Megestrol acetate: How does steroid modification endow it with multi-field medicinal value?
Megestrol acetate, a synthetic progestin, has evolved from its initial role as a contraceptive to a core active pharmaceutical ingredient (API) for palliative treatment of hormone-dependent tumors and intervention for cancer cachexia since its discovery in 1959 and its clinical application in 1963. From precise modification of the steroid nucleus to multi-receptor cross-regulation, from classic anti-tumor applications to novel formulations and metabolic target discovery, it has become a benchmark molecule in the pharmaceutical API field, combining classic and innovative properties, thanks to its stable chemical structure, clear pharmacological effects, and continuous clinical value.
Precise modification of the steroid skeleton lays the foundation for efficacy and drug-like properties.
Megestrol acetate is chemically known as 17α-acetoxy-6-methyl-6-dehydroprogesterone, CAS number 595-33-5, with the molecular formula C₂₄H₃₂O₄ and a relative molecular mass of 384.51. It belongs to the 19-nortestosterone class of steroidal derivatives, with a core skeleton of a cyclopentane- and polyhydrophenanthrene tetracyclic steroid nucleus. In pharmaceutical raw material development, every modification to the molecular structure directly determines solubility, receptor affinity, metabolic stability, and bioavailability. The structural design of Megestrol acetate is a classic example of steroid drug optimization.
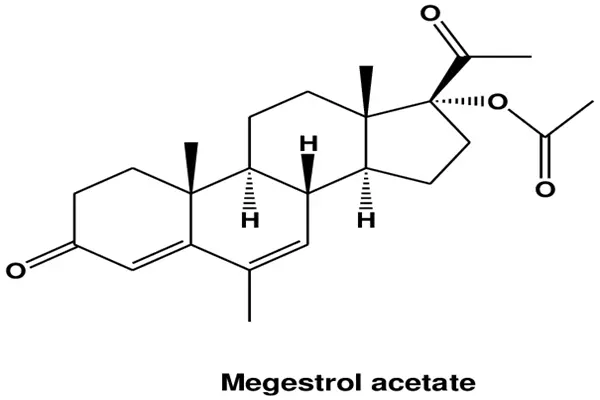
Firstly, the modification of the methyl group and double bond at the C6 position is a key breakthrough in efficacy. Natural progesterone has a hydrogen atom at the C6 position, and its affinity for the progesterone receptor is only about one-third that of melatonin. Megestrol acetate introduces a methyl group at the C6 position and forms a Δ⁶ double bond, enhancing receptor binding pocket adaptability through steric hindrance and electronic effects. In vitro experiments show that its PR binding activity reaches 130% of that of natural progesterone, while significantly reducing aromatase conversion efficiency, decreasing in vivo estrogen bypass production, and avoiding compensatory estrogen elevation in the tumor microenvironment. Sigma-Aldrich in vitro receptor binding experiments confirmed that MA has a binding constant Kd = 0.8 nM for the human progesterone receptor ligand binding domain, far superior to medroxyprogesterone. This structural difference allows it to achieve receptor saturation activation at low doses.
Secondly, C17α-acetylation is a key optimization for drug development. Progesterone has a hydroxyl group at C17. After oral administration, it is rapidly hydrolyzed by gastrointestinal esterases, with a first-pass metabolism rate exceeding 90% and a bioavailability of less than 10%. Megestrol acetate acetylates the C17α hydroxyl group, forming an ester bond protecting group, which delays hepatic metabolism, increasing oral bioavailability to 60%-70% and prolonging the plasma half-life to 14.0±2.12 hours, meeting the clinical requirements for 1-2 daily dosing. In the production of pharmaceutical raw materials, the acetylation reaction needs to be controlled at a temperature of 45-50℃ and a catalyst dosage of 0.5% to ensure selective binding of the acetyl group to the C17α position and avoid carbonyl side reactions at the C3 and C20 positions. The purity of the finished product must be ≥99.6%, with a total impurity content ≤0.3%, including ≤0.1% of 6-dehydrogenation byproducts; otherwise, it will lead to decreased receptor affinity and increased adverse reactions.
