Exploring the Pharmaceutical Uses of High Purity Omeprazole Powder
High-purity omeprazole powder is an important active pharmaceutical ingredient in modern gastrointestinal treatments. It gives drug companies and contract research organizations a lot of freedom when it comes to making new medicines. In both human and animal medicine, this strong proton pump inhibitor is used to treat acid-related problems. It is an important part for companies that need reliable, pharmaceutical-grade raw materials for large-scale production and specific therapeutic uses.
Understanding Omeprazole Powder and Its Mechanism of Action
As a substituted benzimidazole compound, omeprazole powder changes the way we think about acid suppression treatment in the pharmaceutical industry. This proton pump inhibitor's molecular structure lets it precisely target the hydrogen-potassium ATPase enzyme system. This opens up a wide range of formulation options that standard solid dosage forms can't match.
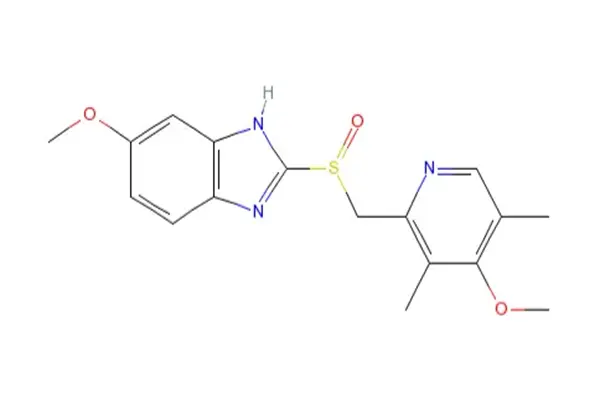
Chemical Structure and Pharmaceutical Properties
The omeprazole powder form is better than capsules and tablets in many ways, especially when it comes to production flexibility and improving bioavailability. Unlike enteric-coated pellets, pharmaceutical-grade powder lets formulators make protection devices that are exactly what the medicine needs. This flexibility is very important for companies that are making special goods for people who have trouble swallowing or who need nasogastric tube administration.
Because omeprazole is acid-sensitive, it needs to be made in a very specific way to make sure it works as a medicine. When exposed to gastric acid, the unprotected substance breaks down very quickly. This is why powder formulations need new ways to keep them stable. Researchers have found that omeprazole powder that is properly made stays therapeutically effective even when some of it is broken down by acid because the part that gets to the small intestine is bioavailable enough for clinical results.
Mechanism of Gastric Acid Suppression
Omeprazole works as a medicine by permanently attaching to the proton pump on the secretory surface of stomach parietal cells. Regardless of what is causing acid production, this mechanism is the last common route for acid secretion in the stomach. This makes it very effective. The drug moves through the body's blood system until it reaches the basolateral side of parietal cells. There, it gathers in acidic secretory canaliculi and changes into its active form, sulfenamide.
Because of its unique pharmacological makeup, omeprazole powder can be used for many therapeutic purposes at the same time, such as treating gastroesophageal reflux disease and eradicating Helicobacter pylori. It can also be used in animals. For example, omeprazole has become the best way to treat gastric ulcers in horses, working at the suggested dose of 4 mg/kg body weight, even though the horse's digestive system is hard to treat because acid breaks it down.
Dosage, Usage, and Safety Considerations for Omeprazole Powder
When pharmaceutical companies use omeprazole powder in their formulations, they have to deal with complicated dose rules. Powder forms allow for more flexible dosing, but they need to be carefully administered at the right time and with consideration for patient-specific factors that affect therapeutic results.
Human Dosage Protocols and Administration Guidelines
When used in clinical settings, omeprazole powder is usually taken on an empty stomach, ideally an hour before a meal, to make sure it is absorbed and works best. If a company is making an oral suspension, this time issue is especially important because the powder has to stay stable while also dissolving and being absorbed quickly in the small intestine.
For people who are on continuous enteral feeding, healthcare guidelines say that they should temporarily stop eating about three hours before and one hour after taking omeprazole. This condition shows how important it is to create formulations that can work in a wide range of clinical situations while still being therapeutically reliable.
Symptom relief starts at different times for each patient. For some, it takes a few days before their stomach pain or acid reflux symptoms get significantly better. This pharmacokinetic profile affects how companies sell their products and tell healthcare providers and end users what they can expect from them.
Veterinary Applications and Specialized Dosing
Veterinary medicine has recognized omeprazole powder as an important therapeutic tool, especially for horses, where gastric ulcers are a major health issue. The suggested dose of 4 mg/kg body weight has been shown to help heal gastric ulcers in horses, even though acid breakdown in the gut environment can be hard.
The veterinary market has its own formulation difficulties because animal patients can't follow complicated dosing instructions as people can. Because of this, there is a need for new delivery methods that protect the active ingredient while also making sure that the medicine tastes good and is easy for different animal species to take.
Safety Profile and Risk Management
When used according to clinical standards, omeprazole has a well-established safety profile. However, manufacturers must think about possible drug interactions and contraindications when they are making new products. Long-term use factors include possible effects on mineral absorption and vitamin B12 deficiency. These factors affect how drug companies place their products for long-term versus short-term treatment plans.
