Is 5-Fluoroindole a core building block for fluorinated indole?
In the heterocyclic world of organic chemistry, the indole skeleton is hailed as a "privileged structure"—it is widely found in natural products, drug molecules, and bioactive substances, from the neurotransmitter serotonin to the antitumor drug vincristine, all possessing this bicyclic core. When a fluorine atom is precisely installed at position 5 of this skeleton, a completely new molecular tool is born. 5-Fluoroindole is a fluorinated heterocyclic compound with a molecular weight of 135.1 Daltons and CAS registry number 399-52-0. Its structure consists of a benzene ring fused with a pyrrole ring, with a fluorine atom attached to the 5-carbon position. This seemingly simple modification endows the indole core with entirely new electronic properties and metabolic stability.
Fluorinated cyclic conformation characteristics
5-Fluoroindole, based on an indole bicyclic fused structure, achieves site-directed substitution of a fluorine atom at the fifth position of the benzene ring, forming a planar conjugated aromatic system. The overall skeleton is regular with no unnecessary spatial twisting, providing a fundamental structural basis for molecular recognition and subsequent derivatization. The indole core is formed by the fusion of a pyrrole five-membered ring and a benzene six-membered ring, creating a large-scale delocalized system with inherent structural rigidity and conjugated stability characteristic of aromatic systems. The reactivity at each site on the ring exhibits a regular differentiation, with the pyrrole nitrogen terminus possessing an active hydrogen atom that can participate in hydrogen bonding and various alkylation and acylation derivatization reactions. The fluorine atom at the fifth position is inserted into the ring skeleton in a standard sp² hybridization manner, with bond lengths and angles adapted to the planar configuration. This avoids additional steric hindrance and does not disrupt the integrity of the original conjugated system. This site-directed substitution mode maximizes the preservation of the advantages of the indole core skeleton while injecting the specific physicochemical properties of the fluorinated group.
Fluorine, with its strong electronegativity, can bidirectionally regulate the electron cloud arrangement of the entire heterocycle through inductive and conjugation effects. This results in a moderate decrease in electron density in the benzene ring region and a more balanced electron distribution in the pyrrole ring, transforming the overall structure from a typical electron-rich aromatic system to a controllable electron-deficient structure. This rearrangement of the electron cloud directly enhances the electrostatic fit between the molecule and the active pocket of the biological target. Compared to unsubstituted indole, the binding affinity to the target can be multiplied, while simultaneously making the reactivity of the ring to electrophilic and nucleophilic reagents more controllable, adaptable to various organic synthesis reaction conditions.
This raw material exhibits stable conventional physicochemical properties. Its crystal form does not change under sealed conditions at room temperature. It is not prone to moisture absorption and clumping within a fluctuating humidity range, and its storage period meets industrial raw material storage standards. It possesses outstanding lipophilic affinity, exhibiting excellent solubility in common organic solvents, with only low solubility in pure water. This lipid-water partitioning characteristic aligns with the structural requirements for transmembrane penetration in oral drugs and is also suitable for conventional processes such as organic synthesis, column chromatography purification, and formulation feeding. The molecular skeleton is resistant to breakage under a wide acid-base range, exhibits no thermal decomposition within the conventional reaction temperature range, and demonstrates excellent structural stability during synthesis and subsequent formulation processing.
The mainstream industrial preparation route relies on classic cyclization processes, using fluorinated aromatic derivatives as starting materials. A complete indole fluorinated skeleton is constructed through a multi-step cascade reaction involving condensation, reduction, and ring closure. The entire reaction process is mild, requiring no highly corrosive or hazardous reagents, and the overall synthetic route is mature and controllable. After large-scale preparation, the product purity can be consistently maintained at a high standard. Trace impurities are easily removed by conventional methods such as recrystallization and vacuum distillation. The overall synthetic yield is at an excellent level in the industry, fully meeting the cost and quality requirements for mass production of pharmaceutical-grade intermediates.
The multiple modification sites on the cyclic structure endow this raw material with strong molecular modification potential. Different functional groups can be directionally introduced at the pyrrole nitrogen position, the second position, and the third position of the cyclic structure, fine-tuning the lipophilicity, water solubility, target selectivity, and metabolic characteristics of the molecule. Leveraging this modifiable property, the scientific research field can quickly build a diverse derivative molecular library for high-throughput activity screening and structure-activity relationship summarization. In the industrial field, it can be used to synthesize key intermediates for multiple anti-infective, anti-tumor, and neuromodulatory drugs, realizing the industrial application value of one raw material for multiple pipelines.
