Madecassoside powder: Why has Centella asiatica active saponin become a core medicinal ingredient for skin repair, anti-inflammation and anti-aging?
Madecassoside powder is a pentacyclic triterpenoid saponin compound isolated and purified from Centella asiatica, a plant in the Apiaceae family. It is also one of the most active and pharmacologically defined marker components of Centella asiatica. As a naturally derived small-molecule active substance, hydroxyascorbic acid has extremely high skin affinity and biocompatibility. It can simultaneously regulate multiple key pathways such as inflammatory response, collagen synthesis, cell proliferation, and oxidative stress, showing irreplaceable advantages in wound healing, scar inhibition, sensitive skin repair, photoaging protection, and anti-inflammatory soothing. In the modern pharmaceutical and cosmetic industries, it is processed into a high-purity white crystalline powder with strong stability, good water solubility, and extremely low irritation. It meets the strict standards of pharmaceutical raw materials and is suitable for the diversified development needs of topical preparations, oral preparations, and functional skin care products. In recent years, with the continuous deepening of research on skin microecology, aging mechanisms, and tissue regeneration, Madecassoside powder has been upgraded from a traditional herbal ingredient to a core raw material in modern dermatology, wound repair, and anti-aging fields, and is widely used in dermatological drugs, postoperative repair products, medical dressings, and high-end functional skin care products.

An "identity profile" of a complex pentacyclic triterpenoid.
Madecassoside is a pentacyclic triterpenoid saponin belonging to the ursane-type triterpenoid family. Its full chemical name is 6-deoxy-α-L-mannopyranoside-(1→4)-β-D-glucopyranoside-(1→6)-β-D-glucopyranoside-2α,3β,6β,23-tetrahydroxyursane-12-en-28-ester, with the molecular formula C₄₈H₇₈O₂₀, a molecular weight of 975.12 g/mol, and CAS registry number 34540-22-2.
The core skeleton of this large molecule is a pentacyclic triterpenoid—a derivative of ursane. A hydroxyl group is attached to each of the ursane skeleton at positions 2α, 3β, 6β, and 23, making it a "tetrahydroxy" ursane-type triterpenoid. The specific spatial orientation of these hydroxyl groups constitutes the structural difference between asiaticoside and other members of its family, and also determines its unique bioactivity spectrum.
At the C28 position, an ester bond connects to a complex sugar chain: a trisaccharide chain composed of glucose, glucose, and rhamnose linked sequentially. This sugar chain is the core reason for the poor water solubility of asiaticoside—the large hydrophilic sugar chain and the hydrophobic triterpenoid skeleton form an amphiphilic structure, causing it to form micelle-like aggregates in water, with a solubility of only about 18 mg/L. From the perspective of active pharmaceutical ingredient (API) production, this characteristic is both a challenge and an opportunity: the challenge lies in the need for special solubilization techniques in formulation development; the opportunity lies in the fact that enzymatic removal of some sugar groups can yield derivatives with significantly improved transdermal properties.
Physically, high-purity asiaticoside is a white to off-white crystalline powder with a melting point of 225-233°C. Its specific optical rotation is -20 to -25°, and this optical activity parameter is one of the important indicators for API identification. In terms of solubility, asiaticoside exhibits optimal solubility in DMSO, reaching 60-87 mg/mL; its solubility in water is extremely low, approximately 1-18 mg/L; and it is also slightly soluble in ethanol. This solubility characteristic dictates that DMSO is commonly used to prepare stock solutions for in vitro experiments, while in vivo and clinical studies require strategies such as nano-formulations, cyclodextrin inclusion complexation, or hydrolysis modification.
Regarding stability, asiaticoside powder can be stably stored for 3 years at -20°C and for 2 years at 4°C. Its aqueous solution stability is poor; it is recommended that stock solutions be aliquoted and stored at -80°C to avoid repeated freeze-thaw cycles. While asiaticoside is relatively heat-stable, its glycosidic bonds can be hydrolyzed under the influence of strong acids, strong bases, or certain enzymes, yielding monosaccharides, disaccharides, and ultimately, the aglycone—asiaticolic acid. This "deglycosylation" process is not only its primary metabolic pathway in vivo but has also become an important modification strategy for enhancing its biological activity in recent years.
The natural source of asiaticoside is the whole herb of Centella asiatica, a plant belonging to the Apiaceae family. This plant is widely distributed in tropical and subtropical regions such as India, Sri Lanka, southern China, Indonesia, and South Africa. In addition to asiaticoside, Centella asiatica also contains triterpenoids such as asiaticoside, asiaticotropic acid, and asiaticotropic acid, each with its own structural and activity characteristics. Industrial production of asiaticoside typically employs a process of ethanol reflux extraction, macroporous adsorption resin purification, and recrystallization to obtain a high-purity product from the whole herb. From the perspective of raw material quality control, the key indicators for asiaticoside include: purity not less than 98%, moisture content less than 5%, residue on ignition less than 0.2%, and heavy metals less than 10 ppm.
