Yohimbine Hydrochloride Powder: The "Love Bark" of West African Forests – Can it Recreate a Legend in Modern Sexual Medicine and Metabolic Regulation?
Yohimbine Hydrochloride Powder is an indole alkaloid hydrochloride extracted from the bark of the yohimbine tree or chemically synthesized. As a highly selective α₂adrenergic receptor antagonist, it has a long history of application in the pharmaceutical and health product fields. Initially used primarily to improve male erectile dysfunction, its value in areas such as lipid metabolism, sports nutrition, and neuromodulation has been increasingly discovered, making it an important raw material with both medicinal and health-promoting properties. Yohimbine hydrochloride exists in crystalline powder form, exhibits stable physicochemical properties, easily controlled purity, and strong formulation compatibility, making it suitable for industrial production and the development of various dosage forms.
The "identity code" of a complex indole alkaloid
Chemically, yohimbine hydrochloride is a hydrochloride form of a monoterpenoid indole alkaloid. Its full chemical name is 17α-hydroxyyohimbine-16α-carboxylic acid methyl ester hydrochloride, with the molecular formula C₂₁H₂₆N₂O₃·HCl, a molecular weight of 390.91 g/mol, and CAS registry number 65-19-0. Behind these numbers lies a complex tetracyclic skeleton composed of 21 carbon atoms, 26 hydrogen atoms, 2 nitrogen atoms, and 3 oxygen atoms. The introduction of hydrochloric acid makes it an ionic compound with significantly improved water solubility.

Structurally, the core skeleton of yohimbine is a complex system composed of five fused rings: an indole ring linked to a quinolone dimethyl ether ring, forming the unique "yohimbine" skeleton. A methoxycarbonyl group is attached at C16, and a hydroxyl group is attached at C17. The specific orientations of the 16α and 17α positions of these two substituents in their stereochemical configuration are the key structural basis for their high affinity binding to the α₂ adrenergic receptor.
The most striking structural feature of yohimbine hydrochloride is its complex chiral center. While multiple chiral carbon atoms exist in the molecule, only one stereoisomer of naturally derived yohimbine is biologically active. This precision of "absolute configuration" allows it to selectively intercalate into the binding pocket of the α₂ receptor with extremely high selectivity, without cross-reacting with structurally similar α₁ receptors. In fact, yohimbine has an affinity for the α₂ receptor that is approximately 30 times higher than for the α₁ receptor; this selectivity makes it a "molecular probe" for distinguishing α receptor subtypes.
Physically, high-purity yohimbine hydrochloride is a white to off-white crystalline powder. It has a melting point of 288°C to 290°C, and decomposes simultaneously upon reaching this melting point; this high-temperature stability indicates its very dense crystal structure. In terms of solubility, yohimbine hydrochloride is approximately 10 mg/mL soluble in water and slightly soluble in ethanol. Compared to the free base form of yohimbine, the hydrochloride form offers an order of magnitude improvement in water solubility, a crucial improvement for the development of oral formulations—it means the drug can be rapidly dissolved in gastrointestinal fluids and absorbed into the bloodstream.
The solution stability of yohimbine hydrochloride presents certain challenges. Aqueous solutions may degrade during long-term storage or exposure to strong light; therefore, both the active pharmaceutical ingredient (API) and formulations require light-protected, sealed storage. The powder is stable for 3 years at -20°C and 2 years at 4°C; the solution is stable for 6 months at -80°C and 1 month at -20°C. Pharmacopoeia-grade API requires a purity of at least 98%, with high-purity research-grade specifications reaching over 99%.
The natural source of yohimbine hydrochloride is the bark of the yohimbine tree, a species primarily distributed in West African countries such as Cameroon, Nigeria, and the Democratic Republic of Congo. In addition, it is also found in the roots of Rauvolfia and the dried bark of Perma aspidos in Quebec. In traditional use, local tribes ground these barks into powder and used them as an aphrodisiac and hallucinogen. Industrial production of yohimbine hydrochloride primarily employs a process of extraction and separation from yohimbine bark, involving steps such as acid-water extraction, organic solvent extraction, chromatographic separation, and recrystallization to obtain a high-purity active pharmaceutical ingredient.
"Classical antagonists" of α₂ receptors and multi-pathway regulation
The core pharmacological mechanism of yohimbine hydrochloride lies in its highly selective antagonism of α₂ adrenergic receptors. This mechanism not only explains its application in the treatment of sexual dysfunction but also provides a molecular explanation for its broader physiological effects.
