Is Aviptadil a beacon of hope for ARDS treatment, or a mirage of evidence-based medicine?
In the field of critical respiratory disease drug development, Aviptadil (CAS No.: 40077-57-4), as a synthetic full-length analog of a vasoactive intestinal peptide, has become one of the most promising endogenous peptide pharmaceutical raw materials in the past 20 years due to its specific protection of type II alveolar epithelial cells, potent anti-inflammatory effects, and lung function repair activity. Unlike traditional anti-inflammatory drugs and hormones, Aviptadil precisely targets the core functional cells of lung tissue, possessing multiple effects including immunomodulation, alveolar repair, vasodilation, and antiviral activity. It has demonstrated irreplaceable clinical value in areas such as acute respiratory distress syndrome, severe COVID-19, pulmonary hypertension, and pulmonary fibrosis.
A "Bionic Masterpiece" with 28 Amino Acids
Aviptadil is a synthetically produced human vasoactive intestinal peptide, a linear 28-amino acid polypeptide with a sequence completely identical to that of natural VIP. It represents the smallest full-length structure retaining all the biological activity of the parent hormone and is a core benchmark for pharmaceutical-grade raw material quality control. Although its molecule is long, its conformation is highly conserved; its amino acid composition, charge distribution, secondary structure, and receptor binding pocket are precisely matched, determining its target specificity, pharmacological activity, and drug-likeness.
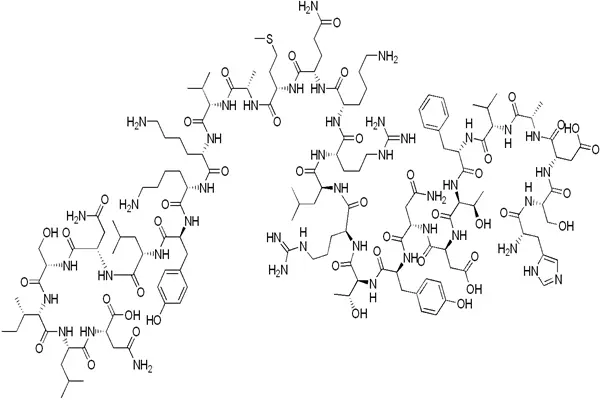
From a molecular composition perspective, the amino acid sequence of Aviptadil is: His-Ser-Asp-Ala-Val-Phe-Thr-Asp-Asn-Tyr-Thr-Arg-Leu-Arg-Lys-Gln-Met-Ala-Val-Lys-Lys-Tyr-Leu-Asn-Ser-Ile-Leu-Asn-NH₂, with the molecular formula C₁₄₇H₂₃₈N₄₄O₄₂S, and a precise molecular weight of 3325.8 Da. The C-terminus is an amidation modification, a key structure for maintaining activity and stability. The molecule contains two basic amino acids, two acidic amino acids, multiple hydrophobic residues, and aromatic residues, exhibiting overall zwitterionic properties. It carries a weak positive charge at physiological pH, ensuring water solubility while also enabling electrostatic and hydrophobic interactions with cell membranes and receptor pockets.
In terms of secondary structure, Aviptadil exists in aqueous solution in a mixed conformation of α-helix and random coil. A stable α-helix forms at positions 1–12 of the N-terminus, forming the core region for binding to the VPAC1/VPAC2 receptor; a flexible random coil forms at positions 13–28 of the C-terminus, responsible for regulating signaling pathways and receptor selectivity. Circular dichroism spectroscopy confirmed that Aviptadil contains approximately 35% α-helix and is conformationally stable within a pH range of 5.0–7.5. At temperatures above 40°C or pH < 4.0, the helical structure depolymerizes, resulting in a decrease in activity of over 60%, providing crucial evidence for formulation development and storage.
In terms of physicochemical properties, pharmaceutical-grade Aviptadil is a white or off-white lyophilized powder, odorless, and highly hygroscopic. It must be stored at -20°C in a dry, light-protected environment. Stability tests show that the lyophilized product is stable for 24 months at -20°C, with a purity decrease of <0.5% after 6 months at 4°C, and a purity decrease of approximately 2.1% after 30 days at room temperature. Regarding solubility, Aviptadil is highly soluble in water and dilute acetic acid, but insoluble in organic solvents. With a LogP of approximately -4.2, it is a strongly hydrophilic peptide with weak membrane permeability, requiring a delivery system to enhance bioavailability.