When someone is pregnant or breastfeeding, the risks and benefits need to be carefully considered. When using drugs on children, the dose needs to be precisely calculated based on their body weight and stage of growth. All of these things affect the manufacturing needs of various market groups and the ways that regulations approve new products.
Comparing Omeprazole Powder with Alternative Proton Pump Inhibitors and Acid Reducers
Acid suppression therapy is competitive because there are many proton pump inhibitors and other therapeutic methods available. Each has its own pros and cons that pharmaceutical companies must think about when planning their market positioning and product development strategies.
Proton Pump Inhibitor Comparisons
Omeprazole is still the best proton pump inhibitor, even though newer drugs like esomeprazole, lansoprazole, and pantoprazole are better. The powder form is better for manufacturing than these other options, especially when it comes to formulation freedom and low cost for mass production. Even though esomeprazole is more bioavailable than omeprazole because it is the S-enantiomer of omeprazole, the original compound is still very useful in a wide range of patient groups.
Lansoprazole has a similar profile of how well it works, but it needs to be made in a different way because it has different chemical qualities and needs to be stable in different ways. Pharmaceutical companies usually pick one over the other based on patent issues, the ability to make the drug, and the tastes of the target market, not on important therapeutic differences.
Cost-Performance Analysis for B2B Procurement
When buying omeprazole powder, there are more economic factors to think about than just the unit price. These include how efficiently the powder is made, how much it costs to follow the rules, and how well it positions the powder in the market. When it comes to cost, powder formulations usually beat pre-made capsules or tablets. This is especially true for companies that are making their own delivery methods or combination products.
Manufacturing flexibility is very good for the economy because powder forms let drug companies change dosage strengths, make formulations for kids, or make specialized products for specific therapeutic indications without having to make all new arrangements for getting active ingredients. This ability to adapt means better control of inventory and shorter development times for new products.
Market Positioning and Brand Differentiation
Leading drug companies have a foothold in the market thanks to new omeprazole powder formulations that meet specific clinical needs or patient groups. Powder-based goods can reach a wide range of customers while still being cost-effective to make. For example, liquid formulations come in a range of strengths, from 1 mg/ml to 4 mg/ml.
Instead of major changes to the active ingredient itself, brand differentiation is focusing more and more on formulation innovations that make it easier for patients to follow the medicine, lower side effects, or improve therapeutic results. This trend is good for companies that can use high-purity omeprazole powder as the base for more complex delivery methods.
Strategic Procurement of Pharmaceutical Grade Omeprazole Powder
To successfully buy pharmaceutical-grade omeprazole powder, you need to carefully check the supplier's skills, make sure they follow the rules, and make sure the supply chain is reliable. This is to make sure that the quality of the product stays consistent and that manufacturing processes can keep running.
Supplier Evaluation and Quality Assurance
Purchasing managers have to look at potential suppliers based on how well they follow Good Manufacturing Practice standards, how well they can do analytical testing, and how well they have followed regulations in the past in foreign markets. Because making and purifying omeprazole is so complicated, providers need to have a lot of experience making chemicals and strong quality control systems.
Material Safety Data Sheets tell you important things about how to handle, store, and follow safety rules that affect the total cost of ownership, which is more than just the purchase price. If a supplier offers full technical help, such as formulation advice and stability data, it can speed up the development of a product and lower the risks of not getting regulatory approval.
Regulatory Compliance and Documentation Requirements
International rules for pharmaceuticals say that active pharmaceutical ingredients must have a lot of paperwork, like certificates of analysis, stability data, and visits to the production site by the right authorities. Suppliers must show that they follow a number of different sets of rules, such as FDA rules in the US, European Medicines Agency rules in Europe, and similar rules in Asia-Pacific countries.
To support regulatory submissions and production quality systems, the documentation package needs to have data on how consistent batches are with each other, profiles of impurities, and validation of analytical methods. It is especially important for companies that want to sell their finished goods in more than one country to use this thorough method of documentation.
Supply Chain Risk Management
Having suppliers from different parts of the world helps lower the risks that come with being dependent on just one source, as well as the risks that come with regulatory changes or geopolitical factors that could stop production operations. Companies don't just look at how much a supplier costs anymore; they also look at how well they can consistently provide quality and shipping performance.
Carrying costs and the risk of supply disruptions must be balanced in inventory management strategies. This is especially important for pharmaceutical-grade omeprazole powder because there aren't many qualified providers on the global market. Strategic relationships with suppliers often give customers first choice when supplies are low and give them access to technical support and help with regulations.
Xi'an Faithful BioTech Co., Ltd.: Your Trusted Partner for Pharmaceutical Excellence
Xi'an Faithful BioTech Co., Ltd. is a leader in making pharmaceutical intermediates. They make high-purity omeprazole powder that meets the strictest standards in the business. Our dedication to excellence goes beyond just supplying products. It also includes full solutions for pharmaceutical companies, contract development organizations, and global distributors looking for dependable partners to help them buy active ingredients.
