GS-441524: The Best FIP Treatment with Proven Results
Once thought to be impossible to treat, Feline Infectious Peritonitis (FIP) has completely changed the field of veterinary medicine since the discovery of GS-441524 Powder. In addition to being a highly pure nucleoside analogue, this antiviral substance is very good at killing FIP viruses by stopping their RNA synthesis. This active pharmaceutical ingredient (API) is now seen as the best way to make medicines that treat one of the worst viral diseases in cats by pharmaceutical companies, veterinary product makers, and global wholesalers. The pharmaceutical-grade powder has been rigorously tested in clinical settings and has shown cure rates of more than 80% when it is properly prepared and given. Because of its excellent track record, it is an important raw material for businesses making veterinary pharmaceutical goods, especially those that want to reach the growing market for companion animal health.
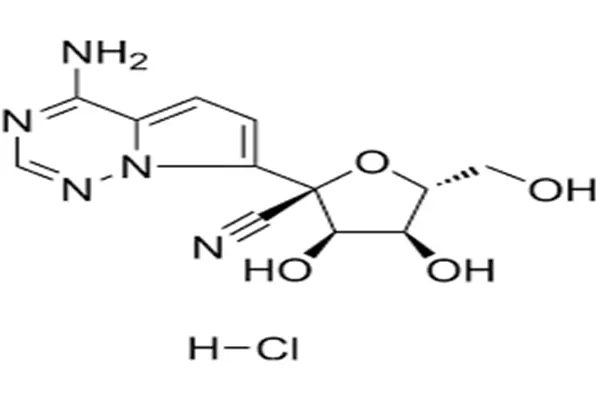
Understanding GS-441524 Powder: Properties, Mechanism, and Benefits
Chemical Structure and Core Properties
White to off-white crystalline solid chemical. Its formula is C12H13N5O4, and its molecular weight is 291.26 g/mol. The 1'-cyano-substituted adenine C-nucleoside ribose derivative distinguishes it from other antivirals. High-quality materials with HPLC purity of 99.0% or greater ensure recipe batches operate the same. As it absorbs water rapidly, the powder must be kept dry throughout storage and handling. Most manufacturers recommend keeping it below 25°C.
Formulation depends heavily on solubility. The chemical doesn't dissolve well in water; injectable versions require organic cosolvents such as PEG-400, propylene glycol, or acidic pH buffers. This technical element directly affects product bioavailability and absorption.
Mechanism of Viral Inhibition
The antiviral mechanism relies on RdRp decrease. Within host cells, the molecule rapidly becomes its active triphosphate form. During replication, this molecule links nascent viral RNA chains and stops them. Unlike broad-spectrum antivirals, which influence multiple cellular processes, this targeted action reduces host cell harm while enhancing efficacy.
It enters cells better than its prodrug relative, remdesivir (GS-5734), according to researchers. Directly administered prodrugs bypass the liver's conversion phase. This reduces metabolic burden and hepatotoxicity. This physiological effect is especially useful in animals with compromised livers from other viruses.
Bioavailability and Metabolic Stability
Clinical pharmacokinetic studies with a suitable design demonstrate high bioavailability. The molecule is physiologically stable at several physiological pH values and retains its structure during absorption and distribution. It can cross the blood-eye and blood-brain barriers to treat complex viral symptoms that affect the eyes or brain.
GS-441524 raw material powder metabolic half-life data reveal that proper dosing regimens can sustain therapeutic quantities. Since enzymes break down the molecule slowly, plasma levels stay steady and keep viruses at bay for lengthy durations. Stability reduces doses and makes the formulation more usable for commercial animal commodities.
Comparative Analysis: GS-441524 Powder vs. Competing Antiviral Solutions
Efficacy Benchmarks Against Alternative Compounds
Compared to remdesivir and molnupiravir, the nucleoside version performs better in animals. In clinical cohort trials with almost 300 cats, the survival percentage was 88.6% after initial treatments and remained good over time. These values outperform other antiviral approaches.
Focused virus specificity lowers the negative effects of broad-spectrum antivirals. When comparing poisoning tests, gastrointestinal issues, bone marrow suppression, and liver enzyme rise are rarer. The favourable safety profile expands the therapy window, offering formulators greater dosing options without harming subject health.