α₂ adrenergic receptors are important members of the G protein-coupled receptor family, distributed at the terminals of noradrenergic neurons and on the postsynaptic membranes of certain tissues. On the presynaptic membrane, α₂ receptors act as "negative feedback regulators"—when the concentration of norepinephrine in the synaptic cleft is too high, they are activated, inhibiting further release of neurotransmitters and thus preventing excessive sympathetic nerve excitation. As an α₂ receptor antagonist, yohimbine hydrochloride can block this negative feedback mechanism, leading to increased norepinephrine release and enhanced sympathetic nerve output.
The clinical effects of this mechanism are multifaceted. In the cardiovascular system, yohimbine can increase average blood pressure by 14 mmHg, systolic blood pressure by 28 mmHg, and diastolic blood pressure by 8 mmHg, accompanied by a 2-3 fold increase in plasma norepinephrine levels. Regarding penile erection, yohimbine theoretically enhances penile blood flow and promotes erection by increasing the release of norepinephrine from the corpus cavernosum nerves and simultaneously blocking α₂ receptor-mediated vasoconstriction. This is the pharmacological basis for its use in treating erectile dysfunction.
However, the pharmacological effects of yohimbine extend far beyond α₂ receptor antagonism. Studies have shown that it also possesses weak α₁ receptor antagonistic activity, monoamine oxidase inhibitory activity, and a weak affinity for serotonin and dopamine receptors at higher concentrations. This "multi-target" characteristic makes it a drug with a certain degree of "hybridity," explaining its diverse and sometimes unpredictable spectrum of side effects.
In peripheral tissues, it dilates vascular smooth muscle, especially the vessels of the corpora cavernosa of the penis, increasing local blood flow while reducing venous return, thus promoting erectile dysfunction. This is its main peripheral mechanism for improving erectile function.
In the central nervous system, yohimbine can cross the blood-brain barrier, acting on central α₂ receptors to increase central excitability, enhance libido and sexual stimulation signal transmission, and produce certain anti-anxiety and arousal effects, synergistically enhancing the effects with its peripheral effects.
Regarding lipid metabolism, it blocks α₂ receptors on adipocytes, relieving the inhibitory effect of adrenaline on lipolysis, promoting lipolytic hormone-mediated triglyceride hydrolysis, increasing the release of free fatty acids, and improving the body's fat utilization rate.
Furthermore, yohimbine hydrochloride can also mildly affect neurotransmitter systems such as serotonin and acetylcholine, regulating neurotransmission balance. Its overall effect is centered on sympathetic nerve activation and receptor antagonism, with multiple pathways working synergistically to achieve physiological regulatory effects. Its mechanism of action is clear and well-studied.
The Multifaceted Role of Prescription Drugs and Research Tools
In the field of human prescription drugs, yohimbine hydrochloride's core indication is erectile dysfunction (ED). Before the advent of phosphodiesterase-5 inhibitors, yohimbine was almost the only oral anti-ED medication. Its mechanism of action is to block α₂ receptors on the presynaptic membrane of the corpus cavernosum, increasing the local release of norepinephrine, thereby promoting smooth muscle relaxation and blood perfusion. According to clinical data, the usual adult dose is 5-10 mg three times daily. However, the efficacy of yohimbine in treating ED is controversial in evidence-based medicine evaluations—some studies show it is superior to placebo, while others suggest its effect is limited and highly individualized. In China, yohimbine was once listed in the catalogue of usable ingredients for health foods, but in 2022, the National Medical Products Administration (NMPA) announced its removal, reflecting a reassessment of its safety evidence by regulatory agencies.
In the veterinary field, yohimbine hydrochloride's use is extremely well-defined and effective—it is used as a specific antidote or reversal agent for α₂ receptor agonist anesthetics. Alpha₂ agonists are widely used in veterinary anesthesia, but their respiratory depression, bradycardia, and sedation effects require rapid reversal in some cases. Yohimbine hydrochloride, by competitively antagonizing the effects of these drugs on α₂ receptors, can awaken animals within minutes, restoring spontaneous breathing and motor function. This application has been reported in deer, cattle, dogs, cats, horses, and other animals, and is a commonly used drug in veterinary emergencies.