From the perspective of pharmaceutical raw materials, the structural optimization logic of Megestrol acetate is clear: site-specific modification of the steroid nucleus enhances receptor affinity and oral stability, crystal form control ensures formulation quality, and precise impurity management reduces toxic side effects. Its molecular structure retains the core activity of progestins while avoiding the drug-like defects of natural hormones, making it a classic model for structure-effect relationship research in steroidal raw materials and providing a structural design template for subsequent progestin-based anti-tumor drug development.
From contraception to palliative care for cancer, covering a wide range of clinical needs.
Core Application 1: Palliative Treatment of Hormone-Dependent Tumors
Megestrol acetate is a first-line palliative treatment active pharmaceutical ingredient (API) for advanced breast cancer and endometrial cancer, particularly suitable for postmenopausal breast cancer patients resistant to tamoxifen. Phase III clinical trials showed that daily administration of 160 mg Megestrol acetate to advanced breast cancer achieved an objective response rate of 22%-28%, a disease control rate of 56%-62%, and a median progression-free survival of 4.2 months, comparable to aromatase inhibitors with better tolerability. For endometrial cancer, Megestrol acetate inhibits abnormal endometrial proliferation, resulting in a tumor shrinkage rate of 18%-25% in advanced patients, alleviating symptoms such as vaginal bleeding and abdominal pain, and improving quality of life. Furthermore, Megestrol acetate also has adjuvant therapeutic effects on prostate cancer and kidney cancer. Its weak anti-androgen effect can inhibit the AR signaling pathway in prostate cancer cells, and when combined with docetaxel, it can increase the PSA reduction rate in advanced prostate cancer by 30%.
At the pharmaceutical raw material level, Megestrol acetate for tumor treatment must meet the requirements of high purity and low impurities, with heavy metal content ≤10ppm and microbial limits meeting sterile preparation standards to avoid infection caused by impurities in patients with advanced tumors. High-performance liquid chromatography (HPLC) is used throughout the industrial production process to ensure that the raw materials are free of estrogen-like impurities, preventing stimulation of tumor cell proliferation.
Second core application: Intervention in cancer cachexia-anorexia syndrome
This is the most clinically valuable new use of megestrol acetate, and it is currently the only drug for treating cachexia recommended as a Class 1 drug by the CSCO guidelines. Cancer cachexia is characterized by weight loss, muscle atrophy, and loss of appetite, affecting approximately 50%-80% of patients with advanced cancer, and 20% of patients die directly from cachexia rather than from the cancer itself. A phase III controlled trial conducted by Loprinzi et al. showed that daily administration of 800 mg of megestrol acetate to treat cancer cachexia resulted in an average weight gain of 5.2 kg, a 65% improvement in appetite scores, and a lean meat retention rate of 78% within 8 weeks, superior to traditional formulations prior to medroxyprogesterone acetate nanocrystals.
The advent of nanocrystalline Megestrol acetate raw material further breaks through application bottlenecks: traditional Megestrol acetate suspension requires a high-fat meal for administration, and its bioavailability is greatly affected by diet; nanocrystalline raw material has a particle size D90≤200nm, does not require a high-fat meal, and the average weight gain over 12 weeks is 5.8kg, which is 2.3 times that of non-nanocrystalline formulations, with an 80% shorter onset time, filling the gap in medication for patients with anorexia and dysphagia. In the production of pharmaceutical raw materials, nanocrystalline Megestrol acetate uses high-pressure homogenization technology, controlling the homogenization pressure at 1500bar and the number of cycles at 15 to ensure uniform particle size, avoid agglomeration, and achieve a dissolution rate ≥90%.
Core Application Three: Gynecological Contraception and Treatment of Endometriosis
Megestrol acetate was initially used as a raw material for short-acting contraceptives. It works by inhibiting the secretion of luteinizing hormone (LH) from the pituitary gland, blocking ovulation, and achieving a contraceptive success rate of over 99%. Low-dose megestrol acetate can regulate the endometrial cycle, treat dysfunctional uterine bleeding and endometriosis, reduce menstrual flow by 50%-70%, and relieve dysmenorrhea symptoms. In addition, megestrol acetate is also used for wasting syndrome in HIV-infected patients and malnutrition intervention in chronic obstructive pulmonary disease. Clinical data show that after 12 weeks of treatment for HIV-related wasting syndrome, the average weight gain was 3.8 kg, CD4+ cell count remained stable, and there were no significant metabolic side effects.