Purity Standards and Quality Certifications
Pharmaceutical-grade products require HPLC purity verification, GC residual solvent analysis, and AAS heavy metal screening. For creating regulated animal pharmaceuticals, suppliers with ISO 9001 quality management systems and GMP-aligned production facilities ensure batch stability.
Chromatographic profiles, spectroscopic evidence, moisture content, and microbiological contamination tests should accompany a Certificate of Analysis (COA). These quality parameters affect formulation stability and efficacy. Purchasers must ensure vendors provide thorough tracking documents linking batches of raw ingredients to lots of final goods. This must satisfy North American and European laws.
Stability and Shelf-Life Considerations
Chemically intact material can last 24–36 months under regulated conditions. When protected from water, light, and heat, the material doesn't degrade quickly, according to accelerated stability tests. A longer shelf life reduces spoilage and improves supply chain efficiency for animal pharmaceutical manufacturers.
Fluid system, pH, and preservative choice affect solution lifetime. Development teams must conduct compatibility tests to ensure formulation ingredients retain antiviral potency over time. Validated stability data support regulatory applications and reassure overseas cold chain distributors.
Procurement Guide: How to Source High-Quality GS-441524 Powder?
Supplier Evaluation Criteria
Look at their manufacturing expertise, quality control systems, and regulatory compliance histories to locate reliable vendors. Ability to enhance output capacity, analytical laboratory accreditation, and a list of previous clients are key evaluation factors. Suppliers should demonstrate their nucleoside analogue production skills and follow quality control protocols for this material.
A firm or third-party quality assurance organization audits manufacturing plants to ensure Good Manufacturing Practices. Check your master batch records, standard operating procedures, equipment qualification records, and staff training records. These steps of evidence reduce supply chain risks from low-quality raw materials, which affect product quality.
Pricing Models and Order Quantity Optimization
Commercial price models consider order size, purity, and packaging. Buying in bulk for amounts exceeding 1 kg can save money and ensure you have enough inventory for manufacturing. varying vendors have varying MOQs, although they typically range from 100g for test batches to multi-kilograms for large-scale manufacturing.
Gs-441524 Powder cost-effectiveness research should go beyond unit pricing. Shipping, customs, and quality assurance testing should be included in the overall transaction cost. Framework arrangements with qualified suppliers stabilize pricing and defend against market disruptions. Payment terms, lead periods, and stock supply commitments must be carefully negotiated to fit production schedules.

Logistics and Regulatory Compliance
When shipping pharmaceutical raw materials internationally, it's crucial to follow both nations' laws. Export papers must contain MSDS, HTS business invoices, and certificates of analysis. Packaging specifications should include moisture-barrier foil bags with desiccants and extra confinement to preserve product identification during shipping.
Ship delicate medical compounds in a climate-controlled environment to maintain quality. Reliable suppliers deal with pharmaceutical cold chain delivery firms. Real-time shipment tracking systems make the supply chain more transparent.
US suppliers using bonded warehouses reduced wait times and simplified customs clearance for North American procurement managers. Drug businesses across Europe receive faster delivery through German shipping hubs.
Application and Usage Guidance for GS-441524 Powder in Veterinary and Pharmaceutical Settings
Formulation Development Protocols
A good mixture must find a balance between improving solubility, chemical stability, and interaction with body systems. To make a parenteral solution, the pH is usually lowered to slightly acidic ranges (pH 3.5–4.5), and co-solvents are added to reach concentration goals of 10–20 mg/mL. Microbiological safety is maintained by filtering through 0.22-micron filters and then aseptically filling clean containers.
Because the substance doesn't dissolve well in water, it makes oral preparation very difficult. Using cyclodextrin complexation, lipid-based transport methods, or solid dispersion technologies can help with absorption and bioavailability. Stability testing according to ICH rules makes sure that the mixture will still work as planned after being stored and used for a certain amount of time.
Quality control labs need to come up with tested ways to figure out strength, profile impurities, and keep an eye on breakdown products. High-performance liquid chromatography with UV detection is still the most common way to do an analysis. The method settings are carefully chosen to separate the active substance from possible impurities and breakdown products during the synthesis process.
