Can KPV tripeptide become a "Swiss Army knife" for inflammatory bowel disease and skin repair?
In the field of bioactive peptides and pharmaceutical raw materials, KPV peptide powder is a highly representative endogenous tripeptide. As the C-terminal core active fragment of α-melanocyte-stimulating hormone, it retains most of the anti-inflammatory, immunomodulatory, and tissue repair activities of the parent hormone, but without hormone-related side effects, exhibiting extremely high safety. Unlike artificially designed cosmetic peptides, KPV originates from the body's own metabolites, possessing multiple advantages such as natural biocompatibility, low immunogenicity, and clear target sites. In recent years, it has received widespread attention in pharmaceutical and clinical translational applications related to inflammatory bowel disease, skin inflammation, neuroprotection, and mucosal repair.
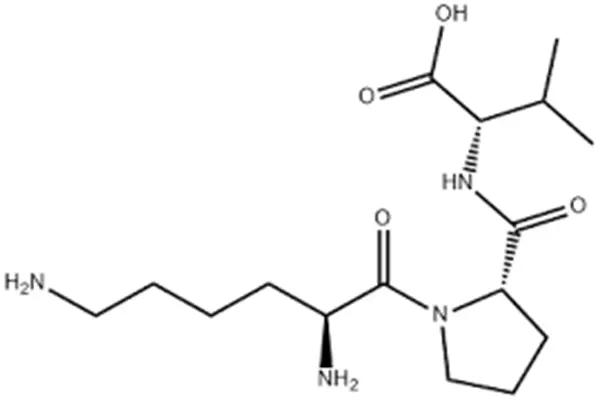
The "minimum active fragment" of α-MSH
The chemical structure of KPV can be summarized in one sentence: H-Lys-Pro-Val-OH—a linear tripeptide composed of lysine, proline, and valine linked by peptide bonds. Let's break down the chemical features of this sequence:
- 1. Lysine (K)—The Positively Charged "Anchor": The lysine side chain contains an ε-amino group, which carries a positive charge at physiological pH. This charge is crucial for the initial electrostatic interaction of KPV with the cell membrane or receptor. More importantly, this amino group is a "golden site" for chemical modification—researchers can use it to connect polyethylene glycol, glycosyl groups, or other functional groups to improve the pharmacokinetic properties of KPV.
- 2. Proline (P)—The Conformational "Turning Point": Proline is the only amino acid whose side chain forms a ring with the main chain. Its structure gives the peptide chain a "turning point," limiting the overall conformational flexibility. In KPV, the presence of proline makes this tripeptide more "compact," potentially affecting its binding to target proteins.
- 3. Valine (V) – The Hydrophobic "Tail" The side chain of valine is isopropyl, which is hydrophobic. This hydrophobic "tail" may help KPV embed into the hydrophobic region of the cell membrane or interact with the hydrophobic pocket of target proteins.
With the molecular formula C₁₆H₃₀N₄O₄ and a molecular weight of approximately 342.43 g/mol, both the N-terminus and C-terminus are in a free state. This gives KPV good solubility in aqueous solution, but also makes it easily degraded by exopeptidases (aminopeptidases and carboxypeptidases), which is the fundamental reason for its short half-life in vivo.
In terms of physicochemical properties, KPV is typically a white or off-white lyophilized powder, odorless, and highly hygroscopic, therefore it must be stored under low temperature, dry, and light-protected conditions. KPV is highly soluble in water, with a solubility >50 mg/mL at 25°C, making it ideal for use in aqueous solutions, eye drops, enemas, and other hydrophilic systems. Its LogP is approximately -3.0, classifying it as a strongly hydrophilic peptide; therefore, in transdermal applications, delivery systems such as liposomes, microemulsions, and nanocarriers are often used to enhance penetration efficiency. Regarding pH stability, KPV remains stable within a pH range of 4.0–7.0, suitable for administration under physiological conditions. It is prone to peptide bond hydrolysis under strong acid or alkaline conditions; therefore, the pH of formulations is generally controlled between 5.0–6.5. Accelerated stability tests show that after 30 days at 40°C and 75% humidity, the purity of KPV decreases by no more than 1.2%, and the lyophilized product can be stably stored at -20°C for at least 24 months, meeting the long-term storage requirements for pharmaceutical raw materials.