Multi-receptor cross-regulation achieves dual effects of anti-tumor and appetite stimulation.
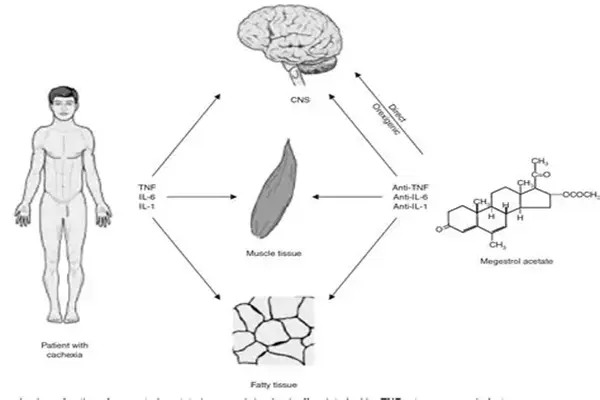
Megestrol acetate, a high-affinity agonist of PR (progesterone receptor), binds to PR in tumor cells, forming a PR-MA complex that translocates into the nucleus, inhibiting the transcription of downstream genes of the estrogen receptor and blocking ER-mediated tumor cell proliferation. In vitro cell experiments showed that treatment of MCF-7 breast cancer cells with megestrol acetate for 48 hours reduced the expression of proliferation-related proteins such as Cyclin D1 and c-Myc by 60%-70%, arrested the cell cycle at the G0/G1 phase, and increased the apoptosis rate by 35%. In PR-positive endometrial cancer cells, megestrol acetate also downregulated vascular endothelial growth factor expression, inhibited tumor angiogenesis, and reduced tumor blood supply. Nude mouse xenograft experiments showed that the tumor volume in the megestrol acetate treatment group was reduced by 58% compared to the control group, and the microvessel density decreased by 45%.
From the perspective of a pharmaceutical raw material expert, this product crosses the blood-brain barrier, activates hypothalamic GR and neuropeptide Y receptors, inhibits the leptin signaling pathway, and enhances the sensitivity of the appetite center. Animal experiments showed that after MA treatment in rats, hypothalamic NPY expression was upregulated by 2.1 times, leptin receptor expression decreased by 58%, food intake increased by 45%, and body weight increased rapidly. Clinical studies have confirmed that this product can reduce serum leptin levels by 32% and increase ghrelin levels by 28%, reversing anorexia symptoms at the neuroendocrine level.
Repurposing old drugs and formulation innovation: Expanding the new boundaries of pharmaceutical raw materials
Traditionally, megestrol acetate is primarily used for gynecological tumors and prostate cancer, but recent research focuses on non-small cell lung cancer (NSCLC) and neurodegenerative diseases. In vitro experiments show that megestrol acetate inhibits the EGFR/PI3K/AKT pathway in NSCLC cells, achieving a 38% inhibition rate of proliferation in gefitinib-resistant cells. In nude mouse xenograft experiments, its combination with osimertinib increased tumor shrinkage by 40% and reversed targeted drug resistance. Currently, a phase II clinical trial of megestrol acetate for the treatment of locally advanced NSCLC is underway. Preliminary data show that combined with chemotherapy, it can improve disease control rates by 25% and alleviate cachexia symptoms in 70% of patients.
Research in the field of neuroprotection has found that megestrol acetate, by activating the C/EBPβ pathway, inhibits aromatase expression in the hippocampus of rats with ischemia-reperfusion injury, reduces excitotoxicity, shrinks cerebral infarction volume by 42%, and improves neurological function scores by 50%. At the same time, megestrol acetate can inhibit microglial activation and reduce neuroinflammation, providing a new direction for the treatment of Alzheimer's disease and Parkinson's disease. It is currently in the preclinical research stage.
