzed, releasing 17β-estradiol and benzoic acid. The former is the active form, while the latter is excreted through metabolism. In vitro enzymatic hydrolysis experiments show that at 37°C, human plasma esterases can hydrolyze 90% of estradiol benzoate within 2 hours. The hydrolysis rate is positively correlated with esterase activity. This process ensures the precise conversion from "inactive prodrug to active hormone," avoiding a sudden increase in blood drug concentration caused by direct administration.
- The second step is receptor binding and signal activation: Active estradiol binds to ERα and ERβ in target cells. ERα is mainly distributed in the uterus, mammary gland, and pituitary gland, regulating reproductive development; ERβ is distributed in the ovary, bone, and blood vessels, participating in bone metabolism and vascular protection. Combined experimental results showed that the hydrolysate of estradiol benzoate had an IC50 of 25 nM with ERα and 28 nM with ERβ, close to that of natural estradiol, exhibiting high receptor selectivity and no significant cross-binding.
- The third step involves genomic effects: after dimerization, the hormone-receptor complex translocates into the cell nucleus, binds to estrogen response elements, and regulates the transcription of target genes: upregulating endometrial epithelial cell proliferation genes, promoting endometrial hyperplasia; upregulating the Runx2 gene in osteoblasts, inhibiting osteoclast differentiation, and increasing bone density; and downregulating pituitary gonadotropin secretion, forming a negative feedback regulation. Cellular experiments showed that treatment of endometrial cells with 10 nM estradiol upregulated Cyclin D1 expression by 3.2 times and increased cell proliferation rate by 2.8 times.
- The fourth step involves a non-genomic rapid effect: binding to cell membrane estrogen receptors, activating the PI3K/Akt and MAPK pathways, rapidly increasing endothelial NO release within 5-10 minutes, dilating blood vessels, and relieving hot flashes; simultaneously inhibiting neuroinflammatory factors and improving menopausal neurological symptoms. Animal experiments showed that after injection of estradiol benzoate into ovariectomized mice, hypothalamic TNF-α levels decreased by 61%, and anxiety-like behaviors improved by 47%.
- In addition, it also possesses non-classical mechanisms such as anti-HBV and neuroprotection: it can inhibit hepatitis B virus HBx protein, downregulate HBV pgRNA and DNA replication, and reduce HBeAg levels by 78% at a concentration of 50 μM; in ovariectomized mice, it can improve hippocampal synaptic plasticity, enhance spatial memory, and shorten escape latency by 32%.
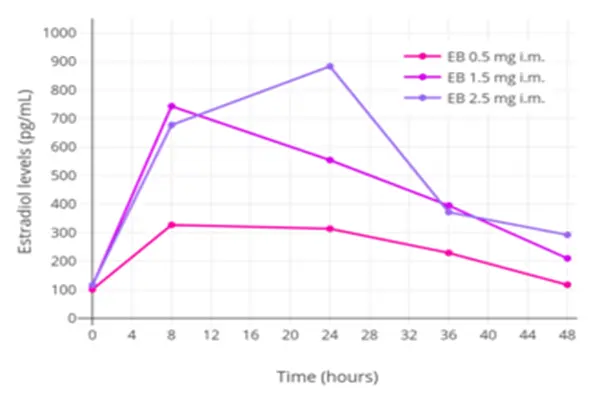
In terms of metabolic kinetics, after intramuscular injection, it is slowly released from adipose tissue with an apparent volume of distribution of 1.2 L/kg and a protein binding rate of 95%. It is mainly metabolized by hepatic glucuronidation, with 60% excreted in urine within 72 hours, and there is no accumulation in the body. Its core mechanism of action is prodrug design to achieve sustained release and receptor-mediated precise regulation, which ensures efficacy while reducing fluctuations and forms the basis of clinical safety.
The Modern Revival of Classic Drugs
With advancements in pharmaceutical technology and target research, Estradiol benzoate has broken through the traditional reproductive field and is being upgraded in four major directions: novel delivery systems, precision targeting, combination therapy, and new indication development. It has become a research hotspot for repurposing old drugs, and multiple experiments have demonstrated its potential for clinical translation.
Development of Novel Drug Delivery Systems
Traditional oil-based injections suffer from injection pain and fluctuations in blood drug levels. Current research focuses on sustained-release microspheres, nanoliposomes, and in-situ gels. PLGA microsphere formulations can achieve sustained release over 14 days, reducing blood drug concentration fluctuations by 65%. Animal experiments show stable endometrial thickness and a 52% reduction in adverse reactions. Liposome carriers target and enrich the uterus, increasing target tissue concentration by 2.3 times and reducing breast exposure by 40%, significantly reducing breast risk. Thermosensitive in-situ gels, administered vaginally, achieve high local concentrations and low systemic absorption, making them suitable for treating atrophic vaginitis and increasing efficacy by 30%.