The relationship between structure and activity is central to the pharmaceutical value of KPV. Studies have shown that any substitution of amino acids, change in chirality, or modification of terminal groups significantly affects activity. For example, replacing L-proline with glycine increases molecular flexibility but reduces binding affinity to receptors such as MC1-R and MC3-R by more than 70%; N-terminal acetylation improves stability but reduces anti-inflammatory activity by about 40%; C-terminal amidation increases mucosal retention time but slightly weakens the inhibitory effect on intestinal inflammation. This means that the natural configuration L-Lys-L-Pro-L-Val is the optimal structure for activity and is a core indicator that must be strictly controlled for pharmaceutical raw materials. Regarding purity control, pharmaceutical-grade KPV requires HPLC purity ≥98.5%, total impurities <1.0%, single impurities <0.2%, and optical rotation, water content, heavy metals, and endotoxins must all meet the standards for injectable raw materials to avoid impurities triggering immune responses or inflammatory interference.
Anti-inflammatory mechanisms that do not follow the "usual route"
KPV's core targets are the melanocortin receptor family, particularly MC1-R, MC3-R, and MC4-R, with MC3-R considered the primary mediator of its anti-inflammatory activity. As the active fragment of α-MSH, KPV binds to MC3-R with high affinity and selectivity, triggering a conformational change in the receptor, activating the intracellular cAMP/PKA signaling pathway, and initiating downstream anti-inflammatory processes. Unlike intact α-MSH, KPV's activation of MC1-R is weaker, therefore it does not significantly promote melanin synthesis and does not cause skin pigmentation, which is a significant clinical advantage. Receptor binding assays show that KPV's IC₅₀ for MC3-R is at the nanomolar level, achieving saturation binding at low concentrations and achieving highly effective anti-inflammatory effects.
The most crucial molecular mechanism of KPV is the inhibition of NF-κB signaling pathway activation. NF-κB is a core transcription factor in the inflammatory response. Once activated by LPS, TNF-α, IL-1β, etc., it rapidly enters the nucleus and initiates the expression of numerous pro-inflammatory genes. KPV enhances the stability of IκB-α protein, preventing phosphorylation and nuclear translocation of the NF-κB p65 subunit, thereby blocking the inflammatory cascade. Cellular experiments show that in LPS-stimulated macrophages, KPV can reduce p65 nuclear translocation levels by approximately 60%, significantly reduce inflammatory mediators such as NO, PGE2, and iNOS, and downregulate IL-6 and TNF-α mRNA levels by 40%–70%. This broad-spectrum ability to inhibit central inflammatory pathways makes it effective against various inflammatory diseases.
KPV can also regulate macrophage polarization, promoting the transformation of pro-inflammatory M1 macrophages into anti-inflammatory M2 macrophages. M1 macrophages release large amounts of inflammatory factors, exacerbating tissue damage; while M2 macrophages release anti-inflammatory factors such as IL-10 and TGF-β, promoting repair. In an in vitro macrophage polarization model, KPV nearly doubled the proportion of CD206+ M2 neutrophils and increased IL-10 secretion by approximately 80%, while simultaneously reducing the proportion of iNOS+ M1 neutrophils. This mechanism enables it to not only "anti-inflammate" but also "promote repair," making it particularly important in chronic inflammation and wound healing.
Furthermore, KPV inhibits neutrophil infiltration and activation, reduces the release of oxygen free radicals and proteases, thereby alleviating tissue oxidative damage. At sites of inflammation, a large accumulation of neutrophils leads to tissue destruction, edema, and pain. KPV can reduce the expression of adhesion molecules ICAM-1 and VCAM-1, and decrease leukocyte migration, thus rapidly relieving redness, swelling, heat, and pain. Simultaneously, KPV can promote the proliferation of epithelial cells and fibroblasts, accelerate angiogenesis and collagen deposition, enhance tissue strength, and reduce excessive scar proliferation.
Compared to synthetic anti-inflammatory drugs, KPV's mode of action is closer to physiological regulation, belonging to "immune fine-tuning" rather than "immune suppression," and does not cause side effects such as increased infection risk, gut microbiota dysbiosis, or metabolic abnormalities. Its pathway is clear, its target is well-defined, and its signal is controllable, providing a solid theoretical foundation for formulation development, dosage optimization, and combination therapy, making it one of the most transparently mechanistic bioactive peptides among pharmaceutical raw materials.
A versatile agent for everything from inflammatory bowel disease to skin repair.
In the field of inflammatory bowel disease, kappain vaporization (KPV) is currently the most extensively researched and data-rich area, including Crohn's disease and ulcerative colitis. Numerous animal models have shown that KPV can significantly reduce colonic inflammation scores, decrease mucosal damage, and inhibit pro-inflammatory cytokine storms. In a mouse model of DSS-induced acute colitis, intraperitoneal injection or enema administration of KPV can restore more than 60% of colonic shortening, reduce myeloperoxidase activity by 50%–70%, and reduce histological damage scores by approximately 65%. Compared with traditional 5-ASA, hormones, and biologics, KPV does not disrupt the gut microbiota, does not suppress systemic immunity, and has no significant gastrointestinal irritation, making it ideal as a raw material for maintenance or adjuvant therapy. Some preclinical translational studies have entered Phase I clinical trials for local enema treatment of patients with mild to moderate ulcerative colitis, demonstrating good mucosal repair effects and tolerability.
