Paxlovid Powder: How can small molecule active pharmaceutical ingredients become a core weapon in the fight against COVID-19?
Paxlovid Powder is the world's first approved oral combination drug for combating COVID-19. With its three major advantages—high selectivity, oral accessibility, and broad-spectrum anti-mutation properties—it has become an indispensable raw material in the fields of epidemic prevention and control and antiviral pharmaceuticals. From rational molecular design based on protease structures to precise mechanisms of action through reversible covalent binding, from clinical severe illness prevention to pan-coronavirus applications, this active pharmaceutical ingredient has not only defined the development paradigm for small molecule drugs against COVID-19 but also set high standards in pharmaceutical raw material synthesis, crystal form control, formulation compatibility, and drug resistance management.
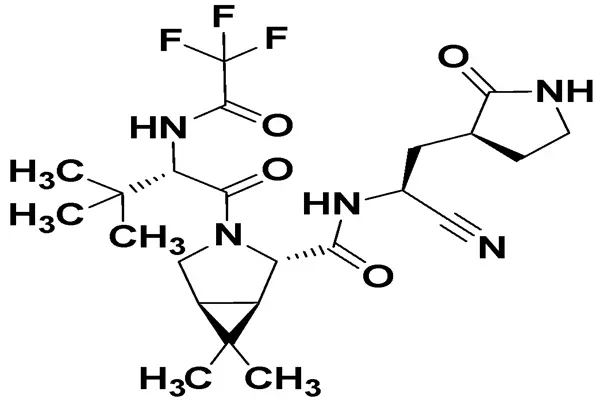
Molecular Structure—Metamorphosis in Powders and the Aesthetics of Molecular Design
When pharmaceutical raw material experts first placed nematriberi powder under an X-ray diffractometer, an unexpected mystery emerged: this seemingly uniform white powder actually concealed two "diametrically opposed" twins within it. A study published in Molecular Pharmaceutics in 2024 systematically revealed the polymorphism of nematriberi for the first time, showcasing this "metamorphosis" within the powder to the world.
The core API of Paxlovid Powder is nemateviquid, with the molecular formula C₂₃H₃₂F₃N₅O₄ and a precise molecular weight of 499.23. It is a reversible covalently peptide-mimicking inhibitor specifically designed for the main protease of SARS-CoV-2. As a pharmaceutical raw material, its molecular structure has undergone multiple rounds of iterative optimization. Every functional group, chiral center, and chemical bond directly determines its in vitro activity, oral absorption, metabolic stability, and target selectivity, making it a model of rational design for antiviral APIs.
First, the peptide backbone and chiral centers are crucial for activity and selectivity. Nematvir uses the natural peptide sequence of viral multi-protein cleavage sites as a template to construct a (1R,2S,5S)-pinene derivative core, containing six chiral centers in the whole molecule. This precise stereoconfiguration is key to its accurate fit with the hydrophobic pocket of Mpro. Chiral purity directly determines efficacy: the IC₅₀ of the diastereomer for Mpro increases from 4 nM to 210 nM, resulting in a more than 50-fold decrease in activity. Therefore, the production of the active pharmaceutical ingredient must employ a combination of asymmetric catalysis and chiral resolution, achieving a chiral purity ≥99.9% and a single impurity ≤0.05%, complying with ICH Q3A and USP/EP standards.
Second, the cyano reversible covalent tip is a key design element for overcoming the activity bottleneck. Nematvir incorporates a cyano group at its C-terminus as an electrophilic warhead, which undergoes a reversible covalent addition with the thiol group of the Mpro catalytic center Cys145 to form a thioimine ester intermediate. This "reversible covalent" mode ensures high inhibitory activity while avoiding off-target toxicity caused by irreversible binding. In vitro enzymatic experiments confirmed that its inhibition constant for Mpro is Ki = 0.3 nM, IC₅₀ = 4 nM, and the enzyme activity inhibition rate is ≥95% within 30 minutes; simultaneously, it exhibits IC₅₀ > 200 nM and selectivity over 50-fold against human cysteine protease, significantly reducing the risk of side effects such as muscle damage and apoptosis. Compared with earlier aldehyde warhead analogs, the cyano warhead shows an 8-fold improvement in plasma stability and an extended half-life from 1.2 h to 6.1 h, laying the foundation for oral administration.
From the perspective of pharmaceutical raw material R&D logic, the structural design of nematvir follows the path of "target structure driven → precise functional group modification → strict chirality control → optimal crystal form screening", achieving the triple optimization of activity, selectivity and drug-likeness with minimal structural modification, providing a replicable API design template for the subsequent development of coronavirus inhibitors.