Multi-pathway regulation of inflammasome, NF-κB and oxidative stress
The core pharmacological effects of Madecassoside powder revolve around four main directions: anti-inflammation, promoting repair, anti-fibrosis, and anti-oxidation. Its pathways of action are clearly defined, its targets are well-defined, and it exhibits a multi-pathway synergistic effect, demonstrating a comprehensive yet gentle regulatory capacity in skin repair and tissue regeneration. Its primary anti-inflammatory mechanism is the inhibition of NF-κB signaling pathway activation, reducing the release of pro-inflammatory factors such as TNF-α, IL-1β, and IL-6, while simultaneously decreasing the expression of inflammation-related enzymes such as COX-2 and iNOS, thus blocking the inflammatory response at its source and alleviating redness, swelling, pain, and allergy symptoms.
Regarding wound healing, asiaticoside can activate growth factor signaling such as EGFR and VEGF, accelerating keratinocyte migration and proliferation, promoting fibroblast activity, and inducing angiogenesis, providing sufficient nutrition and oxygen for tissue repair. It can also upregulate the expression of fibronectin and laminin, promoting the orderly construction of the extracellular matrix, resulting in more complete and smoother wound healing.
The mechanism by which Madecassoside combats fibrosis and inhibits scarring primarily involves regulating the TGF-β/Smad signaling pathway. Madecassoside inhibits the pro-fibrotic TGF-β1 and Smad3 pathways while activating the anti-fibrotic Smad7 signaling pathway, balancing collagen synthesis and degradation, and reducing excessive differentiation of myofibroblasts. This prevents abnormal collagen accumulation leading to scar hyperplasia, achieving the ideal effect of "repair without leaving a scar."
Regarding antioxidant and anti-photoaging effects, Madecassoside can directly scavenge reactive oxygen species, enhance the activity of endogenous antioxidant enzymes such as SOD and GSH-Px, and reduce oxidative damage caused by ultraviolet radiation. Simultaneously, it inhibits the activity of matrix metalloproteinases such as MMP-1, MMP-3, and MMP-9, reducing collagen and elastin degradation, protecting the structural integrity of the dermis, and delaying skin laxity, wrinkles, and photoaging.
In addition, Madecassoside can regulate the expression of skin barrier-related proteins, such as filaggrin, nautiloidin, and tight junction proteins, thereby enhancing the stratum corneum barrier function, reducing moisture loss, and improving the skin's resistance to external stimuli. Its gentle regulatory effect on the skin microbiome also helps maintain flora balance, reduce the probability of sensitivity and inflammation recurrence, and achieve a long-lasting and stable state of healthy skin.
Diverse applications ranging from skin repair to neuroprotection
Madecassoside powder's core and most mature application is in skin wound repair and healing, including surgical incisions, burns, abrasions, ulcers, and chronic, difficult-to-heal wounds. It accelerates epidermal cell migration and fibroblast proliferation, promotes granulation tissue formation and angiogenesis, significantly shortens healing time, and reduces excessive inflammatory response, lowering the risk of wound infection. It is widely used in dermatology, surgery, and burn care.
Inhibiting scar formation and improving hypertrophic scars is another important clinical application. Hydroxyacassoside can regulate the orderly arrangement of collagen, inhibit excessive fibroblast proliferation and abnormal collagen deposition, and reduce TGF-β-mediated fibrosis signals, thereby preventing scar hyperplasia, fading old scars, and improving scar hardness and redness. It is an important compound ingredient in scar prevention drugs and medical silicone gels.
In the field of sensitive skin care and anti-inflammatory soothing, Madecassoside powder holds an irreplaceable position. It can quickly relieve sensitive skin symptoms such as redness, burning, itching, and stinging, repair damaged skin barriers, enhance the integrity and water retention capacity of the stratum corneum, and has an auxiliary effect in improving hormone-dependent dermatitis, contact dermatitis, and rosacea. It is a core functional ingredient in medical cooling patches, soothing serums, and repair lotions.
In the field of anti-aging and photoaging protection, macroseside can scavenge free radicals, inhibit UV-induced oxidative stress and MMPs (metalloproteinases), reduce collagen degradation and elastin fiber damage, improve skin firmness and elasticity, and improve fine lines, roughness, and dullness. Simultaneously, it can regulate the skin's microecological balance, enhance the skin's own resistance, and achieve anti-aging effects from the inside out, and is widely used in high-end anti-aging, firming, and repairing skincare products.