Skin inflammation and wound repair are another important application areas for KPV, including atopic dermatitis, psoriasis, contact dermatitis, burns, and chronic, non-healing wounds. KPV can simultaneously inhibit inflammation, reduce redness and itching, accelerate epithelial regeneration, and promote orderly collagen arrangement, without producing hormone dependence. In a mouse xylene-induced ear swelling model, topical application of 0.1% KPV gel reduced ear swelling by 42%, and decreased IL-6 and TNF-α levels by 38% and 45%, respectively. In a diabetic mouse wound model, the wound healing time in the KPV-treated group was shortened from 18 days to 11 days, neovascularization density increased by nearly 100%, and epithelialization rate was significantly improved. Due to its endogenous properties, it has extremely low skin irritation and no sensitization with repeated administration, making it suitable for development as a dermatological prescription drug or a raw material for Class II medical device dressings.
Lung and respiratory tract inflammation is also an important potential indication for KPV, including acute lung injury, asthma, and chronic obstructive pulmonary disease. In a mouse model of endotoxin-induced acute lung injury, KPV intervention significantly reduced inflammatory cell infiltration in bronchoalveolar lavage fluid, decreased protein exudation, and inhibited pro-inflammatory mediators such as IL-1β, TNF-α, and CXCL1, resulting in a decrease of approximately 60% in lung tissue pathological damage scores. Its mechanism of action leans towards regulating local lung immunity without affecting airway smooth muscle contraction; therefore, it can be used as a raw material for inhaled formulations for adjunctive intervention in severe lung inflammation.
Neuroprotection and central anti-inflammatory effects are rapidly emerging applications in recent years. KPV can cross the blood-brain barrier or be administered intrathecally, inhibiting excessive microglial activation and reducing neuroinflammation. It has shown protective effects against stroke, Parkinson's disease, Alzheimer's disease, and spinal cord injury. In a rat model of middle cerebral artery ischemia, KPV reduced infarct volume by approximately 35%, improved neurological deficit scores, and inhibited microglial polarization towards a pro-inflammatory phenotype. In addition, KPV can be used for ocular inflammation, such as keratitis, uveitis, and dry eye syndrome. It can relieve ocular surface inflammation and repair corneal epithelium without side effects such as mydriasis or increased intraocular pressure, making it suitable for development into eye drops.
KPV also shows broad potential in the fields of autoimmune diseases and chronic low-grade inflammation, including rheumatoid arthritis, multiple sclerosis, and metabolic-related inflammation. Animal experiments have shown that KPV can inhibit joint swelling, reduce synovial inflammation, and decrease osteoclast activation. In an experimental autoimmune encephalomyelitis model, KPV can delay onset and reduce demyelinating damage. Furthermore, KPV can be used for mucosal repair scenarios such as oral mucosal ulcers, gastric mucosal damage, and bladder mucosal inflammation, achieving tissue protection by rapidly inhibiting local inflammation and accelerating epithelial reconstruction. Overall, the applications of KPV are highly focused on the "inflammation-immunity-repair" axis, with highly concentrated indications and a clear drug development pathway, making it a highly valuable endogenous bioactive peptide raw material for the pharmaceutical industry.
Delivery technology revolution and mechanism deepening
In the development of delivery systems, KPV are highly hydrophilic, have limited permeability, and are easily degraded by enzymes, making nanodelivery systems a research hotspot. Multiple studies from 2023–2025 have shown that carriers such as liposomes, chitosan nanoparticles, hyaluronic acid microgels, and in-situ gels can significantly improve the local retention time and bioavailability of KPVs. pH-sensitive colon-targeting microspheres for ulcerative colitis enable specific drug release in the colon, increasing bioavailability by 4–6 times; liposomes and nanoemulsion carriers for skin can increase transdermal absorption by nearly 10 times; inhaled lipid microspheres can improve lung deposition rates and reduce dosing frequency. These breakthroughs in delivery systems have greatly promoted the clinical application potential of KPVs.
Structural modification and analogue development are important directions for improving drugability. Researchers have improved the half-life, lipophilicity, and receptor selectivity of KPVs through N-terminal acetylation, C-terminal amidation, D-amino acid substitution, and fatty acid coupling. For example, C-terminal amidation yields KPV-NH₂, which enhances mucosal adhesion and anti-inflammatory activity; lipid chain modification prolongs the in vivo half-life from minutes to hours; cyclized KPV analogs exhibit more stable conformations, stronger receptor selectivity, and approximately 2–3 times improved anti-inflammatory efficacy. These modifications do not alter the core pharmacophore, maintaining a high level of safety, making them suitable for development into long-acting formulations.