The combination of megestrol acetate and immune checkpoint inhibitors has become a hot research topic: Preclinical experiments have shown that megestrol acetate can remodel the tumor immune microenvironment, increase CD8+ T cell infiltration, and reduce Treg cells. Combined with PD-1 inhibitors, it can achieve a 45% complete tumor regression rate in melanoma mice, which is superior to monotherapy. Furthermore, megestrol acetate combined with CDK4/6 inhibitors in the treatment of HR-positive breast cancer can overcome drug resistance and prolong median progression-free survival by 3.5 months. Combined with metformin, it can reverse megestrol acetate-induced weight gain and reduce metabolic side effects. Phase III clinical trials have completed enrollment and are awaiting final data.
Recent research has found that megestrol acetate regulates adipocyte browning through the GR pathway, inhibiting the conversion of white adipose tissue to brown adipose tissue and reducing energy expenditure; this mechanism could be used to treat excessive emaciation. Simultaneously, megestrol acetate can regulate gut microbiota, increasing the abundance of beneficial bacteria, improving intestinal barrier function in cancer patients, and reducing intestinal inflammation, providing a new target for cachexia intervention. Furthermore, the regulatory mechanism of megestrol acetate on the autophagy pathway has been elucidated: low doses of megestrol acetate inhibit tumor cell autophagy, while high doses induce autophagic cell death, providing a theoretical basis for precise dosage control.
Conclusion
Megestrol acetate, a classic representative of steroidal pharmaceutical raw materials, is based on precise molecular structure design and focuses on multi-target pharmacological mechanisms. After more than 60 years of clinical validation, it has evolved from a contraceptive drug into a core drug for palliative cancer treatment and cachexia intervention, and continues to expand its application boundaries through the latest research. Its development history confirms the core logic of the pharmaceutical raw material industry: high-quality raw materials are the foundation of drug efficacy, while structural optimization and mechanism innovation are key to the continuation of drug value. In the future, with continuous breakthroughs in nano-formulations, combination therapies, and green synthesis technologies, Megestrol acetate will play a greater role in areas such as cancer treatment, nutritional intervention, and neuroprotection, becoming a timeless example of "drug repurposing" and providing a replicable scientific path for global pharmaceutical raw material research and development.
Xi'an Faithful BioTech Co., Ltd. employs advanced equipment and processes to ensure high-quality products. Our Megestrol acetate powder meets international pharmaceutical standards. Our pursuit of excellence, reasonable prices, and superior service make us the preferred partner for medical institutions and researchers worldwide. If you require research or production of Megestrol acetate, please contact our technical team at allen@faithfulbio.com.
References
1.Loprinzi, C. L., Jensen, M. D., Jiang, N. S., & Schaid, D. J. (1990). Controlled trial of megestrol acetate for the treatment of cancer anorexia and cachexia. Journal of the National Cancer Institute, 82(14), 1127–1132.
2.Muss, H. B., Case, L. D., Capizzi, R. L., et al. (1993). High versus standard-dose megestrol acetate in women with advanced breast cancer: A phase III trial. Journal of Clinical Oncology, 11(6), 1043–1049.
3.Ruiz-García, V., et al. (2018). Megestrol acetate for cachexia–anorexia syndrome: A systematic review. Journal of Cachexia, Sarcopenia and Muscle, 9(3), 444–452.
4.Sung, J. H., An, H. S., Jeong, J. H., et al. (2015). Megestrol acetate increases the proliferation, migration, and adipogenic differentiation of adipose-derived stem cells via glucocorticoid receptor. Stem Cells International, 2015, 1–10.
5.House, L., et al. (2018). Metabolism of megestrol acetate in vitro and the role of oxidative metabolites. Xenobiotica, 48(12), 1067–1075.
6.Lim, Y. L., et al. (2022). A systematic review and meta-analysis of the clinical use of megestrol acetate for cancer-related anorexia/cachexia. Frontiers in Oncology, 12, 875642.
7.Chen, Y., et al. (2025). Megestrol acetate is a specific inducer of CYP3A4 mediated by human pregnane X receptor. Biomedicine & Pharmacotherapy, 177, 116689.











