99% Rifampicin API: Chemical Password and Future Journey of Anti tuberculosis' Golden Warrior '
In the long history of anti infective drugs, few molecules have been able to occupy the throne of "first-line treatment" like Rifampicin for more than half a century. Since its appearance in 1965, this striking orange powder has become the most powerful weapon against tuberculosis, the "white plague". It is both a natural gift captured from soil microorganisms and a semi synthetic masterpiece of human chemical intelligence. However, with the wave of drug resistance sweeping through, has this' veteran 'shown signs of fatigue? This article will delve into the molecular heart of 99% Rifampicin API from the perspective of pharmaceutical raw material experts, deconstructing its chemical structure, combat principles, clinical roles, and exploring its "second spring" in the latest scientific research.
Molecular Structure - The Complex Geometry Under That Orange Red Stain
People who first encounter the 99% Rifampicin API are often drawn to its unique orange red color. But this flamboyance is not meant to be pleasing to the eye, but rather a visual representation of its complex chemical structure. As pharmaceutical raw material experts, when we examine this molecule under a microscope and in spectra, we see a carefully crafted and structurally precise 'chemical key'.
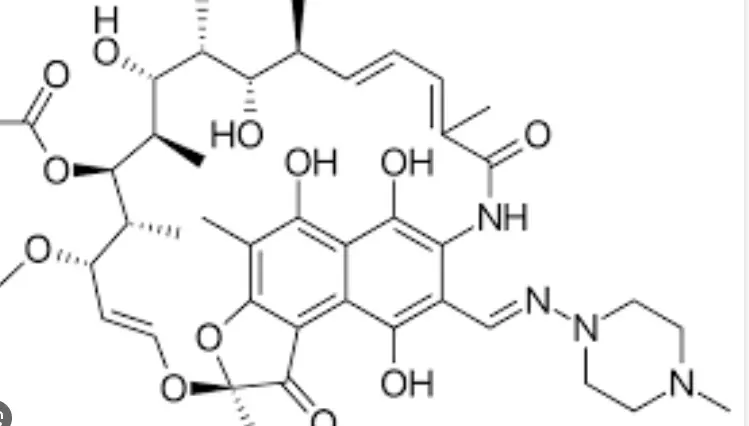
Rifampicin is a semi synthetic derivative of the rifampicin family, with its parent nucleus derived from the fermentation product of Amycolatopsis rifamycinica in the Mediterranean. From the molecular formula, it can be seen that this is a large molecule with 59 heavy atoms and a molecular weight of up to 822.94 Da. In the eyes of chemists, its structure can be considered a work of art: an aromatic core composed of hydrogenated naphthoquinone and hydrogenated naphthoquinone forms a chromophore, which is the optical code that gives it an orange red color; A long and flexible fat bridge chain wraps around the core like an arm, forming a three-dimensional "pocket" structure.
The most critical chemical modification of Rifampicin is the piperazine imine methyl side chain at position 3, which is the key difference between Rifampicin and other rifampicins. This nitrogen-containing alkaline side chain not only greatly improves the lipid solubility of the drug, allowing it to easily cross the complex cell wall barrier of bacteria, but also serves as a "hook" for its tight binding with bacterial targets. In the solid state, Rifampicin exists in multiple crystal forms (such as anhydrous type I, type II, etc.), and the subtle differences in its crystal structure parameters such as space group (usually C121) and unit cell parameters (a, b, c-axis) directly affect the dissolution rate, stability, and bioavailability of the active pharmaceutical ingredient. For example, Ibiapino et al. provided a detailed analysis of the crystal structure of anhydrous Rifampicin type I and its molecular stacking pattern in the monoclinic crystal system, explaining why this crystal form exhibits different physical and chemical properties at specific temperatures and humidity.
From the perspective of stereochemistry, this molecule has 9 chiral centers, and its absolute configuration is crucial for its biological activity. Any reversal of the chiral center may result in the 'key' not being able to open the correct 'lock'. Therefore, when we talk about the 99% Rifampicin API, we are not only discussing a chemical substance, but also a molecular entity with precise three-dimensional arrangement that has undergone billions of years of evolutionary selection and artificial fine modification.