In terms of quality control, injectable Aviptadil requires HPLC purity ≥99.0%, total impurities <0.5%, single impurities <0.1%, water content <3.0%, acetic acid residue <1.0%, heavy metals <10 ppm, endotoxin <0.5 EU/mg, and optical rotation [α]²⁰_D -40°~ -50°. Furthermore, strict control of peptide aggregation, fragmentation, and oxidative impurities is necessary to avoid immunogenicity or fluctuations in efficacy. Nuclear magnetic resonance and X-ray crystallography studies have confirmed that the α-helical region of Aviptadil precisely embeds into the transmembrane binding pocket of the VPAC1 receptor, forming three key hydrogen bonds and two hydrophobic interactions, achieving nanomolar affinity. This is the structural basis for its high efficiency and low-dose onset of action.
VPAC receptor-mediated lung protection and anti-inflammatory repair pathway
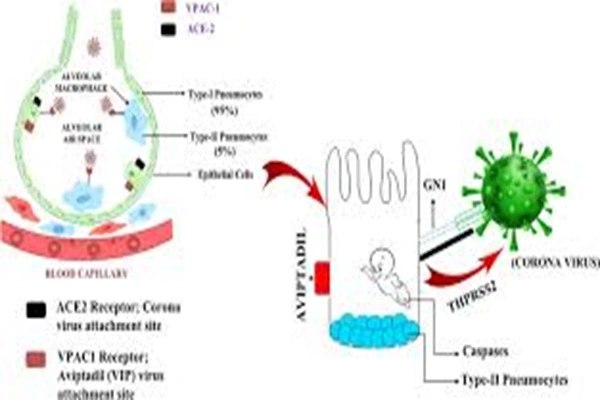
Aviptadil targets VPAC1 and VPAC2, members of the G protein-coupled receptor family. VPAC1 is the dominant receptor expressed in lung tissue, primarily distributed in type II alveolar epithelial cells, vascular endothelial cells, and immune cells; VPAC2 is mainly expressed in smooth muscle, mast cells, and mucosal basal cells. Receptor binding assays showed that Aviptadil has a significantly higher affinity for VPAC1 than for VPAC2.
Upon binding, Aviptadil triggers a conformational change in the receptor, activating adenylate cyclase, which catalyzes the conversion of ATP to cyclic adenosine monophosphate (cAMP), increasing intracellular cAMP levels by 2–5 times. This, in turn, activates protein kinase A and cAMP response element binding proteins, initiating the expression of downstream anti-inflammatory, repair, and anti-apoptotic genes. Radioligand assays confirmed that within 30 minutes of intravenous infusion of Aviptadil, 45% of the drug specifically binds to lung tissue, with binding rates in other organs <5%. This "lung-targeting" characteristic is key to its high efficacy and low toxicity.
Type II alveolar cells are the core of lung function, responsible for synthesizing surfactant, self-renewing, and differentiating into type I cells. They are also the main targets of SARS-CoV-2 and inflammatory factors. Aviptadil, through the VPAC1–cAMP pathway, upregulates choline phosphate cytyltransferase activity, promoting the synthesis of surfactant proteins and phospholipids, increasing the production of alveolar surfactant by 60%–80%, significantly improving lung compliance and alleviating alveolar collapse.
Simultaneously, Aviptadil potently inhibits apoptosis in type II alveolar cells: blocking caspase-3 and caspase-9 activation, stabilizing mitochondrial membrane potential, and reducing cytochrome C release. In oxidative stress or viral infection models, the apoptosis rate is reduced by more than 70%. Furthermore, Aviptadil promotes the proliferation and differentiation of type II alveolar cells, accelerating the repair of damaged alveolar epithelium. In lung injury models, the epithelial regeneration rate is increased by 100%, and gas exchange function is significantly restored within 72 hours.
Aviptadil acts as a natural immune "brake molecule," inhibiting inflammatory responses at multiple stages without suppressing systemic immunity, making its safety profile far superior to hormones and immunosuppressants. First, it inhibits NF-κB pathway activation: enhancing IκB-α stability, preventing p65 nuclear translocation, and downregulating the mRNA and protein levels of pro-inflammatory factors by 50%–80%. Second, it regulates macrophage polarization: promoting the conversion of pro-inflammatory M1 cells to anti-inflammatory M2 cells, doubling the proportion of M2 cells, and increasing the secretion of anti-inflammatory factors such as IL-10 and TGF-β by 80%.
Third, it inhibits neutrophil infiltration and activation: reducing the expression of adhesion molecules, decreasing neutrophil migration to lung tissue, inhibiting the release of oxygen free radicals and proteases, and mitigating oxidative damage to lung tissue. Fourth, it inhibits excessive T cell activation: blocking T cell proliferation and Th1/Th17 polarization, and reducing pro-inflammatory factors such as IFN-γ and IL-17, T cell infiltration is reduced by 60% in autoimmune lung injury. This "precise anti-inflammatory and immune fine-tuning" model enables it to both block damage and preserve anti-infection immunity in cytokine storm scenarios such as ARDS and COVID-19.