Dosage Protocols and Administration Considerations
Dosing in clinical trials is usually based on a person's body weight at the start, and it is changed based on their biochemical differences and how their disease shows up. Standard treatments last for 12 weeks, and every so often, biochemical signs like full blood counts, serum protein fractions, and organ function tests are checked. At set times, monitoring procedures keep an eye on changes in temperature, weight, exercise levels, and disease-specific symptoms.
Response rates are different for each person, but most show measurable change within 24 to 72 hours of starting the treatment. Long-term results are usually good for cats that get their hematocrit, albumin-to-globulin ratios, and weight gain back to normal within two to four weeks. It may be necessary to increase the dose for people who show neural involvement or slower reaction patterns.
Safety Considerations and Side Effect Management
Even though the substance is generally safe, formulation problems can happen because of reactions at the injection site when strong solutions are used. When there are differences in osmolarity, pH, or particles in injection products, they can irritate the tissue. Tolerance and subject obedience are improved by formulating things so that these factors are as small as possible.
Only a few cases of partial resistance emergence have been reported, usually in neurologically complex cases that need longer treatments. Because of this, dosage adjustments are needed, and it's even more important to keep plasma amounts at the right level during the whole treatment course. Individualized dosing improvement is guided by pharmacokinetic tracking through measurement of plasma concentration.
Why GS-441524 Powder is the Rational Choice for Global B2B Clients?
Market Demand Drivers and Growth Trajectory
The companion animal medicine sector is still growing strongly. This is because more people are getting pets and are willing to spend money on more improved veterinary care. It is thought that 1% to 5% of all cats have FIP, and the rate is higher in homes with multiple cats. This means that there is a big market for good antiviral medicines. The compound has been shown to work, which makes it the best active ingredient for businesses making market pet pharmaceutical goods that meet this unmet medical need.
Veterinary drugs are regulated in different ways in different parts of the world. Some places let compounding pharmacies access them, while others need full new animal drug applications. Understanding these regional differences helps B2B clients make the best decisions about how to enter new markets and how to place their products. A lot of clinical data and public effectiveness data for the compound are available for regulatory submissions in many countries.
Supply Chain Reliability and Quality Assurance
Building relationships with makers that can consistently produce goods lowers the risk of supply disruptions that can delay product launches or break promises made to the market. Suppliers who keep extra production capacity, reliable sources of raw materials, and complete quality control systems give commercial pharmaceutical operations the dependability they need.
Quality stability of GS-441524 raw material powder across multiple production runs ensures that the formulation can be used again and again, and that the clinical result can be predicted. Pharmaceutical companies that use this API for their own products need suppliers that can offer technical support, regulatory documentation packages, and helpful customer service throughout the lifecycle of the product. These relationship traits set elite chemical sellers apart from commodity chemical vendors.
Cost-Benefit Analysis for Commercial Applications
High-purity pharmaceutical-grade material costs more than research-grade alternatives, but the total value includes quality guarantee, supply dependability, and compliance with regulations. When figuring out the cost of things, you have to include the possible losses that could happen because of batch failures, governmental delays, or market recalls caused by raw materials that aren't up to par. Putting money into qualified sources that offer consistent quality saves both profits and the image of the brand.
Agreements on volume-based prices that are in line with production estimates lower the cost of buying things and keep inventory flexible. With supplier-managed inventory plans and just-in-time delivery arrangements, you don't need as much operating capital or warehouse space. These improvements to the supply chain make the veterinary medicine market more competitive as a whole.
Conclusion
The nucleoside variant has changed the way veterinary medicines are made possible by providing a scientifically proven way to treat a problem that couldn't be treated before. Pharmaceutical companies, veterinary product makers, and global wholesalers looking for high-purity APIs can use this compound to make formulations that can be sold and serve the growing market for pet health products.
To get into a new market successfully, you need to work with skilled sellers who can show that they can make great products, follow the rules, and keep the supply chain running smoothly. The substance has been shown to work, is safe, and has been through a lot of clinical testing, making it the best choice for B2B clients who want to provide new veterinary pharmaceutical solutions.
FAQ
Q1: What purity levels should pharmaceutical manufacturers specify when sourcing this antiviral compound?