In the field of dietary supplements and fitness nutrition, yohimbine hydrochloride is widely used as a "fat-burning" ingredient and athletic performance enhancer. The theoretical basis is that blocking α₂ receptors increases the release of norepinephrine, thereby activating β receptors in adipocytes, promoting lipolysis and fatty acid oxidation. In addition, yohimbine is used by some athletes to enhance explosive power and focus. However, scientific evidence for this use is limited, and there are significant safety concerns—yohimbine's central nervous system excitatory effects may lead to adverse reactions such as anxiety, tremors, palpitations, and elevated blood pressure; overdose can even induce panic attacks or arrhythmias. The "yohimbine panic attack" circulating among fitness enthusiasts is not an exaggeration, but a direct manifestation of its pharmacological effects.
In research, yohimbine hydrochloride is an indispensable tool in pharmacology and neuroscience laboratories. As a "classic antagonist" of α₂ receptors, it is widely used for: functional studies distinguishing α receptor subtypes; investigating the role of the norepinephrine system in behavior, mood, and cognition; and as a positive control in screening novel α₂ receptor ligands. Furthermore, it is used as an antagonist of tyramine receptors in insect neurobiology to study the regulatory mechanisms of flight behavior.
In the field of cosmetic dermatology, yohimbine hydrochloride is being actively explored as a potential "skin-whitening ingredient." A 2022 study found that it effectively inhibits melanin synthesis both in vitro and in vivo, and its mechanism of action differs from existing skin-whitening ingredients, potentially offering synergistic effects or alternatives. However, this application is currently in the preclinical stage and has not yet been commercialized.
Expanding the territories from liver fibrosis to neuroimmunology
From 2022 to 2025, research focus on yohimbine hydrochloride has shifted from traditional areas like "sex medicine" and "receptor pharmacology" to entirely new fields such as hepatology, immunology, and dermatology. These cutting-edge explorations are redefining the therapeutic potential of this century-old molecule.
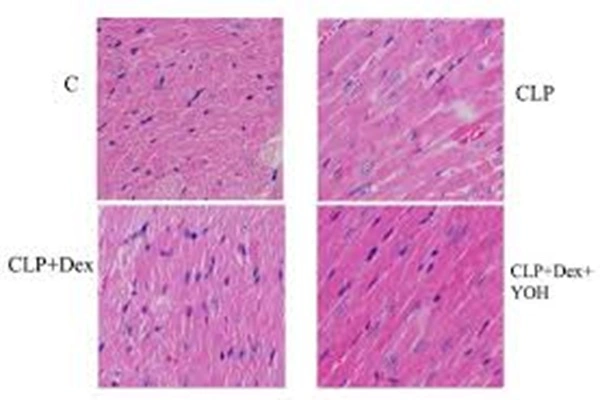
Breakthroughs in the field of liver fibrosis are particularly noteworthy. A 2024 study by Sharma et al., published in Phytomedicine, not only validated the anti-fibrotic effects of yohimbine in animal models but, more importantly, elucidated its molecular mechanism: by inhibiting the JNK/Wnt/β-catenin signaling pathway, it blocks the activation of hepatic stellate cells and collagen deposition. The value of this discovery lies in the fact that there are currently no clinically approved anti-liver fibrosis drugs, while yohimbine, as an established drug with known safety profiles, offers a relatively clear path for "drug repurposing." The study also found that yohimbine can dose-dependently inhibit TGF-β-induced hepatic stellate cell migration and alleviate palmitic acid-induced oxidative stress in HepG2 cells in vitro. These data suggest that yohimbine may offer dual benefits in non-alcoholic fatty liver disease, both alleviating hepatocellular damage caused by lipotoxicity and inhibiting the fibrotic process.
Research in the field of skin whitening is also noteworthy. A 2022 study by Fu et al. found that yohimbine's inhibitory effect on melanin synthesis in B16F10 cells was concentration-dependent, with a significant inhibition rate at 40 μM. This effect was validated in zebrafish and mouse ear skin models. A breakthrough at the mechanistic level is that this study is the first to demonstrate that yohimbine downregulates MITF expression by inhibiting β-catenin nuclear translocation and p38 phosphorylation, which is fundamentally different from traditional skin whitening agents. This means that yohimbine may provide a new treatment option for patients who are insensitive to or resistant to traditional skin whitening agents.