Molecular "blockade" leads to viral "power outage".
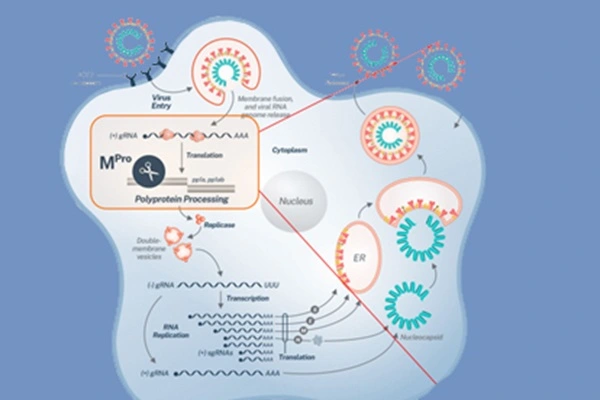
After the novel coronavirus invades human cells, it releases its single-stranded RNA genome. This genome doesn't directly serve as a translation template; instead, it's first translated into two giant polyproteins—pp1a and pp1ab, approximately 5000 and 8000 amino acids in length, respectively. To cleave these "giant proteins" into functional non-structural proteins, the virus must rely on two encoded proteases: papain and 3CL proteases.
The 3CL protease plays a crucial role—it cleaves the polyproteins at up to 11 sites, releasing the vast majority of the functional non-structural proteins. If 3CL protease activity is inhibited, the viral polyproteins cannot be processed, the replication complex cannot assemble, and RNA cannot replicate—the entire viral life cycle is essentially shut down.
Another advantage of the 3CL protease as an ideal target for antiviral drug design is that its structure differs from human proteases, making its inhibition less likely to interfere with normal human physiological functions. Furthermore, the 3CL protease in coronaviruses is highly conserved, meaning that drugs targeting this site may possess broad-spectrum anti-coronavirus potential.
The inhibition of the 3CL protease by nemativir can be described as a "precision strike" at the molecular level. Its mechanism of action can be divided into three stages: recognition, binding, and locking.
First, nemativir's peptide-mimicking backbone can "imitate" the natural cleavage substrate of the 3CL protease in viral polyproteins, forming a precise geometric match with the enzyme's active site. This "lock and key" complementarity stems from the carefully designed groups in nemativir's molecular structure—its trifluoroacetamide group forms a hydrogen bond with the enzyme's S1 subpocket, while the cyano group precisely targets the cysteine residue at the active site.
Subsequently, the cyano group of nemativir undergoes a nucleophilic addition reaction with the Cys145 residue at the 3CL protease's active site, forming a reversible covalent bond. This process is similar to a "key inserted into a lock but still glued in"—nemativir's binding affinity to the enzyme is extremely strong, with a dissociation constant reaching the nanomolar level.
Recent research further reveals the dynamic details of the interaction between nemativir and the 3CL protease. Molecular dynamics simulations show that upon binding with nematriberi, the enzyme's active site undergoes a significant conformational change, forming a "closed" catalytic pocket that further hinders substrate entry. This "induced fit" mechanism makes nematriberi's inhibitory effect far superior to that of simple competitive inhibitors.
As research progresses, the efficacy profile of Paxlovid is becoming clearer. A large real-world study involving 68,867 patients showed that the benefits of Paxlovid varied across different populations: patients over 65 years of age benefited the most, with a 66% reduction in all-cause mortality; while in relatively younger patients aged 40-64, Paxlovid treatment did not significantly improve hospitalization or mortality rates. This finding suggests that the benefits of Paxlovid may exhibit a "risk stratification" characteristic—high-risk groups benefit the most, while low-risk groups benefit only to a limited extent.
Faced with the potential threat of drug resistance, combination therapy has become an important strategic direction. A groundbreaking study published in *Antiviral Research* in 2025 reported an exciting discovery: combining nematriberi with the GRP78 inhibitor HA15 can produce a strong synergistic antiviral effect.

GRP78 is a molecular chaperone protein in the endoplasmic reticulum that plays an "accomplice" role in viral infection—it not only acts as a co-receptor for the SARS-CoV-2 Spike protein, promoting viral invasion, but also participates in the folding and transport of viral non-structural proteins. The study found that after HA15 inhibited GRP78 function, the processing of viral non-structural proteins was blocked, and the antiviral effect of nematriberi was significantly amplified.