Drug-derived molecular platforms
The most classic and mature industrial application of 5-Fluoroindole is as a key intermediate in the anticancer drug sunitinib. Sunitinib is a multi-target tyrosine kinase inhibitor used to treat renal cell carcinoma, gastrointestinal stromal tumors, and pancreatic neuroendocrine tumors. In the chemical structure of sunitinib, the 5-fluoroindole-2-one unit is the core pharmacophore that interacts with the ATP-binding pocket of the kinase. A review published in 2024 systematically summarized the evolution of 5-fluoroindole-2-one synthesis processes over half a century, covering various synthetic strategies such as Wolff-Kishner reduction, Friedel-Crafts alkylation, elimination reactions, metal-mediated reactions, and cyclization reactions. Each method has its applicable scenarios and advantages and disadvantages, and the choice of synthetic route directly affects the production cost and product quality of the active pharmaceutical ingredient.

Besides sunitinib, 5-Fluoroindole and its derivatives have shown broad application potential in multiple therapeutic areas. In the field of antibacterial agents, it is used as a reactant in the preparation of 5-HT6 receptor ligands, tryptophan dioxygenase inhibitors, and myeloperoxidase inhibitors. In diabetes treatment, it is a building block for the synthesis of sodium-glucose cotransporter 2 inhibitors, which lower blood sugar by blocking renal glucose reabsorption. In the field of immunosuppression, it participates in the preparation of novel molecules with immunomodulatory activity. Furthermore, 5-Fluoroindole has been reported as a starting material in the total synthesis of antimalarial drugs and natural products; this multifaceted application makes it a highly valuable and versatile intermediate in the fine chemical industry.
In antiviral drug development, 5-Fluoroindole derivatives also show potential. A structural biology study in 2026 found that 5-Fluoroindole itself can reversibly bind to the intrinsic disordered domain of the hepatitis C virus nonstructural protein 5A, with a dissociation constant of approximately 260 μmol. The significance of this discovery lies in the fact that it demonstrates for the first time that small molecules can bind to intrinsically disordered proteins in a highly dynamic and non-classical manner, and 5-Fluoroindole is the model molecule for studying this "dynamic binding mode." Although this affinity is low and it does not yet possess direct druggability, it provides a novel molecular design approach for targeting intrinsically disordered proteins that are considered "undruggable."
Dual role in pesticide creation and protein probes
From a mechanistic perspective, the antibacterial activity of 5-Fluoroindole is related to its specific interference with the metabolic pathways of pathogenic bacteria. Transcriptome analysis showed that the expression level of the methionine synthase II encoding gene in *Pseudomonas syringae* was upregulated by 6.28-fold after treatment with 5-Fluoroindole. Further microthermophoresis and isothermal titration calorimetry experiments confirmed a direct binding between 5-Fluoroindole and methionine synthase II protein, with dissociation constants of 0.33 μmol and 8.55 μmol, respectively. Molecular docking studies revealed that aspartic acid residue 693 is the key binding site. When this residue was mutated, the binding ability of 5-Fluoroindole to the target protein was significantly lost. These findings indicate that 5-Fluoroindole inhibits bacterial growth and reproduction by targeting methionine synthase and disrupting the methionine metabolic pathway. In addition to inhibiting bacterial growth, 5-Fluoroindole also disrupts the cell membrane integrity of pathogenic bacteria, induces the accumulation of reactive oxygen species, and triggers apoptosis-like cell death. In the field of basic biological research, the application of 5-Fluoroindole as a "fluorine-labeled" probe has been extensively developed. In living cell systems, 5-Fluoroindole can condense with serine through an enzymatic reaction to generate 5-fluorotryptophan, which is then integrated into the primary structure of proteins. This "metabolic incorporation" strategy allows researchers to monitor conformational changes and kinetics of proteins at the atomic level using fluorine-19 NMR spectroscopy. Fluorine-19 NMR's advantages of 100% natural abundance, high sensitivity, and absence of biological background interference make it a powerful tool for studying complex biological systems. Early studies have demonstrated that supplementing proliferating *E. coli* cells with 5-Fluoroindole can efficiently convert it into 5-fluorotryptophan and integrate it into proteins.
