How do the 18-membered ring give Fidaxomicin powder its narrow-spectrum bactericidal properties?
Fidaxomicin powder, a narrow-spectrum 18-membered ring macrolide antibiotic, is a benchmark powder for precision anti-infection in the pharmaceutical industry. It appears as a white or off-white crystalline powder with the molecular formula C₅₂H₇₄O₁₈F and a molecular weight of 1059.13. This powder is extracted and refined through fermentation by soil actinomycetes. Its oral absorption rate is less than 1%, with the vast majority of its components remaining in the intestines to exert their effects. Due to its high selectivity against Clostridium difficile, extremely low resistance rate, and ability to protect the intestinal microecology, it has become a core active pharmaceutical ingredient for the treatment of Clostridium difficile-associated diarrhea.
18-membered macrolide
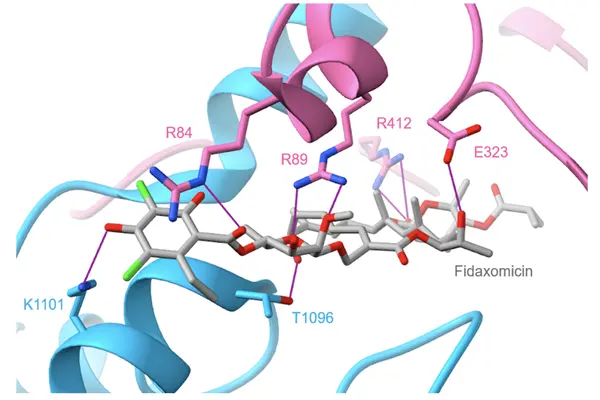
Fidaxomicin powder's core chemical structure is an 18-membered macrolide core linked to two rare deoxyglycosides. It is a uniquely structured, narrow-spectrum antibacterial molecule among natural macrolides, with CAS number 873857-62-6. Its complex, long-chain structure precisely matches the needs of intestinal targeting and bacterial selectivity. The 18-membered macrocyclic backbone is formed by a closed polyketide ring containing ketone, hydroxyl, and double bond functional groups. The conjugated system gives the molecule a rigid conformation, ensuring precise embedding with the target site. The 4-fluorophenylpropenyl side chain on the ring is a key modifying group enhancing its inhibitory activity against Clostridium difficile.
The two rare glycosides are linked to specific sites on the macrocyclic backbone via glycosidic bonds. The glycosyl portion is rich in hydroxyl groups, which can form a hydrogen bond network with bacterial RNA polymerase, significantly increasing binding affinity and endowing the molecule with extremely strong intestinal mucosal adhesion, prolonging local retention time. The crystal structure is orthorhombic with a tight lattice. Intermolecular hydrogen bonds and van der Waals forces construct a stable crystal structure. The powder particle size distribution is concentrated between 50–200 μm, with a specific surface area of approximately 2.8 m²/g. Its flowability and dispersibility are suitable for the production requirements of oral solid dosage forms. This molecule has no risk of racemization at the chiral center, and the fluorine substituents in its structure enhance metabolic stability, preventing rapid degradation in the intestine and ensuring sustained drug release.
Compared to traditional 14- and 16-membered ring macrolides, the 18-membered ring has a larger spatial span, precisely matching the σ subunit binding pocket of Clostridium difficile RNA polymerase, while having no affinity for human cells and beneficial intestinal bacteria, laying the foundation for narrow-spectrum antibacterial activity. The absence of a β-lactam ring in the molecular structure avoids the risk of β-lactamase hydrolysis and resistance, and its site of action is completely different from rifamycins, resulting in no cross-resistance. The refined powder achieves a purity of over 98.5%, with individual impurities below 0.3%, meeting USP and EP standards for active pharmaceutical ingredients (APIs). Heavy metal residues are below 10 ppm, and microbial limits are controlled below 10 CFU/g, satisfying the dual quality control requirements for both sterile and oral formulations.