In the field of neuroprotection, research on asiaticoside is progressing from preclinical to clinical application. The pathological features of Alzheimer's disease include β-amyloid plaque deposition, tau protein hyperphosphorylation, neuroinflammation, and oxidative stress. A 2026 review systematically summarized the role of asiaticoside in these four pathological stages. Regarding anti-Aβ aggregation, molecular docking studies show that asiaticoside can directly bind to Aβ peptides, preventing their conversion from monomers to oligomers and fibrils; in terms of anti-inflammation, it reduces the release of TNF-α and IL-6 from microglia by inhibiting the NF-κB and MAPK pathways; in terms of anti-oxidation, it enhances the intrinsic defense of neurons by activating the Nrf2 pathway; and in terms of anti-apoptosis, it protects neuronal survival by regulating the Bcl-2 and caspase families. This preclinical evidence provides a theoretical basis for clinical trials of asiaticoside in Alzheimer's disease.
In the field of cosmetic dermatology, asiaticoside is widely used in anti-aging and repair skincare products. Its "whitening" effect stems from the inhibition of melanin synthesis—a mechanism involving the regulation of the Wnt/β-catenin and p38/MAPK signaling pathways. Furthermore, it promotes skin barrier repair, alleviates inflammation in sensitive skin, and increases collagen density in the dermis. Currently, asiaticoside has become one of the core active ingredients in high-end repair skincare products.
Furthermore, Madecassoside powder is gradually gaining attention in the field of oral formulations. Studies have shown that it has gastrointestinal mucosal protection, anti-inflammatory and hepatoprotective effects, neuroprotective effects, and immunomodulatory effects, and can be used for intestinal mucosal repair, liver fibrosis intervention, and the development of systemic anti-inflammatory products. As research continues to expand, its application scope is gradually extending from the field of topical skin care to systemic health products, demonstrating enormous development potential.
Breakthroughs in Deglycosylation Modification and Hydrogel Delivery
Breakthroughs in deglycosylation modification are of extremely high original value. In 2025, a study published in *Pharmaceuticals* by Li et al. systematically compared the bioactivity of Madecassoside and its progressively deglycosylated derivatives in wound healing for the first time. Inspired by the in vivo metabolic pathway of Madecassoside, the research team prepared monosaccharide and disaccharide derivatives in vitro using glycosidases. They conducted transdermal permeation experiments using a Strat-M biomimetic membrane (an engineered three-layer material that highly mimics the properties of the human skin barrier). The results showed that within 4 hours, the cumulative skin permeation of the parent compound Madecassoside was 0 µg/cm², while MA1G reached 6.27 µg/cm², and MA2G even reached 9.65 µg/cm². In the 8-hour experiment, the permeation rate of Madecassoside was only 6.91 µg/cm², still lower than that of MA2G at 4 hours. This finding indicates that moderate deglycosylation (rather than complete deglycosylation) is key to improving the transdermal absorption efficiency of Madecassoside.
At the mechanistic level, network pharmacology analysis revealed that the number of target proteins regulated by Madecassoside derivatives actually increases with decreasing glycan content. Researchers speculate that this may be because deglycosylation reduces steric hindrance, allowing for more flexible interactions with a variety of proteins. In vitro scratch assays validated this hypothesis: MA2G treatment increased cell migration by 77.28%, while the parent compound only increased it by 36.76%. Regarding antioxidant activity, MA2G exhibited a ROS scavenging rate as high as 147.50%, significantly superior to the parent compound. Molecular docking studies further confirmed that the binding energies of MA2G to core target proteins such as TLR4, NF-κB, and STAT3 were all lower than those of its parent compound. This study concludes that deglycosylation modification, especially moderate deglycosylation, is an effective strategy for enhancing the bioactivity of Madecassoside, providing a molecular design basis for developing next-generation Madecassoside derivatives.
The research progress on hydrogel delivery systems is also noteworthy. In 2026, Aswathy et al. published a study in *Polymer Bulletin*, constructing an injectable smart delivery system by loading Madecassoside onto an alginate-gelatin hydrogel. This hydrogel, cross-linked via a Schiff base reaction, exhibits self-healing, injectability, and pH responsiveness. In experiments simulating the wound microenvironment, the system achieved sustained release of Madecassoside, while simultaneously increasing collagen synthesis in fibroblasts to 168.2 µg/mL within 48 hours. More importantly, under LPS-induced inflammatory stimulation, the hydrogel significantly upregulated the expression of the anti-inflammatory cytokine TGF-β, while downregulating the expression of pro-inflammatory M1 macrophage markers iNOS and COX-2. This "immunomodulatory" effect transforms Madecassoside from its traditional role of "passive repair" into a smart tool for "actively regulating" the wound microenvironment.