Use -- from the cornerstone of tuberculosis treatment to versatile
In medical microbiology textbooks, the name Rifampicin is always closely associated with "first-line anti tuberculosis drugs". But as the ultimate destination of active pharmaceutical ingredients, its usage scope is much broader than this. From deadly mycobacteria to stubborn gram-positive cocci, to common pathogens in ophthalmic clinics, 99% Rifampicin API plays multiple protective roles after formulation processing.
Its core use is undoubtedly the treatment of tuberculosis. According to the professional information in Merck's diagnosis and treatment manual, Rifampicin has strong antibacterial activity against the vast majority of Gram positive bacteria and some Gram negative bacteria. In standard short course chemotherapy regimens for tuberculosis (such as the 2HRZE/4HR regimen), Rifampicin (R) works alongside isoniazid (H), pyrazinamide (Z), and ethambutol (E) as the core force for sterilization and sterility. Its unique value lies not only in its ability to kill rapidly proliferating tuberculosis bacteria, but also in its ability to target the "stubborn" microbiota with low metabolism, effectively preventing disease recurrence and shortening the treatment cycle from the past 18-24 months to 6-9 months.
However, Rifampicin never goes it alone. Due to the high susceptibility of bacteria to developing drug resistance, its clinical application strictly follows the iron law of "combination therapy". In addition to tuberculosis, this joint strategy has been widely applied to:
Non tuberculous mycobacterial infection: such as lung disease caused by Mycobacterium avium complex (MAC) in bird cells.
Leprosy: Used in combination with drugs such as dapsone, it is a key component of leprosy combined chemotherapy (MDT).
Antibiotic resistant Staphylococcus aureus infection: For difficult to treat Staphylococcus aureus infections such as artificial joint infections, endocarditis, osteomyelitis, etc., Rifampicin is often used in combination with vancomycin or other anti staphylococcal drugs due to its ability to penetrate biofilms and kill intracellular bacteria.
Chemical prophylaxis: Rifampicin alone can effectively eliminate the colonization of Neisseria meningitidis or Haemophilus influenzae in the nasopharynx, and is used to prevent the onset of disease in close contacts.
Ophthalmic local medication: Rifampicin is used in eye drops to treat trachoma, conjunctivitis, and keratitis, demonstrating its value in the field of local anti infection.
At the formulation level, 99% of Rifampicin API is processed into various dosage forms such as capsules, tablets, injections, eye drops, etc. However, its chemical properties are active, sensitive to light, heat, and moisture, and there are issues with its polymorphic form, which places extremely high demands on the formulation process. For example, when used in combination with aminosalicylic acid (PAS), there is a physical interaction between the two that can affect absorption, so it is clinically recommended to take medication at intervals. As raw material experts, we are well aware that after shaping this "orange red" into medicine, it is not only necessary to ensure its chemical purity, but also to ensure that it can be released and dissolved in the correct posture in the complex internal environment, and ultimately reach the battlefield.
Working principle - Sniper bacteria's' transcription factory '
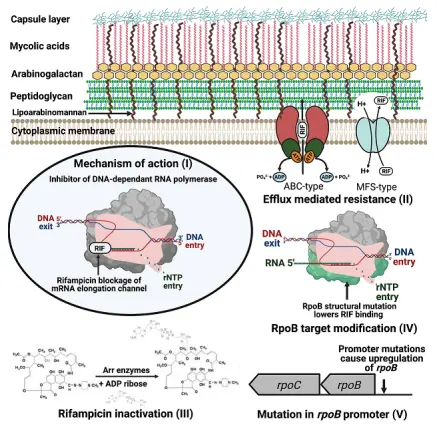
Why has Rifampicin become the "ace" in the fight against tuberculosis? The answer is hidden in a microscopic 'molecular factory' - the RNA polymerase (RNAP) of bacteria. Understanding this intricate targeting mechanism is the cornerstone of our evaluation and improvement of drugs.