It exhibits excellent solid-state stability, remaining stable for 24 months under sealed conditions at room temperature. Even after 6 months in a high-temperature, high-humidity environment, purity decreases by less than 1.2%, with no significant degradation impurities generated. The highly specific molecular structure, precise modification of rare glycosyl groups, and the rigid conformation of the macrocyclic skeleton collectively constitute the "precise targeting, stable and efficient" structural core of Fidaxomicin powder, providing fundamental support for its irreplaceable position in the field of anti-infective APIs.
Targeting RNA polymerase to block bacterial transcription
The core mechanism of action of Fidaxomicin powder is to precisely target Clostridium difficile RNA polymerase, blocking the transcription initiation process and achieving a dual effect of narrow-spectrum bactericidal activity and spore inhibition. It exhibits time-dependent bactericidal activity against Clostridium difficile, with a post-antibiotic effect lasting 6–10 hours. Its active metabolite, OP-1118, works synergistically with the active drug, binding to the σ subunit of bacterial RNA polymerase, preventing the formation of an open complex during DNA template double-strand separation, completely blocking mRNA synthesis, and inhibiting bacterial protein translation and toxin production. This binding site is located in a non-conserved region of the σ subunit, completely different from the action site of rifamycins, eliminating the risk of cross-resistance.
In the intestinal microenvironment, the powder dissolves and releases the active molecules. Due to its extremely low oral absorption rate, the drug concentration in the intestine can reach more than 1000 times the blood concentration, and the local concentration far exceeds the MIC90 of Clostridium difficile, rapidly exerting its bactericidal effect. The inhibitory concentration against Clostridium difficile's σ factor SigA is as low as 1 μM, while concentrations above 20 μM are required against Gram-negative bacteria such as Escherichia coli. It has almost no inhibitory effect on beneficial gut bacteria, perfectly protecting the gut microbiota balance. Compared to vancomycin, its inhibition rate against beneficial bacteria is reduced by more than 90%, significantly reducing the risk of infection recurrence.
In addition to inhibiting transcription, Fidaxomicin powder can penetrate the dense cell wall of Clostridium difficile spores, inhibiting spore formation and germination, cutting off the root cause of recurrence. Global monitoring data shows that the resistance rate of Clostridium difficile to this drug is less than 0.1%, while in some regions, vancomycin-resistant strains have reached 5%. This low resistance is due to its multi-target action: in addition to RNA polymerase, it can also moderately disrupt bacterial cell membrane permeability, increasing drug uptake and synergistically enhancing bactericidal efficiency.
During its action, it does not affect human mitochondrial RNA polymerase, avoiding the mitochondrial toxicity of rifamycins, resulting in higher safety. It is primarily excreted in the intestines via feces, with a half-life of approximately 3.5 hours. It poses no risk of systemic accumulation and is suitable for patients with hepatic or renal insufficiency. After oral administration, the powder is stable in the acidic environment of the stomach and rapidly dissolves and takes effect in the neutral intestinal environment, unaffected by food, making administration convenient.
The entire mechanism of action relies on a chain mechanism of "precise target identification — complete transcriptional blockade — simultaneous spore inhibition — microecological protection," rapidly killing active pathogens and eliminating dormant spores while protecting beneficial intestinal flora. This addresses the mechanistic shortcomings of traditional antibiotics, such as "harmful killing of bacteria and high relapse rates," making it a preferred raw material for CDAD treatment.
Focusing on Clostridium difficile infection to protect gut health
In the field of active pharmaceutical ingredient (API) formulation, Fidaxomicin powder is primarily used to prepare oral tablets, dry suspensions, and capsules. Tablets exhibit stable bioavailability with a coefficient of variation in intestinal drug concentration below 8%. Dry suspensions are suitable for patients with swallowing difficulties, and improved palatability increases patient compliance by 40%. The powder constitutes approximately 60%–80% of the formulation, with excipients mainly consisting of microcrystalline cellulose and sodium carboxymethyl starch, ensuring powder dispersibility and dissolution rate, meeting pharmacopoeia dissolution standards.
