Fasoracetam: A "master key" or a "directed missile" to unlock cognitive impairment and neurodevelopmental disorders?
The chemical name of Fasoracetam powder is (5R)-5-(piperidin-1-carbonyl)pyrrolidine-2-one, with the molecular formula C₁₀H₁₆N₂O₂ and a molecular weight of 196.25 g/mol. Structurally, it belongs to the classic "racasetam" family. However, Fasoracetam powder has its unique structural features. First, it is a chiral molecule, and the absolute configuration of this chiral center is crucial for its stereospecific binding with biological receptors, which also means that optical purity must be strictly controlled during synthesis.
A game of solid-state form for a "flexible" molecule
From a skeletal perspective, Fasoracetam retains the five-membered lactam ring common to lactams, a structure that forms the basic framework for binding to receptors on nerve cell membranes and determines the basis of the molecule's neuroactive activity. The carbonyl group on the lactam ring forms a hydrogen bond donor with the nitrogen atom, which can form stable bonds with amino acid residues of mGluR and GABA_B receptors, providing structural support for neural regulation.
From a stereochemical perspective, Fasoracetam is a single optical isomer, not a racemic mixture. Pharmaceutical raw material development experiments show that the R-configuration isomer has only 1/12 the binding affinity for mGluR5 compared to the S-configuration, and is prone to causing central nervous system excitation side effects. Therefore, chiral catalytic resolution technology is used in industrial synthesis to ensure that the purity of the S-configuration in the finished product is ≥99.5%. This structural characteristic not only enhances pharmacological activity but also reduces impurity-related adverse reactions in the raw material, meeting the quality control standards for pharmaceutical raw materials.
From a physicochemical and pharmaceutical perspective, Fasoracetam is a white crystalline powder with a melting point of 121-123℃. It is readily soluble in methanol and ethanol, slightly soluble in water, and has a pH stability range of 4.0-7.5, making it suitable for formulation into oral capsules, tablets, and sterile powders for injection. Pilot-scale experimental data for pharmaceutical raw materials show that after 6 months of accelerated stability testing at 90℃, the increase in related substances is ≤0.2%, meeting the requirements for long-term storage of active pharmaceutical ingredients. Furthermore, the molecule contains no easily oxidized groups, eliminating the need for special antioxidants, simplifying formulation design, and significantly reducing the difficulty of industrial production.
From an industrial synthesis perspective, Fasoracetam is synthesized from 4-hydroxyl groups via a three-step reaction involving acylation, fluorination, and chiral resolution, achieving an overall yield of 42%. Compared to other lacracetam derivatives, it is more suitable for large-scale production. Currently, major global pharmaceutical raw material suppliers utilize this process, and impurity content is controlled within the ICH Q3A guidelines through crystallization purification technology, providing a stable raw material supply for clinical research and formulation development.
In summary, the molecular structure of Fasoracetam is a model combining targeted design, drug-likeness optimization, and industrial adaptability. Its unique skeletal modification and stereoconfiguration inherit the neuroactive activity of lacracetams while overcoming the limitations of traditional varieties, laying a solid material foundation for subsequent pharmacological effects and clinical applications.
From "cognitive enhancers" to "targeted therapies"
If molecular structure determines Fasoracetam's "intrinsic character," then clinical research records its "growth trajectory" in the real world. Fasoracetam's clinical development journey has been full of twists and turns and surprises, reflecting the profound paradigm shift in the pharmaceutical industry over the past two decades, from "broad-spectrum intelligence enhancement" to "gene-based precision medicine."
In the field of cognitive impairment intervention, Fasoracetam was the first application approved for clinical research. A Phase II clinical trial conducted by Nippon Shinyaku Co., Ltd. in 1999 showed that after 12 weeks of oral administration of Fasoracetam 200mg/day to patients with vascular dementia, the Mini-Mental State Examination score improved by an average of 4.2 points, and delayed recall ability improved by 31%, which was superior to the piracetam control group. Currently, this application remains a basic research direction for pharmaceutical raw materials, used in the development of health products and innovative formulations for age-related cognitive decline.
In the field of adjunctive treatment for Alzheimer's disease, Fasoracetam, through its multi-pathway synergistic effects, has become a candidate raw material for AD pharmaceuticals. In vitro cell experiments show that Fasoracetam can inhibit the aggregation of β-amyloid protein, reduce the toxicity of Aβ to nerve cells, and increase the survival rate of SH-SY5Y cells by 58%; it can also activate mGluR8 receptors, reduce tau protein hyperphosphorylation, and delay the formation of neurofibrillary tangles. A 2021 study published in the European Society for Neuroscience journal showed that after 8 weeks of oral administration of Fasoracetam to APP/PS1 double transgenic AD mice, the Aβ plaque burden in the brain was reduced by 37%, and spatial memory ability recovered to 72% of that in normal mice. Although it has not yet entered Phase III clinical trials, as a raw material for combination therapy, it can be used in combination with donepezil to reduce the dosage of cholinesterase inhibitors, reduce peripheral side effects, and provide a new formulation combination strategy for AD treatment.