The mechanism of action of Rifampicin can be regarded as a model of "targeted sniping". It belongs to bactericidal antibiotics, and its target is the core machine that bacteria rely on for survival - DNA dependent RNA polymerase. This enzyme is responsible for reading genetic information from DNA, synthesizing messenger RNA (mRNA), and initiating protein production. Without RNA, bacterial life activities would come to a standstill.
In 2001, Campbell et al. published a landmark study in the journal Cell, revealing for the first time at the atomic level how Rifampicin "locks" the enzyme through crystal structure analysis. They found that Rifampicin molecules do not directly attack the active center of the enzyme, but bind to a deeply buried, hydrophobic 'pocket' on the RNA polymerase beta subunit. This binding point is located deep within the DNA-RNA channel, approximately 12 angstroms away from the active center of the enzyme. When
Rifampicin is embedded, it physically blocks the way out of the extending RNA strand. Specifically, when the RNA strand is synthesized to the length of the 2nd to 3rd nucleotide, it will experience steric hindrance conflicts with the Rifampicin molecule bound here, leading to premature termination of transcription. It's like on a production line, just two steps away from the product, a huge rock blocks the way, and all subsequent processes cannot proceed.
It is precisely this clever mechanism of "one man holding the key, ten thousand men unable to open" that endows Rifampicin with powerful bactericidal activity. In addition, Rifampicin also has excellent pharmacokinetic properties: it is well absorbed orally (with a bioavailability of up to 90% -95%), has strong tissue penetration, and is particularly easy to accumulate in polymorphonuclear granulocytes and macrophages, which enables it to kill Mycobacterium tuberculosis that "hides" inside cells.
However, strong selection pressure inevitably leads to drug resistance. The counterattack of bacteria also occurs on RNA polymerase. The most common drug-resistant mutation is a point mutation in the rpoB gene encoding the RNA polymerase beta subunit, resulting in amino acid changes at the Rifampicin binding site (such as the most common mutation β S450L), making it difficult for the drug to effectively "lock" onto the target. This is also why Rifampicin can never be used alone - monotherapy almost inevitably leads to the rapid emergence of drug resistance.
Latest research direction - New transmission of old medicine and "using the spear of the child to attack the shield of the child"
Facing the increasingly severe challenge of drug-resistant tuberculosis (MDR-TB), this veteran who has served for more than half a century has not withdrawn from the historical stage. On the contrary, the latest scientific research is giving Rifampicin a whole new vitality. At the end of 2025, a groundbreaking study from Rockefeller University showed us the infinite possibilities of "old medicine new transmission".
This study, published in Nature Microbiology, proposes a disruptive strategy called 'vertical inhibition'. Traditional combination therapy typically targets different pathways of bacteria, such as cell walls and protein synthesis, but scientists at Rockefeller University have taken the opposite approach: they have decided to set up two consecutive obstacles on the same pathway. Specifically, they found a compound codenamed AAP-SO ₂, which also targets RNA polymerase but has a completely different binding site from Rifampicin. Its function is to specifically inhibit the transcription elongation stage (RNA strand elongation process).
The experimental design is exquisite: although the most common Rifampicin resistant mutation (β S450L) in tuberculosis bacteria allows the bacteria to escape the attack of Rifampicin, it comes with a "cost" - this mutation causes RNA polymerase to become abnormally slow and clumsy during the transcription extension stage. At this point, if AAP-SO ₂ is added, it is equivalent to adding another blow to the already struggling extension pathway of the bacteria, completely pushing them into a dead end.
The research results are exciting:
Synergistic effect: In a rabbit model simulating the dormancy and aggregation state of tuberculosis, the combination of AAP-SO ₂ and Rifampicin showed a strong synergistic effect, which could increase the efficacy of Rifampicin by 30 times.
Containing drug resistance: AAP-SO ₂ can effectively eliminate strains carrying drug-resistant mutations, making it difficult for drug-resistant mutations to establish themselves in the microbiota and even being eliminated in reverse.
