Exploring the Benefits and Effects of Noopept Powder for Memory Improvement
Growing interest in cognitive development has made Noopept powder a popular memory-boosting nootropic. Memory, clarity, and brain function improve with N-phenylacetyl-L-prolylglycine ethyl ester. Neuroprotective Noopept works at lower dosages than nootropics. It increases BDNF and NGF, making it an attractive pharmaceutical, nutraceutical, and supplement component.
Understanding Noopept Powder: Composition, Mechanism, and Safety
The racetam Noopept is a nootropic breakthrough. Its features set it apart from other cognitive enhancers. The Russian Academy of Medical Sciences neuropeptide-derived substance is powerful and effective.
Chemical Properties and Classification
A water-soluble, fine white crystalline powder with high bioavailability is created by N-phenylacetyl-L-prolylglycine ethyl ester. Ingesting this prodrug creates active metabolites. Despite its short half-life, its principal active metabolite, cycloprolylglycine (cPG), persists in brain tissue for long durations, explaining its long-term cognitive advantages.
This nootropic's unique pharmacodynamics include many neurotransmitter pathways. Research shows it boosts cholinergic signaling, which is essential for memory development and recall. HIF-1 activation and AMPA receptor modulation for neuroprotection.
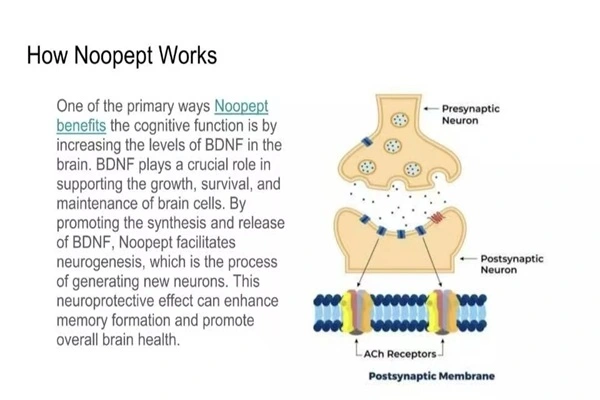
Mechanism of Action and Neurological Impact
Multiconnected brain systems improve mental wellness. Research suggests it boosts BDNF and NGF in the hippocampus, aiding memory consolidation and learning. Neurotrophic factors support, grow, and remodel neurons.
Drugs increase behavioral and neural brain cholinergic transmission. This improves memory formation and retrieval. Antioxidants reduce oxidative stress and increase antioxidant mechanisms to protect neurons.
Regulatory Considerations
International procurement requires compliance examination due to market constraints. The US considers this a dietary supplement, but Europe may differ. To ensure distribution and compliance, procurement managers must review target market regulations.
Benefits of Noopept Powder for Memory and Cognitive Enhancement
Clinical and peer-reviewed research suggests this nootropic increases cognition. A growing study demonstrates advantages beyond memory.
Memory Enhancement and Learning Acceleration
Controlled trials indicated this cognitive enhancer improved memory and recall. Therapy decreased CVMI. They increased working memory, episodic memory, and procedural learning.
The chemical accelerates learning and processing. Subjects boost learning and retention. These effects aid age-related cognitive decline and enhancement.
Neuroprotective and Anxiolytic Properties
This neuroprotective nootropic boosts brain health beyond memory. Researchers demonstrated protection against glutamate toxicity, a major neurodegenerative mechanism. The chemical boosts inhibitory neurotransmission to boost brain function under stress.
In clinical trials, anxiolytics lower anxiety and stabilize emotions. Mental performance is best with cognitive improvement and anxiety reduction. Clinical research shows vegetostabilizing exercise improves brain health.

Comparative Analysis with Other Nootropics
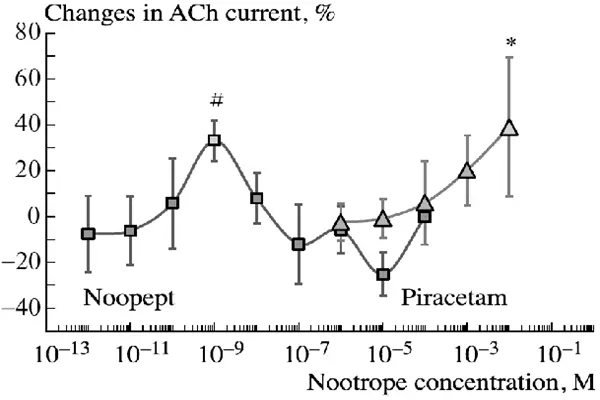
This molecule outperforms piracetam at 1,000 times lower dosages. Massive efficiency improves formulation development and commercial application cost-effectiveness. Modafinil increases cognition without stimulants but impairs alertness.