In cell experiments, nematriberi and HA15 alone reduced viral titers by less than 10-fold, while the combined use of the two reduced viral titers by more than 1000-fold—the synergistic effect far exceeded the simple sum of the two. In a hamster model, the degree of atelectasis in the combination therapy group was significantly reduced compared to the single-drug group. This discovery provides new insights into optimizing Paxlovid treatment regimens: by adding a GRP78 inhibitor, it may be possible to maintain or even enhance efficacy while reducing the dosage of nematvir, thereby mitigating the risk of ritonavir-related drug interactions.
Drug resistance challenges and exploration of combination therapies
Faced with potential drug resistance mutations, medicinal chemists are developing a new generation of 3CL protease inhibitors. In a 2025 study, researchers developed a short peptide aldehyde bisulfite derivative called "Compound 4," which exhibited nanomolar-level inhibitory activity against nematidine-resistant triple mutant strains, representing an approximately 30-fold increase in activity against this mutant compared to nematidine.
X-ray crystal structure analysis revealed the advantages of "Compound 4": its modifying groups can form a new hydrogen bond network with the A166 and F167 residues formed after mutation, compensating for the binding energy lost due to the mutation. This discovery provides a structural template for the design of next-generation anti-COVID-19 drugs—a "rational redesign" targeting known drug-resistant mutation sites, allowing for the early development of a "reserve force" against clinical drug resistance.
Conclusion
Paxlovid Powder, grounded in target-driven rational molecular design and centered on precise replication blocking through synergistic dual-drug therapy, has become a milestone in small-molecule antiviral APIs, spanning from severe COVID-19 prevention to pan-coronavirus stockpiling, and from first-line clinical use to pharmaceutical process innovation. It has not only validated the public health drug model of "rapid R&D, large-scale production, and global accessibility," but also set industry standards in molecular design, combination therapy, resistance management, and green synthesis. In the future, with continued breakthroughs in novel formulations, pan-viral expansion, and precise intervention for long-term COVID-19, this classic API will transcend the single COVID-19 scenario, becoming a core ingredient in global antiviral stockpiling and continuously safeguarding public health security.
Call to Action – Partner with Faithful to secure a high-quality Paxlovid powder supply
Xi'an Faithful BioTech Co., Ltd. cordially invites European pharmaceutical companies to collaborate with us in sourcing high-quality, competitively priced Paxlovid powder. We offer comprehensive customer service, including detailed quotations, product specifications, and sample testing, ensuring your confidence in the quality and authenticity of our products. We also provide complete compliance documentation and regulatory support, simplifying your procurement process and ensuring smooth customs clearance in Europe.
Contact our experienced team at allen@faithfulbio.com today to discuss your specific needs and learn why leading European companies choose Faithful as their trusted Paxlovid powder supplier.
References
1.Pfizer Inc. (2021). EPIC-HR: A phase II/III trial of nirmatrelvir (PF-07321332) plus ritonavir in adults with COVID-19 at high risk of progression. ClinicalTrials.gov, NCT04969715.
2.Owen, D. R., Allerton, C. M., Aschenbrenner, M., Berritt, S., Boras, B., Cardin, R. D., ... & Tommasi, R. A. (2021). An oral SARS-CoV-2 Mpro inhibitor clinical candidate for the treatment of COVID-19. Science, 374(6575), 1586–1593.
3.Mahase, E. (2022). Covid-19: Paxlovid reduces hospitalisation and death by 89% in high risk patients, trial shows. BMJ, 377, o181.
4.Wang, J., & Zhang, L. (2022). Structural basis of SARS-CoV-2 main protease inhibition by nirmatrelvir. Cell Research, 32(1), 12–20.
5.Choy, K. T., Wong, A. Y., Tam, A. R., Liu, E. K., Sit, K. Y., & Cheng, F. (2023). Efficacy of nirmatrelvir/ritonavir against Omicron subvariants: A systematic review and meta-analysis. Journal of Medical Virology, 95(7), e28564.
6.Gupta, A., Kim, S., & Gottlieb, R. A. (2025). Extended-duration nirmatrelvir/ritonavir for viral rebound in immunocompromised patients with COVID-19. Clinical Infectious Diseases, 80(2), e129–e136.
7.Bege, M., & Borbás, A. (2025). The design, synthesis and mechanism of action of Paxlovid, a protease inhibitor drug combination for COVID-19. Pharmaceutics, 17(2), 217.