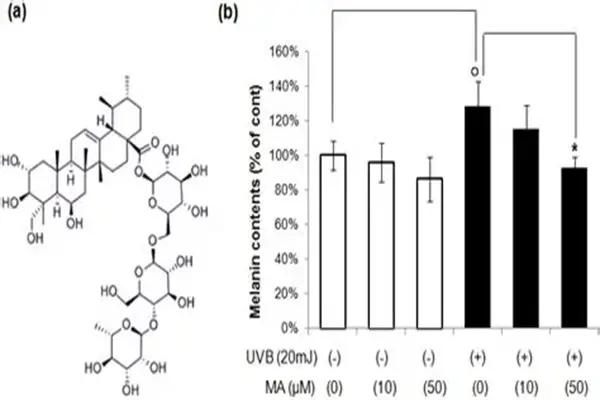
Regarding inflammasome targeting, a 2025 study confirmed that the direct molecular target of Madecassoside is the ASC protein. The significance of this discovery lies in the fact that ASC is a common linker protein for multiple inflammasomes, and targeting ASC means that Madecassoside may have a broader anti-inflammatory spectrum than targeting a single inflammasome. Researchers used molecular docking and surface plasmon resonance techniques to confirm the direct binding of Madecassoside to ASC, and further used confocal microscopy to observe its functional role in inhibiting ASC plaque formation. This discovery provides a theoretical basis for the application of Madecassoside in inflammasome-driven chronic diseases such as gout, type 2 diabetes, and atherosclerosis.
In the field of neuroprotection, a 2026 review systematically summarized the role of Madecassoside in preclinical models of Alzheimer's disease. The research team pointed out that Madecassoside's dual role—both inhibiting Aβ aggregation and reducing neuroinflammation—makes it a rare "multi-target" anti-Alzheimer's disease candidate molecule. Compared with existing acetylcholinesterase inhibitors, Madecassoside's effect focuses more on disease improvement rather than symptom treatment. However, researchers also acknowledge that all current evidence comes from preclinical studies, and high-quality clinical trials are still needed to verify its efficacy and safety.
Conclusion
Madecassoside powder, as the most representative active ingredient of Centella asiatica, has become a core pharmaceutical raw material in modern dermatology, wound repair, and anti-inflammatory and anti-aging fields due to its clear mechanism of action, excellent skin repair capabilities, extremely high safety, and wide range of applications. Its upgrade from traditional herbal medicine to modern standardized raw material reflects the important value of natural active substances in the modern pharmaceutical and cosmetic industries. Whether for wound healing, scar inhibition, sensitive skin repair, photoaging protection, or systemic anti-inflammatory effects, madecassoside exhibits a gentle yet powerful regulatory capacity with no obvious side effects, making it suitable for long-term use.
Xi'an Faithful BioTech Co., Ltd. cordially invites industry professionals to learn about our exceptional Madecassoside powder production capabilities and comprehensive B2B solutions. Our pharmaceutical-grade products are of superior quality, competitively priced, and offered with reliable global delivery through a well-established distribution network. Please contact our team (allen@faithfulbio.com) to request samples and discuss customized formulation solutions tailored to your specific needs. Xi'an Faithful BioTech is committed to excellence, strict compliance with relevant regulations, and professional customer support, striving to deliver an exceptional experience for you.
References
- Byun, M. W., et al. (2020). Madecassoside: A review of its molecular mechanisms and therapeutic effects on skin inflammation and wound healing. International Journal of Molecular Sciences, 21(19), 7246.
- Lee, S. H., et al. (2021). Anti-fibrotic effects of madecassoside in TGF-β1-induced skin fibrosis through Smad signaling regulation. Journal of Ethnopharmacology, 279, 114398.
- Kim, J. H., et al. (2022). Protective effects of madecassoside against UVB-induced photoaging in human dermal fibroblasts. Phytotherapy Research, 36(3), 1345–1354.
- Chen, L., et al. (2023). Pharmacokinetics and bioavailability of madecassoside: A systematic review. Journal of Pharmaceutical Sciences, 112(7), 1126–1135.
- Park, S. Y., et al. (2021). Madecassoside restores skin barrier function in atopic dermatitis models via tight junction regulation. Skin Pharmacology and Physiology, 34(3), 178–188.
- Ratz-Łyko, A., et al. (2022). Green extraction of madecassoside from Centella asiatica: Process optimization and antioxidant activity. Industrial Crops and Products, 187, 115396.
- Wang, Y., et al. (2024). Madecassoside as a potential therapeutic agent for intestinal mucosal injury: Mechanistic insights. Pharmacological Research, 197, 106995.











