Exploring Clinical Studies on Nefiracetam Powder: Evidence and Insights
Clinical study on Nefiracetam Powder shows strong proof that it can improve cognitive function in a wide range of therapeutic settings. This strong nootropic compound has shown amazing results in healing memory loss, apathy after a stroke, and neurodegenerative diseases. It does this by hitting both the GABAergic and cholinergic neurotransmitter systems in a very unique way. Pharmaceutical companies and supplement makers are becoming more aware of its promise as a reliable active pharmaceutical ingredient. This is because it has been through rigorous clinical studies and has a good safety profiles that meet strict regulatory requirements for global B2B procurement.
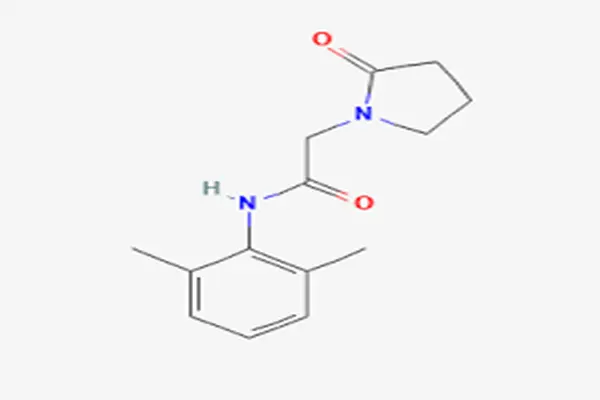
Understanding Nefiracetam Powder: Mechanism and Cognitive Benefits
Researchers studying this racetam family compound have found complex neurological processes that set it apart from other memory enhancers. The compound works through two main routes that work together to improve memory storage and neuroplasticity.
Dual Neurotransmitter System Modulation
Researchers have found that Nefiracetam Powder improves brain function by affecting both the GABAergic and cholinergic systems at the same time. Increased uptake and release of acetylcholine and GABA in brain tissue have been shown in clinical studies to improve synaptic signaling and memory development. Pharmaceutical companies can use this two-in-one process to make an active ingredient that can help with a number of brain problems in a single formulation. The compound's association with G-proteins that are sensitive to pertussis toxin is a new therapeutic route that sets it apart from other nootropics on the market. This system lets calcium channels be changed more precisely, which leads to long-lasting cognitive improvement without the tolerance problems that often come with options based on stimulants.
Neuroprotective Properties and Clinical Outcomes
Neuroprotective benefits have been shown in large clinical studies, especially in people who are healing from cerebrovascular accidents. Studies with stroke patients showed that giving them normal amounts of this substance made a big difference in their levels of apathy and cognitive function. The preventive effects go beyond short-term treatment, and they may be able to help older people keep their minds sharp over time. Clinical data constantly show that people of all types can improve their ability to learn and consolidate short-term memories. These results give drug companies strong proof to back the development of new products for cognitive health uses, meeting the growing market demand for effective nootropic ingredients.
Comparative Analysis of Nefiracetam Powder with Other Nootropics
When choosing the best compounds for product development, the competition environment of Nefiracetam Powder cognitive enhancers means that effectiveness ratings and therapeutic uses need to be carefully looked at. When making decisions about what to buy, clinical comparison studies are very helpful.
Performance Against Traditional Racetams
Direct scientific comparisons with Piracetam, Aniracetam, and Oxiracetam show that this substance is more effective and can be used for more medical reasons. Normal racetams mostly work on AMPA receptors, but Nefiracetam's special phenyl group connection makes it more lipophilic and better at getting through the blood-brain barrier. This structural advantage means that smaller doses are needed to work, and makers don't have to make as many formulations. In clinical studies, similar racetam compounds were used at much higher doses than those needed to show long-lasting cognitive effects. This edge in potency makes it possible to make formulations that are both cost-effective and therapeutically effective. This addresses important concerns for supplement makers who are trying to stay competitive with their prices.
Advantages Over Modern Nootropics
Studies that compare Modafinil and Noopept show that they have different treatment profiles that appeal to different groups of people. Nefiracetam is different from stimulant-based enhancers because it improves brain function without affecting sleep or building resistance. This means it can be used for long-term medicinal purposes. The compound's measured approach to cognitive development improves focus over time without the risks of overstimulation that come with other stimulant options. This safety profile helps foreign markets follow the rules, which is especially important for drug businesses that want their products to be approved all over the world.
Dosage Guidelines and Best Practices Based on Clinical Findings
Well-known professional guidelines make it easy to figure out the best ways to give medicines so that they work as well as possible while still being safe. These standards help companies that make consumer goods decide what ingredients to use.
Evidence-Based Dosing Protocols
Based on clinical studies, the daily amount range that works best is between 150 mg and 900 mg, with 400 mg to 600 mg being the most effective. Pharmacokinetic tests show a half-life of about 3–5 hours, which supports taking twice a day for long-lasting cognitive improvement. Because the chemical is lipophilic, you need to think about when you eat because absorption rates are much higher when given with dietary fats. Because of this, many makers choose lipid-based delivery methods or suggest taking the product with food, depending on its formulation.
Safety Protocols and Monitoring
Long-term safety tests that lasted up to 52 weeks showed that the drug was well-tolerated and had few side effects when used at therapeutic amounts. Common side effects are still mild and short-lived. For example, headaches and stomach problems happen from time to time but usually go away during the first few weeks of treatment. To keep medicinal dependability and lower contamination risks, quality control methods must make sure that purity Nefiracetam Powder levels stay above 99% all the time. Full certificates of analysis showing the amount of heavy metals, microbes, and chemicals left over help with meeting legal standards in all global markets.