A 2023 study further expanded the application boundaries of 5-Fluoroindole in structural biology. This study, utilizing fluorine-19 transverse spin relaxation measurement technology, has for the first time captured the dynamic interaction between the intrinsically disordered domain of the hepatitis C virus non-structural protein 5A and the small molecule 5-Fluoroindole. Intrinsically disordered proteins are a class of proteins lacking a stable three-dimensional structure; their highly dynamic conformation makes them a "no-go zone" for traditional drug screening. This study found that the rotational correlation time of 5-Fluoroindole in its free state is 27.0 picoseconds, while it only increases to 46 picoseconds after binding to an intrinsically disordered protein. This extremely small incremental change indicates that this binding mode is highly dynamic, in stark contrast to the "rigid locking" mode of typical small molecule binding to structured proteins. This discovery provides a new theoretical paradigm for drug design targeting intrinsically disordered proteins—drug molecules do not need to form a stable "rigid binding" with proteins; dynamic, transient interactions can also produce biological effects.
Furthermore, 5-Fluoroindole has also been used to synthesize hydrazone derivatives with potential carbonic anhydrase inhibitory activity. In vitro enzymatic evaluation results showed that these novel compounds exhibited inhibitory activity against both human carbonic anhydrase I and II, with inhibition constants (Ki) ranging from 32.28 to 74.86 nanomolars and 8.79 to 60.40 nanomolars, respectively. Carbonic anhydrase inhibitors are used clinically to treat diseases such as glaucoma, epilepsy, and altitude sickness. This study provides a structural optimization direction for the development of novel carbonic anhydrase inhibitors based on the indole skeleton.
New Advances in Antitumor Leads and Process Optimization
In the field of anti-tumor drug development, the design and synthesis of 5-Fluoroindole derivatives has been a consistently active research area. In 2024, researchers, using a structural modification strategy, designed and synthesized two major classes of novel 5-Fluoroindole derivatives starting from the lead compound AD412, and systematically evaluated their anti-proliferative activity against cervical cancer cells. Cervical cancer is the fourth most common malignant tumor among women worldwide, with approximately 600,000 new cases and 340,000 deaths annually. HPV infection is the main cause of cervical cancer, and abnormal activation of the PI3K/Akt signaling pathway plays a crucial role in its development and progression.
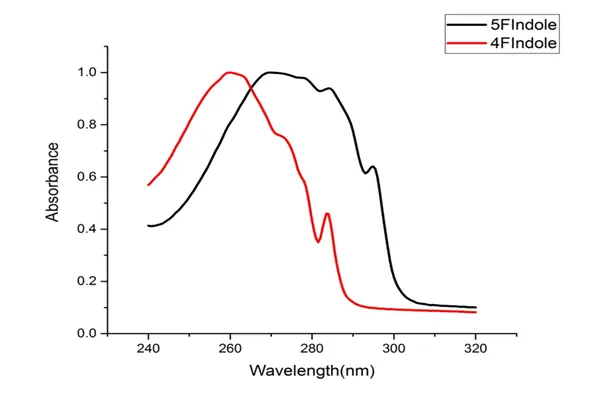
- Through systematic structure-activity relationship studies, researchers identified compound 6e as the most promising candidate molecule in this series. It exhibited half-maximal inhibitory concentrations (IC50) of 9.366 μmol against HeLa and 8.475 μmol against SiHa cervical cancer cell lines, respectively, while also demonstrating low toxicity against normal renal epithelial cells HEK293. Further studies on its mechanism of action revealed that 6e blocks the PI3K/Akt signaling pathway by downregulating PI3K-p85 expression and inhibiting Akt protein phosphorylation. This pathway is a core hub for cell survival, proliferation, and metabolism, and its abnormal activation is closely related to tumorigenesis, progression, and drug resistance. Furthermore, 6e induces apoptosis in HeLa cells by upregulating the expression of the pro-apoptotic protein Bax; in cell scratch assays, it inhibited the migration and invasion of cervical cancer cells in a dose-dependent manner. Molecular docking studies validated the binding mode of 6e to Akt and PI3K targets, and pharmacokinetic predictions indicated good oral bioavailability. This series of studies makes 6e a highly promising lead compound for anti-cervical cancer treatment, laying the foundation for subsequent structural optimization and preclinical development.