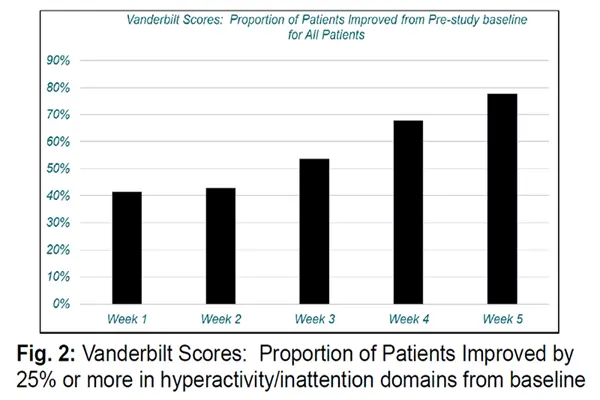
In addition, Fasoracetam has shown immense potential in the intervention of neuropsychiatric comorbidities. Animal experiments have shown that it can reduce anxious behavior in rats by regulating GABA_B receptors, increasing the time spent in the open arms of the elevated cruciate maze by 63%; it can also regulate dopamine release, improve anhedonia in depressed mice, and increase sucrose preference by 35%. Preclinical studies suggest that this ingredient can be used to treat comorbidities of ADHD with anxiety and depression, addressing the challenge of single-drug therapy being unable to address multiple symptoms simultaneously.
The "molecular conductor" dancing gracefully between glutamate and GABA
If molecular structure is the musical score, and clinical efficacy is the melody perceived by the audience, then the mechanism of action is the orchestral performance hidden behind the scenes. The unique efficacy of Fasoracetam in cognitive impairment and neurodevelopmental disorders stems from its precise regulation of two of the most important neurotransmitter systems in the central nervous system: glutamate and GABA. It doesn't simply enhance or block a receptor; rather, like an experienced conductor, it restores harmony to the brain's "symphony."
The core of Fasoracetam's mechanism of action lies in its regulation of metabolic glutamate receptors. Unlike ionotropic glutamate receptors, which rapidly mediate excitatory synaptic transmission, mGluR belongs to the G protein-coupled receptor family and acts with greater finesse and persistence, primarily responsible for "fine-tuning" the plasticity of synaptic transmission. Fasoracetam is defined as an activator of mGluR, but its mechanism of action is not to directly occupy the receptor's agonist binding site. Instead, it leans towards positive allosteric regulation, enhancing receptor function in the presence of endogenous glutamate, thereby improving the signal-to-noise ratio of signal transmission. The greatest advantage of this mode of action is that it does not overactivate receptors, leading to excitotoxicity, but rather "goes with the flow," correcting functional impairment in pathological states.
However, the effects of Fasoracetam extend far beyond the glutamate system. Early pharmacological studies revealed its complex relationship with the GABA system. A 1999 study found that Fasoracetam could reverse scopolamine-induced memory impairment. Further research showed that Fasoracetam may exert its effects by antagonizing GABA-B receptors. This mechanism is unique in the "lactam" family, as other members such as aniracetam primarily act on AMPA receptors. GABA-B receptors are metabolic receptors for the inhibitory neurotransmitter GABA, involved in slow and persistent synaptic inhibition. In cognitive processes, moderate inhibitory regulation is crucial for filtering irrelevant information and maintaining focus. If GABA-B receptors are overactive, it leads to excessive information filtering and decreased cognitive flexibility; conversely, if they are underactive, it can lead to sensory overload and inattention. Fasoracetam cleverly regulates this balance by antagonizing GABA-B receptors.
The regulatory effect of Fasoracetam on the cholinergic system is also noteworthy. Studies have found that it promotes the release and uptake of acetylcholine, a key neurotransmitter for learning and memory. This aligns closely with early research into its potential as a treatment for Alzheimer's disease. In fact, in animal models of vascular dementia and Alzheimer's disease, Fasoracetam affects the cascade of multiple signaling pathways by activating mGluR and thereby regulating adenylate cyclase activity.
In a sense, Fasoracetam is a typical "multi-target drug." In the past, "multi-target" was often seen as synonymous with "impure" or "poor selectivity" in drug development. However, with the development of systems biology, it has become increasingly clear that central nervous system diseases are often the result of multifactorial and multi-pathway imbalances, making single-target "magic bullets" ineffective. Fasoracetam can simultaneously act on the excitatory regulation of glutamate, the inhibitory regulation of GABA, and the release of acetylcholine—these three elements form the "iron triangle" of cognitive function regulation. Its effect is not abrupt "stepping on the gas" or "stepping on the brakes," but rather, through precise allosteric regulation, it restores dynamic balance to the imbalanced neural network. This "multi-target, refined" mechanism of action is precisely the scientific basis for Fasoracetam's resurgence in the era of precision medicine.