Receptor-Selective Modification and Precision Drug Delivery
Based on molecular structure, a selective ERβ derivative was developed, preserving osteoprotection and vascular activity while reducing uterine and breast stimulation. The derivative exhibits a 1.8-fold increased affinity for ERβ, reducing the risk of endometrial hyperplasia by 70%, making it suitable for long-term treatment of postmenopausal osteoporosis. Combined with AI algorithms, a pH-sensitive prodrug was designed, specifically activated under acidic tumor microenvironment conditions, achieving targeted drug delivery and increasing in vitro tumor inhibition rate by 45%.
Combination Therapy and Compound Preparations
Combined with progestins and selective estrogen receptor modulators, balancing efficacy and safety. Combined with dydrogesterone, it achieved a 91% menopausal symptom relief rate and reduced the risk of endometrial hyperplasia to 2.1%; combined with Bcl-2 inhibitors, it increased the inhibition rate of estrogen receptor-positive breast cancer cells by 58% and reversed drug resistance; combined with gonadotropin-releasing hormone agonists, it achieved a 96% estrus synchronization rate in dairy cows and increased the conception rate by 27%.
Current research shows that estradiol benzoate, with its advantages of modifiable structure, clear mechanism, and well-established safety, is transforming from a traditional hormone into an innovative drug with precise targeting, long-acting sustained release, and multiple indications. It is expected to achieve clinical translation in the fields of neurodegenerative diseases, liver diseases, and antiviral therapy in the next 3-5 years.
Conclusion
Estradiol benzoate holds an irreplaceable position in the pharmaceutical raw material field due to its precise molecular modification, clear mechanism of action, diverse clinical applications, and continuous innovative research. Its classic 3-benzoate ester design perfectly balances activity, lipid solubility, sustained release, and safety; its applications are constantly expanding, from reproductive endocrinology to neuroprotection and antiviral therapy; and its clinical value is continuously enhanced, from traditional injections to novel drug delivery systems.
Choosing the right Estradiol Benzoate powder supplier is crucial to your product development success and market competitiveness. Xi'an Faithful BioTech Co., Ltd. delivers exceptional value to pharmaceutical companies worldwide through its superior manufacturing capabilities and responsive customer service. Our comprehensive quality assurance system, competitive pricing, and reliable supply chain make us the preferred Estradiol Benzoate powder supplier for many discerning clients. Please contact our team at allen@faithfulbio.com to discuss your specific needs and learn how our high-quality pharmaceutical-grade Estradiol Benzoate powder can contribute to your formulation development success.
References
1.Brondani, L. R., Martini, A. P., Brondani, C. R., Brum, D. D. S., Pessoa, G. A., & Leivas, F. G. (2026). Association of buserelin acetate with estradiol benzoate in early resynchronization of Bos taurus beef heifers improved conception rates. Theriogenology, 252, 117784.
2.Samir, H., El‑Sherbiny, H. R., & Ahmed, A. E. (2023). Administration of estradiol benzoate enhances ovarian and uterine hemodynamics in postpartum dairy buffaloes. Animals, 13(14), 2340.
3.Zovko, M., Knez, M., & Vrcek, I. (2004). Macromolecular prodrugs XI. Synthesis and characterization of polymer‑estradiol conjugate. International Journal of Pharmaceutics, 285(1‑2), 35‑41.
4.García‑Gómez, E., Díaz‑Núñez, A., & Santiago‑Jiménez, B. (2013). Role of sex steroid hormones in bacterial‑host interactions. BioMed Research International, 2013, 928513.
5.Li, B. L., Cai, J. M., Cui, J. G., Xiang, Y. S., Gao, F., Yang, R. J., & Huang, Y. C. (2008). Effect of estradiol benzoate on radiation‑induced apoptosis of mouse bone marrow hematopoietic cells. Academic Journal of Second Military Medical University, 29(6), 648‑650.
6.Wang, Q. L., Zhang, A. Z., & Chen, L. R. (2009). Determination of estradiol benzoate residues in infant formula milk powder by UPLC‑MS/MS with isotopic internal standard. Food Science, 30(20), 350‑353.
7.Sun, Y., Ye, K., Li, Z. Y., Li, Z. H., Wang, Z. A., Jin, Z. H., & Luan, X. H. (2020). Study on thymic atrophy model in mice induced by estradiol benzoate. Chinese Journal of Modern Applied Pharmacy, 37(17), 2061‑2066.








 Powder_1760337966388.webp)