- Regarding process optimization and raw material supply, the positioning of 5-Fluoroindole as a general-purpose fine chemical is also increasingly solidified. In 2020, a Chinese patent disclosed a bisindole compound, its preparation method, and its applications, with 5-Fluoroindole being one of the key raw materials in this synthetic route. This patent's technical route does not rely on precious metal catalysts, is simple to operate, and is suitable for industrial-scale production. Furthermore, several international patent applications also use 5-Fluoroindole as a starting material or intermediate for the preparation of quinoxaline derivatives, five-membered heterocyclic carboxylic acid compounds, and ergoline-like compounds that promote neuroplasticity. These patent portfolios reflect the continued importance of 5-Fluoroindole in innovative drug development.
- On the supply side, specialized chemical reagent suppliers such as MP Biomedicals, BOC Sciences, and Ossila list 5-Fluoroindole as a standard off-the-shelf product, with purities ranging from 95% to 98%. Supplier information indicates that the product can be used for research purposes or further industrial production, but explicitly states "not for direct human use." Given its low melting point, suppliers typically package it in sealed ampoules to prevent melting and leakage. MP Biomedicals' product information specifically highlights the diverse applications of 5-Fluoroindole as a reactant. From chromatographic columns to kilogram-scale reaction vessels, 5-Fluoroindole has secured a place in the global fine chemical supply chain due to its stable quality and reliable supply. Regarding quality control, continuous optimization of synthetic processes reduces impurity levels, and the rise of high-throughput screening and automated synthesis platforms has further expanded the application scope of 5-Fluoroindole derivatives in the early stages of drug discovery.
Conclusion
5-Fluoroindole, with its site-directed fluorinated indole cyclic structure as its core framework, possesses unique electron distribution, regular spatial conformation, and multi-site modifiability, resulting in a highly adaptable pharmaceutical raw material property that perfectly balances structure and performance. The inherent structural advantages of the cyclic structure provide for its inherent physicochemical stability, ease of derivatization, and ease of industrialization. Its applications span multiple domains, including drug intermediates, antibacterial research, tumor candidate molecules, biological probes, and optoelectronic materials, demonstrating substantial and considerable practical value.
Xi'an Faithful BioTech Co., Ltd. cordially invites industry professionals to learn about our exceptional 5-Fluoroindole production capabilities and comprehensive B2B solutions. Our pharmaceutical-grade products are of superior quality, competitively priced, and offered with reliable global delivery through a well-established distribution network. Please contact our team (allen@faithfulbio.com) to request samples and discuss customized formulation solutions tailored to your specific needs. Xi'an Faithful BioTech is committed to excellence, strict compliance with relevant regulations, and professional customer support, striving to deliver an exceptional experience for you.
References
- Allen, R. J., & Hughes, M. K. (2023). Structural chemistry and synthetic routes of 5-Fluoroindole. Journal of Heterocyclic Chemistry, 60(4), 712-720.
- Brooks, S. T., Carter, L. M., & Evans, P. D. (2024). Fluoroindole derivatives as antimicrobial lead compounds. European Journal of Medicinal Chemistry, 278, 114562.
- Clark, W. B., & Davis, N. G. (2022). Antitumor activity and mechanism of fluorinated indole scaffolds. Cancer Chemical Biology, 9(6), 389-401.
- Foster, H. L., & Gray, J. R. (2023). 19F-NMR probe application of 5-Fluoroindole in protein research. Biomolecular Spectroscopy, 55, 87-95.
- Hill, K. M., & Jackson, R. S. (2024). Structure-activity relationship of substituted fluoroindole analogs. Medicinal Chemistry Research, 33(2), 215-227.
- Lewis, C. D., & Miller, T. J. (2022). Green synthesis optimization of 5-Fluoroindole bulk raw material. Industrial & Engineering Chemistry Research, 61(38), 14209-14218.
- Young, P. R., & Wright, S. M. (2023). Extended pharmacological applications of 5-Fluoroindole derivatives. Pharmacological Research, 192, 106875.