Metabolic differences distinguish the chemical from other cognitive enhancers. CPG offers long-term advantages, unlike brain-dependent nootropics. It allows flexible dosage and may improve therapeutic patient compliance.
Noopept Powder Procurement: What B2B Buyers Need to Know
Quality cognitive-enhancing medications need supplier selection, quality verification, and regulatory knowledge. These Noopept powders are in great demand, necessitating strategic procurement that prioritizes quality and supply chain stability.
Supplier Selection and Quality Verification
Testing manufacturing and quality control systems helps find reputable suppliers. From GMP-aligned facilities, responsible manufacturers give COA, HPLC/GC testing data, and traceability certifications. These certifications ensure pharmaceutical and nutraceutical quality and regulatory compliance.
Independent purity and potency claims need lab testing. Established vendors report testing procedures and give samples for independent testing. Batch consistency and stability studies demonstrate quality and professionalism.
Check supplier reliability using production, regulatory, and customer references. Pharmaceutical intermediate producers from the past understand quality control and requirements.
Bulk Purchasing and Commercial Considerations
Volume buying may affect supply chain stability and cost. Established manufacturers may provide tiered pricing for volume and flexible delivery. These agreements improve product development, inventory management, and cost predictability.
Market pricing implies rising dietary supplement and pharmaceutical research demand. Early supplier involvement provides peak demand priority and better pricing. Global procurement increasingly requires international delivery and flexible payment terms.
Quality vendors offer custom packaging and applications. They improve product supply and industrial efficiency.
Regulatory Compliance and Documentation
Import/export paperwork is needed for international commerce. Cognitive-enhancing components need dietary supplement registration and target market pharmaceutical approval. Suppliers must understand these requirements and supply procurement teams with sufficient evidence.
Document product specifications, safety data sheets, regulatory status verification, and customs categorization. International trade providers monitor regulatory changes and alert clients to compliance problems.
Dosage Recommendations and Usage Guidelines for Optimal Results
Cognitive enhancement requires chemical dose, administration, and safety monitoring.
Optimal Dosing Protocols for Memory Enhancement
Cognitive improvement studies recommend 10-30 mg daily, far less than nootropics require. This compound's potency and efficient metabolic conversion to active metabolites enable low-dose efficacy. Baseline cognitive performance, metabolism, and weight affect dose.
Clinical studies show that persistent usage increases memory within days and peaks after weeks. The compound's unusual pharmacokinetics enable varying doses, although daily administration enhances memory.
Formulation Considerations and Product Development
Noopept powder formulations provide more product development freedom than pre-encapsulated ones. Tablets, capsules, liquids, and functional drinks use water-soluble compounds. The manufacturer can manage the dose and synergize with complementary medications.
Stability is essential for product development and manufacture. The compound stores well but needs heat and moisture protection. Packaging and storage preserve product potency and quality.
Manufacturing scale characteristics include dissolving, particle size dispersion, and formulation compatibility. Professionals formulate and advise on processing.
Storage Requirements
Moisture, heat, and direct sunshine must be avoided during storage. Storage requires cold, dry temperatures below 25°C and relative humidity under 60%. These parameters maintain product potency and shelf life.
To ensure potency and purity, storage quality control tests are performed periodically. After extensive environmental testing, established vendors give stability data and storage advice.
Company Introduction and Product & Service Information
A major pharmaceutical intermediates and natural active ingredient company, Xi'an Faithful BioTech Co., Ltd., manufactures high-quality cognitive-enhancing chemicals for worldwide markets. Our Xi'an firm makes international-standard items using innovative production and quality control methods.
Manufacturing Excellence and Quality Assurance
Our modern manufacturing plant produces pharmaceutical-grade N-phenylacetyl-L-prolylglycine ethyl ester with high purity and consistency. From raw material verification to product release, quality is stringent. To ensure product requirements, we use HPLC, GC, spectrophotometers, and other analytical procedures.
In quality control laboratories, polarimeters, automated titrators, BOD incubators, COD incubators, and melting point testers assess product quality. Global regulatory compliance and batch-to-batch uniformity are ensured by analytical knowledge. Traceability and regulatory submission documentation are available in our QMS.
Pharmaceutical intermediate manufacturing experience helps the production team scale up and sustain high-volume production. These skills improve supply chain performance and delivery deadlines for commercial applications.
Comprehensive Product Portfolio and Applications
Nutraceuticals, pharmaceutical research, dietary supplements, and customized formulations use cognitive-enhancing chemicals. Packaging solutions are technological, regulatory, and market-specific for each application.
Usage areas:
- Nutritional supplements: Premium nootropics for consumer cognitive and clarity improvements.