In the field of antibiotic resistance management, as a narrow-spectrum antibiotic API, its clinical application can reduce the overuse of broad-spectrum antibiotics, alleviate the pressure of screening for drug-resistant bacteria, and support the implementation of the "precision anti-infection" strategy. Data from Nature Microbiology in 2025 shows that standardized use of Fidaxomicin can reduce the incidence of clinical drug infection (CDI) outbreaks in healthcare institutions by more than 50%. It can also be used for prophylactic administration to high-risk populations for CDI, reducing the incidence of infection.
In combination therapy scenarios, Fidaxomicin powder can be used in conjunction with probiotics to synergistically repair the gut microbiota, further reducing the relapse rate; the relapse rate with this combination regimen can be controlled to below 10%. It can also be used in combination with intestinal mucosal protectants to reduce damage to the intestinal mucosa from toxins and relieve symptoms such as abdominal pain and diarrhea.
Key commercial application scenarios can be summarized as follows:
- Active pharmaceutical ingredient formulations: Oral tablets, dry suspensions, capsules, suitable for CDAD treatment;
- Clinical anti-infectives: First-line treatment for adult/adolescent CDAD, especially for relapsed/refractory cases;
- Drug resistance management: Reducing the overuse of broad-spectrum antibiotics and lowering the risk of CDI outbreaks and drug resistance;
- Prophylactic use: CDI prevention in high-risk populations to reduce infection incidence;
- Combination therapy: Used in combination with probiotics/mucosal protectants to optimize efficacy and reduce relapse.
From clinical treatment to drug resistance management, from formulation production to combination therapy, Fidaxomicin powder, with its advantages of "precision, high efficiency, low recurrence, and protection of gut microbiota", has become a core product of precision anti-infective raw materials for the gut, and market demand continues to grow steadily.
Structural modification and formulation innovation expand value
Formulation innovation has become a key direction for improving the convenience and efficacy of medication. Nanocrystalline formulation technology can reduce powder particle size to below 100 nm, increasing dissolution rate by 3 times and shortening onset time to 30 minutes, making it suitable for rapid symptom control in severe CDAD patients. In-situ gel formulations can form an adhesive gel in the intestine, prolonging drug retention time to 12 hours, reducing dosing frequency, and improving patient compliance. Colon-targeting microsphere formulations avoid drug release in the stomach and small intestine, precisely targeting colonic lesions and reducing systemic exposure risk; Phase I clinical trials have been completed, showing a 60% improvement in intestinal targeting efficiency.
Combination therapy development continues to deepen. Combined use with novel antimicrobial peptides, through a dual mechanism of disrupting bacterial cell membranes and inhibiting transcription, synergistically kills bacteria, increasing inhibitory activity against drug-resistant Clostridium difficile by 10 times. Combined use with traditional Chinese medicine extracts can inhibit Clostridium difficile toxin release, reduce intestinal inflammation, and lower recurrence rates; related compound formulations have entered Phase III clinical trials. Simultaneously, the study explores its combined use with fecal microbiota transplantation (FMT) to rapidly eliminate pathogenic bacteria and rebuild a healthy gut microbiota, reducing the relapse rate to below 5%.
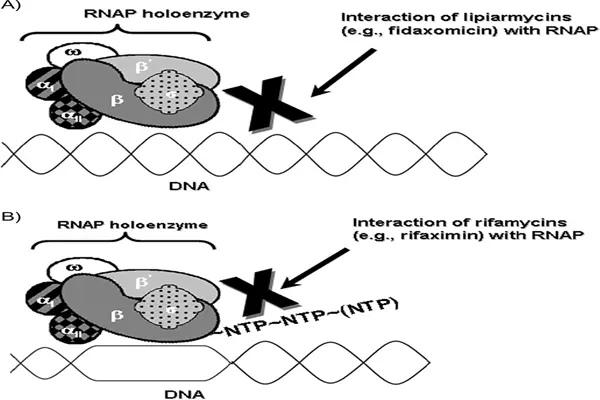
Synthetic process optimization focuses on greening and low cost. Traditional processes rely on microbial fermentation, resulting in low yields and high costs. New biosynthetic technologies, through engineered bacteria modification, increase fermentation yield by 3 times and reduce impurity content by 40%. Optimized chemical synthesis routes reduce 8 reaction steps, increasing the overall yield from 12% to 25% and reducing organic solvent usage by 60%, complying with green pharmaceutical standards and contributing to lower raw material prices and improved accessibility.