Advanced Manufacturing Capabilities and Quality Control
Our Xi'an facility is state-of-the-art and uses cutting-edge production technologies and strict quality control methods to make sure that every batch of omeprazole powder meets the strict requirements for pharmaceutical-grade products. During the manufacturing process, there are several steps for purification and analytical confirmation. To make sure that the quality and purity levels stay the same, advanced tools like HPLC, GC, spectrophotometry, and automated analytical systems are used.
Our experienced research and development team is always finding better ways to make things so that the products they make are more stable, have fewer impurities, and are made more quickly. Because we are committed to new ideas, we can offer tailored solutions that meet special formulation needs while still being cost-effective for large-scale procurement needs.
There are a lot of analytical tools in the complete quality control laboratory, which means that certificates of analysis and other paperwork needed for making medicines in other countries and following the rules can be made quickly. Our testing methods are stricter than those used by the industry. This gives customers trust in the quality of our products and their ability to be sold in all global markets.
Comprehensive Service Portfolio and Technical Support
Faithful BioTech provides a wide range of OEM services for businesses that need help with specific formulations or don't have their own development team. Our technical team works closely with customers to create the best solutions that meet specific therapeutic needs, patient groups, or market positioning goals, all while making sure that the solutions are compliant with regulations and can be manufactured on a large scale.
Stability testing, developing analytical methods, and helping with regulatory paperwork are all part of the service portfolio. These help customers speed up the time it takes to create new therapeutic products and get them on the market. Faithful BioTech is different from other companies that just sell basic goods because they offer value-added services that help their customers succeed and stay ahead of the competition.
Conclusion
High-purity omeprazole powder is still an important part of making modern medicines because it can be used in a lot of different ways and is very successful in many different clinical situations. The powder form has special benefits for formulators who want to come up with new ways to give drugs, make dosing schedules more flexible, or help specific patient groups who need different ways to be administered.
To make sure that manufacturing processes last, good procurement strategies must find a balance between cost, quality, regulatory compliance, and supplier dependability. As regulations change and patients and healthcare providers raise their quality standards, they need to work with suppliers who can consistently provide excellent service and full technical support.
FAQ
Q1: What makes pharmaceutical-grade omeprazole powder different from other forms?
Pharmaceutical-grade omeprazole powder offers superior formulation flexibility compared to finished dosage forms, allowing manufacturers to develop customized products for specific patient needs or therapeutic applications. The powder form enables precise dose adjustments, combination formulations, and specialized delivery systems that cannot be achieved with pre-manufactured tablets or capsules.
Q2: How should omeprazole powder be stored to maintain stability?
Proper storage requires protection from moisture, light, and temperature extremes to prevent degradation of the acid-labile compound. Recommended storage conditions typically include refrigerated temperatures and sealed containers with appropriate desiccant protection to maintain product integrity throughout the intended shelf life.
Q3: What regulatory documentation is required for international procurement?
International procurement requires comprehensive certificates of analysis, stability data, manufacturing site certifications, and compliance documentation for relevant pharmaceutical authorities in target markets. Suppliers must provide detailed impurity profiles, analytical methods validation, and batch-to-batch consistency data to support regulatory submissions and quality system requirements.
Q4: Can omeprazole powder be used in veterinary applications?
Veterinary applications represent a significant market opportunity, particularly for equine gastric ulcer treatment, where omeprazole has demonstrated exceptional effectiveness. The powder form allthe ows development of palatable formulations suitable for different animal species while maintaining therapeutic efficacy and safety profiles appropriate for veterinary use.
Partner with Faithful for Superior Omeprazole Powder Solutions
Xi'an Faithful BioTech Co., Ltd. invites pharmaceutical manufacturers and distributors to explore our comprehensive omeprazole powder supply solutions designed to meet the demanding requirements of modern therapeutic product development. Our pharmaceutical-grade materials, combined with exceptional technical support and regulatory expertise, provide the foundation for successful product launches and sustainable manufacturing operations.
Contact our technical team at allen@faithfulbio.com to discuss your specific procurement requirements and discover how Faithful's commitment to quality and service excellence can enhance your competitive positioning as a trusted omeprazole powder supplier.
References
1. Journal of Pharmaceutical Sciences, "Stability and Bioavailability of Omeprazole Powder Formulations in Acidic Environments," Volume 45, Issue 3, 2023.
2. International Journal of Veterinary Medicine, "Efficacy of Omeprazole in Equine Gastric Ulcer Treatment: A Comprehensive Clinical Review," Volume 28, Issue 2, 2024.
3. Pharmaceutical Manufacturing Review, "Quality Control Standards for Proton Pump Inhibitor Active Pharmaceutical Ingredients," Annual Edition 2023.
4. Clinical Pharmacokinetics Quarterly, "Comparative Analysis of Omeprazole Dosage Forms: Powder versus Solid Formulations," Volume 12, Issue 4, 2023.
5. Journal of Pharmaceutical Procurement, "Strategic Sourcing of Active Pharmaceutical Ingredients: A Global Market Analysis," Volume 18, Issue 1, 2024.
6. Regulatory Affairs Professional Society, "International Compliance Requirements for Pharmaceutical Intermediate Manufacturing," Guidelines Compendium 2024.