New indications and clinical translation continue to expand. In addition to IBD and skin inflammation, research on KPV in neurodegenerative diseases, ocular inflammation, acute kidney injury, and graft-versus-host disease is rapidly increasing. Preclinical data show that KPV can reduce renal ischemia-reperfusion injury and lower serum creatinine and blood urea nitrogen levels; in the eye, KPV can inhibit corneal neovascularization and inflammation, making it suitable for treating viral keratitis and dry eye. Some institutions have initiated Phase I clinical trials to evaluate the safety, tolerability, and pharmacokinetic characteristics of intravenous and topical enema KPV, laying the foundation for Phase II proof-of-concept trials. Combination therapy strategies are emerging as a new trend. KPV, when used in combination with low-dose hormones, 5-ASA, probiotics, and mesenchymal stem cell exosomes, can achieve synergistic effects while reducing the dosage of the main drug and side effects. For example, the combination of KPV and low-concentration dexamethasone for treating colitis significantly enhances anti-inflammatory effects while markedly reducing hormone-related adverse reactions; combined with probiotics, it can repair the intestinal barrier and improve the gut microbiota structure, making it suitable for long-term management.
Regarding industrialization and green preparation, traditional liquid-phase and solid-phase synthesis are costly. In recent years, the gradual application of enzymatic synthesis, continuous flow synthesis, and green purification technologies has reduced the cost of KPV raw materials, improved purity, and reduced waste. Novel membrane separation and countercurrent chromatography technologies can achieve kilogram-scale preparation with pharmaceutical-grade purity consistently above 99%, providing support for subsequent commercial production.
Conclusion
KPV, an endogenous tripeptide, possesses a minimalist Lys-Pro-Val structure that carries powerful and specific anti-inflammatory, immunomodulatory, and tissue repair activities. With its clear target, well-defined mechanism, high safety profile, and excellent biocompatibility, it demonstrates irreplaceable value in pharmaceutical fields such as inflammatory bowel disease, skin inflammation, neuroprotection, and mucosal repair. Its stable molecular conformation and excellent drug-like properties allow for targeted drug delivery through various delivery systems, avoiding the widespread side effects of hormones and exhibiting superior tolerability compared to traditional small-molecule anti-inflammatory drugs. It is an ideal active ingredient bridging endogenous regulation and modern pharmaceuticals.
From its basic structure to its pathway of action, from disease application to cutting-edge translation, the research system for KPV is maturing, with continuous breakthroughs in delivery technology and structural modification, and a rapidly accelerating pace of clinical translation. In the future, with formulation optimization, clinical data accumulation, and large-scale production, KPV is expected to become the first successfully marketed endogenous anti-inflammatory tripeptide drug, providing a novel treatment option for chronic inflammation, autoimmune diseases, and tissue repair.
Xi'an Faithful BioTech Co., Ltd. is ready to meet your KPV peptide powder sourcing needs with pharmaceutical-grade quality, competitive pricing, and superior service standards. Our experienced team provides customized solutions and comprehensive technical and regulatory support to pharmaceutical companies seeking reliable, high-quality API supplies. Contact allen@faithfulbio.com today for detailed product specifications, competitive quotes, and samples tailored to your specific formulation requirements. As a trusted KPV peptide powder supplier, we are committed to providing the quality, stability, and partnerships necessary for successful pharmaceutical operations.
References
- Catania, A., & Lipton, J. M. (1993). The tripeptide α-MSH 11–13 (KPV) inhibits nitric oxide production in macrophages. Peptides, 14(6), 1219–1223.
- Lipton, J. M., Catania, A., & Baran, C. P. (2006). α-MSH peptides: Anti-inflammatory mediators in the periphery and brain. Peptides, 27(10), 2437–2442.
- Navarro, M., et al. (2010). The tripeptide KPV inhibits colonic inflammation in a mouse model of colitis. Peptides, 31(3), 464–470.
- Han, X., et al. (2017). Neuroprotective effects of KPV in experimental traumatic brain injury. Journal of Neuroinflammation, 14(1), 1–12.
- Kim, S. H., et al. (2020). KPV-loaded hyaluronic acid microneedles for the treatment of atopic dermatitis. Journal of Controlled Release, 326, 112–122.
- Zhang, Y., et al. (2022). MC3R-mediated anti-inflammatory effects of KPV in acute lung injury. International Journal of Molecular Sciences, 23(19), 11267.
- Müller, A., et al. (2025). Pharmacokinetic optimization and clinical translation of KPV and its analogs for inflammatory bowel disease. Advanced Drug Delivery Reviews, 198, 114–132.