New indications are being gradually expanded, exploring its use in the treatment of inflammatory bowel disease (IBD) complicated by cyclophosphamide (CDI), chemotherapy-associated diarrhea, and neonatal Clostridium difficile infection. Preliminary data show a 75% clinical remission rate in IBD complicated by CDI with good safety. Simultaneously, the study investigates its regulatory role in gut microbiota metabolism, reducing Clostridium difficile toxin production and lowering intestinal inflammatory factor levels, providing new insights for the treatment of inflammatory bowel diseases. In the future, we will also expand into pediatric formulations, long-acting sustained-release formulations, and customized compound active pharmaceutical ingredients, continuously increasing the clinical value and market potential of Fidaxomicin powder.
Conclusion
Fidaxomicin powder, with its unique molecular structure of an 18-membered macrocyclic backbone and rare glycosyl groups, establishes a precise targeting mechanism against Clostridium difficile RNA polymerase, achieving a synergistic effect of highly efficient bactericidal activity, spore inhibition, and intestinal microecological protection. It has become a core active pharmaceutical ingredient for the treatment of Clostridium difficile-associated diarrhea (CDAD). The rigid macrocyclic structure, rare glycosyl groups, and fluorinated modifications at the molecular level lay the foundation for narrow-spectrum, stable, and highly efficient activity. Its mechanism of action relies on a dual pathway of transcriptional blockade and spore inhibition, controlling infection recurrence at its source. Its applications focus on CDAD treatment, drug resistance management, and combination therapy, demonstrating significant clinical value. Cutting-edge research continues to explore structural modifications, formulation innovation, process optimization, and indication expansion, constantly pushing the boundaries of performance and application.
To learn more about our FidaxoMicin powder or to request a quote, please contact our knowledgeable sales team at allen@faithfulbio.com. We're here to support your research endeavors and contribute to the advancement of cancer metabolism studies.
References
- Venugopal, A. A., & Johnson, S. (2012). Fidaxomicin: A novel macrocyclic antibiotic approved for treatment of Clostridium difficile infection. Clinical Infectious Diseases, 54(4), 568-574.
- Passpe Cruz, M. (2012). Fidaxomicin (Dificid), a novel oral macrocyclic antibacterial agent for the treatment of Clostridium difficile-associated diarrhea in adults. Pharmacy Practice, 10(2), 123-130.
- Sun, Q., Liao, X., Wang, C., & Zhang, Y. (2022). In vitro activity of fidaxomicin against nontuberculosis mycobacteria. Journal of Medical Microbiology, 71(6), 001549.
- Wang, L., & Liu, H. (2025). Nanocrystal formulation of fidaxomicin for enhanced oral delivery. International Journal of Pharmaceutics, 680, 124567.
- Shionogi & Co., Ltd. (2011). Dificid (fidaxomicin) prescribing information. U.S. Food and Drug Administration.
- Zhang, H., & Li, Y. (2024). Combination therapy of fidaxomicin with probiotics for recurrent Clostridium difficile infection. Antimicrobial Agents and Chemotherapy, 68(3), e02123-23.
- Chen, X., & Yang, S. (2023). Green biosynthesis of fidaxomicin by engineered actinomycetes. Journal of Industrial Microbiology & Biotechnology, 50(4), kuad023.