Attacking dormant bacteria: this combination can effectively kill those dormant bacteria hiding in granuloma that are difficult to reach with conventional drugs, and directly point to the pain point of tuberculosis that is difficult to cure.
At the same time, for the treatment of Rifampicin resistant tuberculosis (RR-TB), the clinical community is also actively exploring a safer and more effective program. A randomized controlled study published in Dovepress in 2025 compared the safety and tolerability of the novel Oxadixyl drug Contazolamide with Linezolid in the short-term treatment of Rifampicin resistant tuberculosis. The research results showed that in the background regimen containing TMC-207, Levofloxacin, and other drugs, the incidence of adverse events in the Contazolamide group (14.3%) was much lower than that in the Linezolid group (92.3%), especially the common bone marrow suppression and peripheral neuropathy in the Linezolid group, which did not occur in the Contrazolamide group.
This suggests that even in the field of Rifampicin failure, the safety and tolerance of the whole treatment can be improved by optimizing the "supporting role" in the joint scheme.
These cutting-edge studies have pointed us in two directions: one is to strengthen the core position of Rifampicin through new joint strategies such as "vertical inhibition", and even "turn waste into treasure", using drug-resistant mutations themselves as a breakthrough point for attack; The second is to develop safer alternative drugs and optimize combination therapies to address the already developed drug resistance.
Conclusion
As observers standing at the source of drug production, looking back at the story of Rifampicin, we cannot help but feel a myriad of emotions. This complex molecule, coming from the soil, has saved billions of lives with its unique skill of accurately targeting bacterial transcription factories. Its structure is a masterpiece of chemistry, its use is the cornerstone of medicine, and its principles are the poetry of science.
Nowadays, in the face of the increasing pressure of drug-resistant bacteria, Rifampicin has not given up, but has instead shown new vitality under the brilliant pen of scientists. Whether it is to use the "vertical inhibition" strategy to turn drug-resistant mutations into fatal weaknesses, or to adjust the role in the new combination therapy, it indicates that the story of this "golden warrior" is far from over. As Jeremy Rock from Rockefeller University said, "By gaining a deeper understanding of the molecular mechanisms that occur when bacteria develop drug resistance, we can begin to rationally design clinical response strategies." In the eyes of pharmaceutical raw material experts, Rifampicin is not only an established product, but also a continuously evolving legend, and its future is waiting for more chemical and biological wisdom to continue writing.
Xi'an Faithful BioTech Co., Ltd. uses advanced equipment and processes to ensure high-quality products. We produce high-quality 99% Rifampicin API Powder that meet international drug standards. Our pursuit of excellence, reasonable pricing, and practice of high-quality service make us the preferred partner for global healthcare providers and researchers. If you need to conduct scientific research or production of Rifampicin , please contact our technical team through the following methods sales1@faithfulbio.com.
Reference
1.Sigma-Aldrich. (n.d.). Rifampicin European Pharmacopoeia (EP) Reference Standard. Retrieved from Sigma-Aldrich product page.
2.Wang, J., Xue, Y., Nie, W., Ma, L., Chu, N., & Du, Y. (2025). Safety and tolerability of contezolid versus linezolid in the short-term treatment of rifampicin-resistant pulmonary tuberculosis: A randomized controlled study. Infection and Drug Resistance, *18*, 3307–3315.
3.National Center for Biotechnology Information. (2026). Rifampicin. PubChem Compound Summary for CID 135398735. Retrieved from .
4.Werth, B. J. (2024). Rifamycins. In MSD Manual Professional Edition.
5.Campbell, E. A., Korzheva, N., Mustaev, A., Murakami, K., Nair, S., Goldfarb, A., & Darst, S. A. (2001). Structural mechanism for rifampicin inhibition of bacterial RNA polymerase. Cell, *104*(6), 901–912.
6.The Rockefeller University. (2025, December 12). This new, one-two punch could knock out drug-resistant TB. Rockefeller News.