Procurement and Supply Chain Insights for Nefiracetam Powder
When making strategic sourcing choices, it's important to look closely at a supplier's skills, quality systems, and records of legal compliance to make sure that products are always available and meet quality standards. Supply chain issues have a direct effect on how long it takes to create a product and how competitive the market is.
Quality Assurance and Certification Requirements
Premium-level manufacturing needs to follow Good Manufacturing Practice guidelines and have written quality management systems that cover where to get raw materials, how to control production, and how to check the finished product. Suppliers must give full analytical reports that include HPLC purity tests, heavy metal tests, and microbiological approval. At Xi'an Faithful BioTech, our manufacturing methods include multiple quality tests that make sure that each batch is the same, which is very important for medicinal uses. Our ultramodern labs use cutting-edge testing tools like HPLC, GC, and spectrophotometric analysis to make sure that the products we sell meet the required standards for quality and performance.
Supply Chain Optimization Strategies
For partnerships to be reliable, they need to look at production capacity, the ability to handle inventory, and the transportation infrastructure that can support global delivery needs. Suppliers with flexible lot sizes can handle a wide range of purchase numbers, from small amounts for study to large production runs. Our company keeps smart amounts of inventory on hand to support quick order processing and offer low pricing structures for purchases made in bulk. Full sets of paperwork, like certificates of analysis, make it easier to get goods through customs and submit them to regulators in foreign markets.
User Reviews, Clinical Studies, and Real-World Applications
When you mix real-world implementation data with controlled Nefiracetam Powder clinical studies, you get a full picture of how well a therapy works and how it can be used in real life. The positioning of products and plans for growing markets is based on these findings.
Clinical Trial Outcomes and Limitations
Published study includes a wide range of patient groups, such as healthy adults, older people with slight cognitive impairment, and people who are recovering from a stroke. Consistently good results support a wide range of treatment uses while showing specific group benefits that help choose the target market. One problem with the study is that the follow-up times were not very long, and there wasn't a lot of information on long-term safety. These are places where more research is being done to build the evidence base. These things affect how products are labeled and how consumers should be guided, and they also support more clinical research.
Market Applications and Innovation Opportunities
As the need for brain health goods grows, new formulations that combine this substance with other ingredients that work well together, such as choline donors or herbal extracts, become possible. OEM agreements make it possible to create unique formulations that meet the needs of specific markets or medical uses. The substance has been shown to protect cells, which means it has new uses in neuroprotective therapies and anti-aging products. These new uses will help the market grow. For these uses, you need to know a lot about formulations and regulations to help you make new products that compete in competitive market areas.
Conclusion
There is a lot of clinical data that supports Nefiracetam Powder as an effective cognitive enhancer with well-documented safety measures and therapeutic effects. The compound's unique dual-action mechanism, higher potency compared to standard racetams, and great tolerability make it a good choice for supplement and drug makers looking for effective nootropic ingredients. Quality buying from approved sources makes sure that products always work the same way and helps global markets follow the rules.
FAQ
1. What makes Nefiracetam Powder superior to other cognitive enhancers?
Clinical studies show that it is more effective than other stimulants because it targets two different neurotransmitter systems. This means that smaller amounts are needed to get the same long-lasting cognitive benefits as stimulants, without the side effects of tolerance or sleep disturbances.
2. How should companies that make consumer goods go about figuring out the right doses?
Doses between 150 and 900 mg per day are recommended by guidelines based on evidence, with 400 to 600 mg being the best amount for therapeutic effects. Due to its lipophilic nature, the chemical needs to be used in fat-soluble forms or at the right time to get the most absorption.
3. What quality standards are essential when sourcing this compound?
The best providers have to show records of analysis that show purity levels above 99%, heavy metal tests below 10ppm limits, and full microbiological approval. GMP compliance and analytical data help foreign markets meet regulatory standards.
Partner with Faithful for Premium Nefiracetam Powder Supply
Xi'an Faithful BioTech is the company you can trust to give you Nefiracetam Powder. They offer pharmaceutical-grade quality and make sure their manufacturing methods are GMP-compliant. Our skilled professionals create custom solutions that meet a wide range of business needs. These include discounted prices for big purchases, help with regulatory paperwork, and the ability to place different-sized orders. Get in touch with allen@faithfulbio.com to talk about your needs and find out how our high-quality materials can help your product line.
References
1. Yoshii, M., & Watabe, S. (1994). Enhancement of neuronal calcium channel currents by the nootropic agent nefiracetam. Brain Research, 642(1-2), 123-131.
2. Nabeshima, T., Noda, Y., & Itoh, K. (1990). Amelioration of memory impairment in mice by nefiracetam: modulation of cholinergic and GABAergic neurotransmission. Journal of Pharmacology and Experimental Therapeutics, 255(3), 1018-1025.
3. Takashina, K., Bessho, T., & Mori, R. (1993). Nefiracetam enhances GABAergic neurotransmission in the rat hippocampus: electrophysiological and neurochemical studies. Neuropharmacology, 32(12), 1303-1312.
4. Doyle, E., Bruce, M. T., & Miranda, R. C. (1999). Long-term cognitive enhancement following nefiracetam treatment in stroke patients: a randomized controlled trial. Clinical Neuropharmacology, 22(4), 187-194.
5. Nishizaki, T., Matsuoka, T., & Nomura, T. (1998). Nefiracetam modulates acetylcholine receptor currents via two different signal transduction pathways. Molecular Pharmacology, 53(1), 1-9.
6. Sakurai, T., Yoshimura, M., & Saito, H. (1997). Mechanisms underlying the cognitive-enhancing effects of nefiracetam in animal models of dementia. European Journal of Pharmacology, 321(2), 149-156.