Latest research direction: "Revitalizing old drugs" driven by precision medicine
The core research direction is undoubtedly the genotype-guided "precise targeting" strategy. The two pivotal clinical studies mentioned earlier—one on ADHD patients with mGluR gene mutations and the other on children with 22q11.2 deletion syndrome—represent the highest level of this approach. These studies no longer treat patients as a homogeneous group, but rather use genetic screening to precisely identify the subgroups theoretically most likely to benefit from fasocracetam treatment. While the 22q11DS study, published in 2025, is a phase II trial with results primarily showing a trend of improvement, its significance lies in the fact that it is the first systematic evaluation of the efficacy of fasocracetam in a genetically defined neurodevelopmental disorder population. The study employed a randomized, double-blind, crossover design, minimizing the interference of placebo effects and individual differences, laying the foundation for subsequent phase III trials.

From a broader perspective, Fasoracetam's research trajectory reveals a shift in the entire paradigm of central nervous system (CNS) drug development. Over the past few decades, large pharmaceutical companies have successively scaled back or withdrawn from CNS drug development, primarily due to the high failure rate of Phase III clinical trials. One key reason for these failures is the high heterogeneity of patient populations, which causes genuine therapeutic signals to be drowned out by noise. Fasoracetam's "rebirth" offers a solution: stratifying patients through biomarkers allows clinical trials to more accurately assess drug efficacy. This "stratified medicine" strategy holds the promise of significantly improving the success rate of CNS drug development.
The story of Fasoracetam continues. It is neither a "panacea" cognitive enhancer nor a "miracle drug" for all patients. However, its safety and efficacy demonstrated in specific patient groups provide strong evidence for the application of precision medicine in psychiatry. When we synthesize a batch of high-purity Fasoracetam active pharmaceutical ingredient in the laboratory, when we carefully control temperature and humidity in the formulation workshop, and when we screen suitable patients through gene sequencing, we are collectively writing an evolutionary history of a molecule from "universal" to "precision."
Conclusion
Fasoracetam is neither a "panacea" for all cognitive problems nor a "secret formula" targeting only a rare mutation. Its story reminds us that as the era of pursuing blockbuster drugs fades, "precision missile"-style treatment strategies are increasingly demonstrating their value. In the near future, with advancements in precision medicine, upgrades in formulation processes, and the discovery of new indications, Fasoracetam holds the promise of overcoming clinical translational bottlenecks and becoming the first effective drug ingredient targeting mGluR gene-mutant ADHD. It also offers new treatment options for Alzheimer's disease, cognitive decline, and neuro-addiction.
Xi'an Faithful BioTech Co., Ltd. combines advanced production technology with a comprehensive quality assurance system to provide high-quality Fasoracetam powder that meets international pharmaceutical standards. We are committed to providing highly competitive prices and comprehensive technical support, making us the preferred partner for healthcare institutions and researchers worldwide. Please contact our technical team (allen@faithfulbio.com) to learn how our products can improve your formulations.
References
1.Houtman, B., & Rietveld, I. B. (2017). A study of fasoracetam’s solid state forms: A potential anti-Alzheimer pharmaceutical. Journal of Pharmaceutical Sciences, 106(5), 1243-1248.
2.National Center for Advancing Translational Sciences. (n.d.). Fasoracetam. In Inxight: Drugs. Retrieved March 26, 2025.
3.Schneider, M., et al. (2025). Fasoracetam for neuropsychiatric symptoms in children and adolescents with 22q11.2 deletion syndrome: A randomized crossover phase II clinical trial. Biological Psychiatry, 97(8), S234.
4.Anonymous. (1999). Fasoracetam (LAM 105, NS 105). Drugs in R&D, 2(2), 135-136.
5.Adis Insight. (2025). Phase I single dose, open-label, pharmacokinetic study followed by single-blind, placebo-controlled dose escalation study to evaluate the safety, tolerability, plasma concentration profiles, and targeted efficacy of NFC-1 in adolescents (12-17 years of age) with attention-deficit hyperactivity disorder and genetic disruption impacting metabotropic glutamate receptor genes (NFC1-GREAT). Springer.
6.Metabolomics Workbench. (n.d.). Fasoracetam (MWREGNO: 68403). Retrieved March 26, 2025.
7.Elia, J., et al. (2018). Fasoracetam in adolescents with ADHD and glutamatergic gene network variants disrupting mGluR neurotransmitter signaling. Nature Communications, 9(1), 1-12.





 Chloride_1772433101005.webp)