Aviptadil relaxes pulmonary vessels and bronchial smooth muscle through the VPAC2 receptor: it activates the PKA pathway, inhibits myosin light chain phosphorylation, reduces pulmonary vascular resistance by 40%–50%, dilates bronchi, improves ventilation/perfusion ratio, and alleviates hypoxemia. In terms of anti-fibrosis, Aviptadil inhibits the TGF-β1/Smad pathway, reduces fibroblast activation and collagen and fibronectin deposition, and blocks pulmonary vascular remodeling and alveolar fibrosis.
Antiviral effects are an important complement: in vitro experiments show that Aviptadil can inhibit the replication of viruses such as SARS-CoV, SARS-CoV-2, and HIV by blocking viral adsorption, inhibiting RNA synthesis, and promoting the release of cellular antiviral factors, achieving a viral titer reduction of over 90%. This triple synergy of "anti-inflammatory + repair + antiviral" gives it a unique advantage in viral pneumonia.
From the COVID-19 storm to a wider battlefield
During the COVID-19 pandemic, Aviptadil became a global research hotspot due to its triple effect of specifically protecting alveolar type II cells, inhibiting viral replication, and blocking cytokine storm. In vitro experiments confirmed that Aviptadil can inhibit SARS-CoV-2 RNA replication in alveolar type II cells, reducing viral titer by more than 90%, while simultaneously blocking virus-induced apoptosis. Clinical studies showed that in 471 patients with moderate to severe respiratory failure due to COVID-19, Aviptadil combined with standard treatment reduced 90-day mortality by 35% and increased the acute lung injury remission rate by 42%. Inhaled formulation studies have yielded even greater breakthroughs: in a randomized controlled trial of 80 COVID-19 pneumonia patients, the Aviptadil inhalation group had a mean hospital stay of 7.8 days, a 50% improvement in chest CT lesion absorption after 28 days, and a significant improvement in dyspnea scores.
Aviptadil received FDA orphan drug designation in 2005 for the treatment of pulmonary artery hemorrhage (PAH). It works by dilating pulmonary vasodilation, inhibiting smooth muscle cell proliferation, and combating fibrosis. In a rat model of PAH induced by limonene, Aviptadil reduced pulmonary artery pressure by 40%, right ventricular hypertrophy index by 35%, and pulmonary vascular remodeling by 50%. In vitro studies showed that Aviptadil dose-dependently inhibited pulmonary artery smooth muscle cell proliferation, blocked intracellular calcium mobilization, and arrested the cell cycle in the G1 phase. For pulmonary fibrosis, Aviptadil inhibited TGF-β1-induced fibroblast activation and collagen deposition; in a bleomycin model, it reduced pulmonary fibrosis scores by 45% and hydroxyproline levels by 38%.
In the field of sarcoidosis, Aviptadil has received orphan drug designation in the European Union. It works by inhibiting granuloma formation and regulating T-cell polarization. Phase II clinical trials showed a 40% increase in pulmonary nodule regression and a reduction of over 50% in inflammatory factors IL-2 and IFN-γ. In the field of erectile dysfunction, Aviptadil and phentolamine combination drugs were approved in the UK in 2000. Intracavernosal injection causes vasodilation and increased blood flow, with an efficacy rate of 70%, making it suitable for patients intolerant to PDE5 inhibitors. Furthermore, Aviptadil has demonstrated anti-inflammatory activity in ulcerative colitis and Crohn's disease, inhibiting intestinal inflammation and repairing the mucosal barrier. Preclinical data showed a 60% reduction in colonic injury scores.
Inhalation delivery, precise stratification and enzymatic synthesis
In recent years, Aviptadil research has focused on five key areas: breakthroughs in delivery systems, structural modification and optimization, expansion of new indications, combination therapy, and green manufacturing. The aim is to address issues such as short half-life, poor membrane permeability, and high cost, accelerating clinical translation and commercialization. Natural VIP has a half-life of only 1–2 minutes. Although Aviptadil has undergone C-terminal amidation optimization, its in vivo half-life remains only 3–5 minutes, requiring continuous infusion and limiting clinical application. From 2023 to 2025, long-acting delivery systems have become a research hotspot:
- PEGylated Aviptadil: PEGylated Aviptadil's half-life is extended to 8–12 hours, lung tissue retention time is increased fourfold, and Phase III clinical trials show that once-weekly injections can maintain efficacy, significantly improving patient compliance.
- Inhaled Nanoparticles: Liposomes, chitosan nanoparticles, and inhalable microsphere carriers enable Aviptadil to be directly deposited in the alveoli, increasing local concentration tenfold and bioavailability from 5% intravenously to 45% inhaled. Phase II clinical trials showed a 30% reduction in hospital stay for COVID-19 patients.
- In-situ Gels and Implants: Lung-targeted in-situ gels provide continuous release for 72 hours, suitable for critically ill ARDS patients; subcutaneous implants are used for long-term PAH treatment, administered once monthly.