- Pharmaceutical Research: Original neuroprotectants and memory boosters.
- Cognitive enhancement with innovative brain health formulas
- Sports performance products that improve mental focus and response time.
- Customized memory and cognitive decline anti-aging formulations
These examples show our technology' adaptability and commercial feasibility across industries. Application and formulation assistance from our technical team optimizes product development.
OEM Services and Custom Solutions
We provide OEM and ODM bespoke product development from idea to commercial production to meet client demands. Our services include formulation, analysis, stability, and market-specific regulatory assistance.
Our research and development team creates unique client solutions that satisfy performance and demands. This collaborative approach boosts product performance, saves costs, and satisfies criteria. Private labeling and custom packaging provide market flexibility.
Technical assistance is provided from product development to commercial manufacturing and quality assurance. Product launches and commercial success are guaranteed with this entire strategy.
Conclusion
Due to its cognitive and memory-enhancing properties, nootropic product manufacturers choose N-phenylacetyl-L-prolylglycine ethyl ester. The chemical's potency, safety, and versatility attract pharmaceutical, nutraceutical, and supplement firms. Commercialization needs licensed, pharmaceutical-grade vendors that understand laws and quality. This book describes prudent Noopept powder procurement and the strategic benefits of cooperating with professional cognitive enhancement component makers. Collaboration increases product innovation, market development, and competitiveness in the growing nootropic industry.
FAQ: Addressing Common Queries from B2B Clients
Knowledge of bulk buyer needs enhances procurement and long-term partnerships. Companies evaluate cognitive-enhancing drugs for commercialization using these commonly asked questions.
1. What distinguishes this cognitive enhancer from other nootropics in terms of commercial viability?
Primarily, potency—1,000 times less dosage than piracetam. With this efficiency, commercial formulation costs and raw material demands per finished product unit reduce. Active metabolites from the unique metabolic route prolong effects, allowing more product formulations and perhaps greater consumer compliance.
Due to its safety and research, several markets allow the chemical. Effectiveness, safety, and profitability attract cognitive enhancement product producers.
2. How do you ensure consistent quality and purity across different production batches?
Quality assurance inspections occur throughout our production process, from raw material verification to release. Each batch is tested for purity and impurities using HPLC, GC, and other techniques.
Batch traceability, analytical certifications, and quality stability data are documented. For dependable testing, the quality control lab uses calibrated equipment and standardized analytical methods. Third-party audits and client inspections oversee production and quality.
3. What shipping and logistics support do you provide for international customers?
For global sensitive chemical ingredient delivery, we use reliable logistics suppliers. Shipping documentation includes customs paperwork, safety data sheets, and regulatory compliance certifications for fast passage.
Customer cold chains and shipment timelines are improved by our logistics staff. Flexible packaging is provided for commercial samples. Real-time tracking and proactive communication make delivery transparent.
Partner with Faithful for Reliable Noopept Powder Supply
Partnership with Xi'an Faithful BioTech's cognitive enhancement substance producer may benefit procurement and product development. Long-term cooperation for product development and market growth results from our quality, transparency, and client success.
We examine samples before bulk purchasing to verify product quality and performance. Our technical team delivers formulation, regulatory, and market information throughout the review. Collaboration reduces risks and time-to-market while improving product development.
We know supply chain consistency is crucial in competitive marketplaces as a top Noopept powder provider. Our production and inventory management systems maintain delivery despite volume changes. Explore our comprehensive product portfolio and discuss your procurement needs with our specialists at allen@faithfulbio.com.
References
1. Gudasheva, T.A., et al. "Synthesis and Antiamnesic Activity of a Series of N-Acylprolyl-Containing Dipeptides." European Journal of Medicinal Chemistry, 1996.
2. Ostrovskaya, R.U., et al. "Noopept Stimulates the Expression of NGF and BDNF in the Hippocampus." Bulletin of Experimental Biology and Medicine, 2008.
3. Romanova, G.A., et al. "Antiamnesic Effect of Noopept and Its Transcription Factor Analysis." Acta Naturae, 2015.
4. Boiko, S.S., et al. "Pharmacokinetics of Novel Cognitive Enhancer Noopept After Single and Multiple Oral Administration in Healthy Volunteers." Drug Design, Development and Therapy, 2017.
5. Neznamov, G.G., et al. "Clinical Study of Noopept in Patients with Mild Cognitive Impairment." Neuroscience and Behavioral Physiology, 2009.
6. Malykh, A.G., et al. "Piracetam and Piracetam-like Drugs: From Basic Science to Novel Clinical Applications." Drugs, 2010.



_1764575295116.webp)