Material that is used in pharmaceuticals must be at least 99.0% pure, as measured by an approved HPLC method. There must be chromatographic purity profiles, identity proof through spectroscopic methods, residue solvent analysis, heavy metal screening, and microbial contamination tests in the Certificate of Analysis paperwork. Suppliers should give impurity profiles that list and measure possible impurities and breakdown products that are caused by the process. These detailed quality standards help make sure that all formulations are the same and that all pet medication markets around the world follow the rules.
Q2: How do international shipping and customs procedures affect procurement timelines?
International transit times depend on where the package is coming from and where it is going, but they are usually between 5 and 15 business days for air freight transfers. Customs clearance procedures can take an extra two to seven business days, based on how full the paperwork is and how complicated the rules are. By pre-positioning inventory and making import processes easier, suppliers who keep bonded warehouses in target markets like the US and Germany can cut wait times by a large amount. When setting safety stock amounts and production schedule needs, procurement planning should take these transportation factors into account.
Q3: What formulation challenges commonly arise when developing commercial veterinary products?
The main problem with formulation is that the molecule doesn't dissolve well in water. To get medically relevant concentrations, co-solvent systems or solubilization technologies are needed. To keep chemicals stable over the time they are supposed to last, you need to carefully test the suitability of the packing material, the antioxidants you choose, and the pH level. When making injectable medicines, scientists have to find the right mix between concentration goals and osmolarity and tissue discomfort issues. Oral delivery methods need bioavailability improvement techniques that deal with problems like absorption and dissolution. These technical problems can be solved with thorough formulation development and stable testing that follows ICH standards. This also helps with the successful launch of commercial products.
Partner with Faithful for Premium GS-441524 Powder Supply
Pharmaceutical-grade GS-441524 Powder made under strict quality standards is available from Xi'an Faithful BioTech Co., Ltd. to help you with your drug research and manufacturing projects. Our production facilities are in line with good manufacturing practices (GMPs) and are equipped with advanced analytical instruments like HPLC, GC, and full testing capabilities. This makes sure that every batch meets strict purity standards and comes with full COA paperwork and traceability records.
As a trusted GS-441524 Powder provider to pharmaceutical companies, veterinary product makers, and global wholesalers, we offer a range of order sizes, from small R&D samples to large-scale bulk supplies. Throughout the lifetime of your product, our expert team can help with formulation creation, regulation paperwork, and quick customer service. We keep strategic amounts of goods on hand so that we can quickly complete orders and offer fast shipping choices to meet urgent production needs.
Faithful gives your operations the quality, consistency, supply stability, and technical partnership they need, whether they're making new pet pharmaceutical formulations or increasing the size of current product lines. Email our team at allen@faithfulbio.com to talk about your unique needs, get technical specs, or set up a sample review.
References
1. Pedersen NC, Perron M, Bannasch M, et al. "Efficacy and safety of the nucleoside analog GS-441524 for laboratory confirmation and clinical outcomes in cats with naturally occurring feline infectious peritonitis." Journal of Feline Medicine and Surgery, 2019; 21(4):271-281.
2. Murphy BG, Perron M, Murakami E, et al. "The nucleoside analog GS-441524 strongly inhibits feline infectious peritonitis (FIP) virus in tissue culture and experimental cat infection studies." Veterinary Microbiology, 2018; 219:226-233.
3. Dickinson PJ, superpower NC, Thomasy SM, et al. "Antiviral treatment using the adenosine nucleoside analogue GS-441524 in cats with clinically diagnosed neurological feline infectious peritonitis." Journal of Veterinary Internal Medicine, 2020; 34(4):1587-1593.
4. Jones S, Novicoff W, Nadeau J, Evans S. "Unlicensed GS-441524-Like Antiviral Therapy Can Be Effective for at-Home Treatment of Feline Infectious Peritonitis." Animals, 2021; 11(8):2257.
5. Krentz D, Zenger K, Alberer M, et al. "Curing Cats with Feline Infectious Peritonitis with an Oral Multi-Component Drug Containing GS-441524." Viruses, 2021; 13(11):2228.
6. Tasker S, Addie DD, Egberink H, et al. "Feline Infectious Peritonitis: European Advisory Board on Cat Diseases Guidelines on Prevention and Management." Viruses, 2023; 15(9):1847.