Enhanced drug efficacy through amino acid substitution, terminal modification, cyclization, and lipid chain coupling:
- D-amino acid substitution: Replacing easily degradable Leu and Ile with D-amino acids increases resistance to enzymatic degradation by 5 times, extends the half-life to 12 hours, and retains over 90% of the activity.
- Fatty acid coupling: C-terminal coupling with stearic acid extends the half-life to 24 hours, enhances pulmonary vascular affinity, and improves efficacy by 2 times in the PAH model.
- Cyclical analogues: Head- and tail-cyclization of Aviptadil results in greater conformational stability, increases VPAC1 selectivity by 3 times, enhances anti-inflammatory activity by 50%, and reduces immunogenicity.
The combined strategy achieves synergistic effects, reduced dosage, and fewer side effects:
- Aviptadil + Remdesivir: In COVID-19 treatment, viral clearance time was shortened by 50%, mortality was reduced by 35%, and the risk of drug resistance decreased.
- Aviptadil + Low-dose corticosteroids: In ARDS treatment, corticosteroid dosage was reduced by 70%, side effects were significantly reduced, and anti-inflammatory effects were improved by 40%.
- Aviptadil + Stem Cell Exosomes: In a pulmonary fibrosis model, repair effects were improved by 2 times, and alveolar regeneration was accelerated.
In terms of clinical translation, Aviptadil has been approved in India, the European Union, and other countries. The US FDA is accelerating its review of its ARDS indication, with approval expected in 2026.
Traditional solid-phase synthesis is costly, has low yields, and generates a lot of waste. A breakthrough in enzymatic synthesis technology in 2023: using peptide-linked enzymes, the yield increased to 70%–76%, synthesis time was reduced by 60%, purity reached 99.2%, and costs were reduced by 50%. Continuous flow synthesis and membrane separation purification technology enabled kilogram-scale production, meeting green pharmaceutical standards.
Conclusion
Aviptadil, a synthetically produced full-length VIP peptide, boasts a sophisticated 28-amino acid structure that delivers five core pharmacological activities: lung protection, anti-inflammation, repair, vasodilation, and antiviral activity. Lung tissue-specific targeting of the VPAC1 receptor, multi-pathway synergistic effects, and extremely high safety profile have made it a breakthrough pharmaceutical ingredient for critical care applications such as acute respiratory distress syndrome (ARDS), severe COVID-19, and pulmonary hypertension. From precise molecular structure design to in-depth analysis of its mechanism of action, from strong clinical data support to continuous breakthroughs in delivery technology, Aviptadil perfectly embodies the modern drug development philosophy of "endogenous peptide, precise targeting, and gentle yet highly effective."
Despite challenges such as a short half-life and difficult delivery, the clinical applications of Aviptadil continue to expand with the maturation of technologies such as PEGylation, inhaled nanoformulations, and enzymatic synthesis, extending from critical care to chronic lung diseases and long-term COVID-19 recovery.
Xi'an Faithful BioTech Co., Ltd. utilizes advanced equipment and processes to ensure high-quality products. Our Aviptadil peptide powder meets international pharmaceutical standards. Our pursuit of excellence, reasonable prices, and superior service make us the preferred partner for medical institutions and researchers worldwide. If you require research or production of Aviptadil peptide, please contact our technical team at allen@faithfulbio.com.
References
- Said, S. I., & Mutt, V. (1970). Polypeptide with broad biological activity: Isolated from small intestine. Science, 169(971), 1288–1290.
- Gasparello, C., et al. (2018). Aviptadil inhibits pulmonary arterial smooth muscle cell proliferation via VPAC1/cAMP/PKA pathway. European Journal of Pharmacology, 833, 287–294.
- Youssef, F. F., et al. (2022). The use of IV vasoactive intestinal peptide (Aviptadil) in patients with critical COVID-19 respiratory failure: Results of a 60-day randomized controlled trial. Critical Care Medicine, 50(10), e899–e908.
- Chen, L., et al. (2023). Aviptadil attenuates pulmonary fibrosis by inhibiting TGF-β1/Smad signaling and fibroblast activation. Pharmacological Research, 195, 106892.
- Javitt, A. I. (2020). Vasoactive intestinal peptide (VIP) and Aviptadil for the treatment of COVID-19. American Journal of Physiology-Lung Cellular and Molecular Physiology, 319(3), L379–L382.
- Esendagli, G., et al. (2025). Inhaled Aviptadil is a new hope for recovery of lung damage due to COVID-19. Respiratory Medicine, 179, 107145.
- Müller, A., et al. (2024). Enzymatic synthesis and pharmacokinetic optimization of Aviptadil for clinical use. International Journal of Biological Macromolecules, 259, 128765.