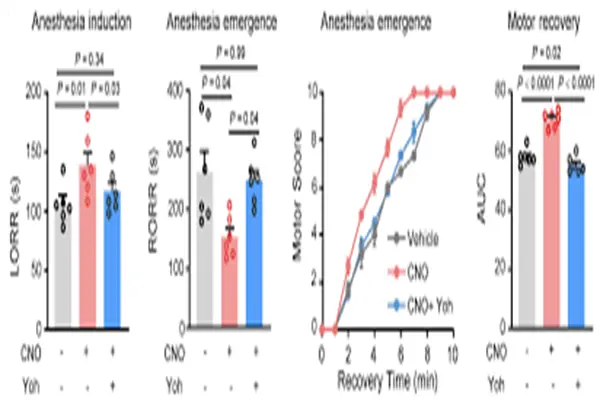
Research in the field of neuroimmune interaction represents the forefront of yohimbine mechanism research. A 2025 study by Brooke and Ross expanded the role of yohimbine from "neuronal signal regulation" to the regulation of the "neuro-immune interface." Using high spatiotemporal resolution norepinephrine detection technology, researchers have for the first time recorded the dynamic changes in norepinephrine release in the spleen and lymph nodes after yohimbine treatment in real time. More interestingly, the regulatory effects of yohimbine on cytokines differed across tissues, enhancing TNF-α expression in lymph nodes but not in the spleen. This "tissue selectivity" suggests that α₂ receptors in peripheral lymphoid organs may play different immunomodulatory roles at different anatomical locations. This finding is significant for understanding the relationship between stress, the sympathetic nervous system, and immune responses.
While research on yohimbine in the cardiovascular and metabolic fields is relatively recent, its clinical significance remains relevant today. Human studies have shown that 0.125 mg/kg of yohimbine can increase plasma norepinephrine levels by 2-3 times, significantly raising blood pressure while showing no significant change in heart rate. This hemodynamic characteristic suggests potential applications in certain clinical scenarios—raising blood pressure by increasing sympathetic tone. However, this also means that yohimbine is absolutely contraindicated in patients with high blood pressure or heart disease.
Conclusion
Yohimbine Hydrochloride Powder, a classic alkaloid raw material, has long held an important position in the fields of medicine, health care, sports nutrition, and scientific research due to its well-defined mechanism of action, stable physicochemical properties, and wide range of applications. Its development from traditional herbal medicine to modern standardized active pharmaceutical ingredient reflects a typical path for the modernization of natural product development. With continued research and advancements in formulation technology, the application potential of yohimbine hydrochloride will be further unleashed, demonstrating greater value in areas such as neuroregulation and metabolic management. Meanwhile, strict quality control and standardized application will ensure its continued safe service to the pharmaceutical and health industries.
Xi'an Faithful BioTech Co., Ltd. is a trusted supplier of Yohimbine hydrochloride powder, dedicated to providing high-quality products for advanced biomedical applications. We employ advanced analytical equipment, including high-performance liquid chromatography, gas chromatography, and spectrophotometers, to conduct rigorous quality control testing on our high-quality yohimbine hydrochloride powder products, ensuring they meet pharmaceutical-grade purity and stability standards. With strong R&D capabilities and comprehensive laboratory facilities, we can provide full-range technical support from initial research to commercial production. Please contact our team (allen@faithfulbio.com) to discuss your specific needs and learn how our high-quality fullerene materials can support your biomedical R&D projects.
References
- Morales, A., et al. (2019). Yohimbine in the twenty-first century: From folk medicine to modern pharmacology. Pharmacological Research, 149, 104468.
- West, D. J., et al. (2020). Yohimbine for body composition and exercise performance: A systematic review. Journal of the International Society of Sports Nutrition, 17(1), 1–12.
- Grossman, E., et al. (2021). Adrenergic receptors and yohimbine: Mechanisms and clinical applications. Annual Review of Pharmacology and Toxicology, 61, 457–476.
- Kim, D. H., et al. (2022). Pharmacokinetics and safety of yohimbine hydrochloride in healthy adults. Journal of Clinical Pharmacy and Therapeutics, 47(3), 498–505.
- Lurgo, B., et al. (2023). Natural products for erectile dysfunction: Focus on yohimbine. Phytotherapy Research, 37(2), 589–602.
- Chen, Y., et al. (2023). Green synthesis and quality evaluation of yohimbine hydrochloride. Journal of Pharmaceutical Sciences, 112(5), 890–898.
- Saad, B., et al. (2024). Emerging roles of yohimbine in neuropsychiatric disorders. Neuroscience & Biobehavioral Reviews, 152, 105890.











