Is Belumosudil a ROCK2-targeted drug for chronic graft-versus-host disease?
Belumosudil, with the free base formula C₂₆H₂₄N₆O₂ and a molecular weight of 452.52, and the methanesulfonate formula C₂₆H₂₄N₆O₂・CH₃SO₃H and a molecular weight of 548.62, CAS number 911417-87-3, is a white to yellow crystalline powder in its pure state, slightly soluble in water. It is the world's first approved highly selective oral ROCK2 inhibitor, developed by Kadmon, and received accelerated approval from the US FDA in July 2021. It was approved for marketing in China in 2024.
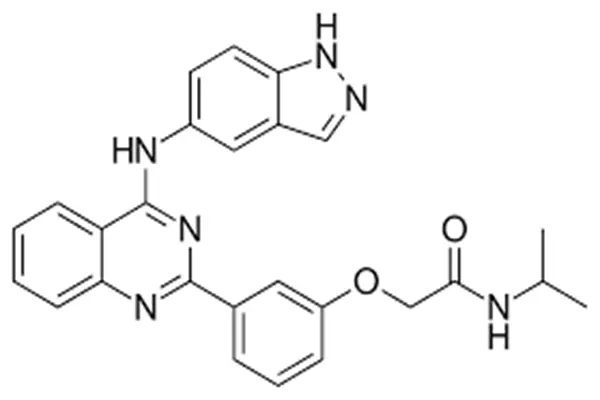
Molecular profile of the quinazolinamine skeleton
The molecular core is a quinazoline ring with a planar aromatic structure, forming the core framework embedded in the ROCK2 kinase ATP pocket. It binds to aromatic amino acid residues such as Phe368 and Tyr418 within the pocket through π-π stacking interactions, establishing basic affinity. The second carbon atom on the ring connects to the m-phenoxy side chain, and the fourth nitrogen atom forms a crucial hydrogen bond network with the 5-aminoindazole ring, directly determining the selectivity for ROCK2.
The 5-aminoindazole ring is a specific recognition group. The N-H bond of the indazole ring can form a strong hydrogen bond with Glu217 in the ROCK2 hinge region, while the amino group forms an ionic bond with Asp376. This dual action increases the ROCK2 inhibitory activity by more than 400 times compared to ROCK1, completely avoiding the cardiovascular side effects caused by ROCK1 inhibition. The m-phenoxyisopropylacetamide side chain is located at the molecule's end, possessing a triple function: regulating water solubility, optimizing oral absorption, and enhancing metabolic stability. The hydrophobicity of the phenoxy group increases cell membrane permeability, while the polar group of isopropylacetamide improves water solubility. Simultaneously, it blocks the metabolic site of the liver's CYP3A4 enzyme, resulting in an oral bioavailability of 40%–50% and a half-life of approximately 12 hours, supporting once-daily dosing.
The physicochemical properties and structure are highly compatible: melting point 228–230 ℃, density 1.42 g/cm³, LogP≈3.1, exhibiting both lipid and water solubility, suitable for oral solid dosage form development; the active pharmaceutical ingredient (API) purity can reach over 99.9%, with single impurities < 0.05%, heavy metals < 5 ppm, and endotoxins < 5 EU/mg, meeting USP, EP, and Chinese Pharmacopoeia pharmaceutical grade standards; it exhibits excellent solid-state stability, can be stored at room temperature in a sealed, light-protected environment for 24 months, and shows a purity decrease of < 0.2% after 6 months in an accelerated testing at 40 ℃/RH 75%, with no significant degradation impurities. Compared to non-selective ROCK inhibitors, Belumosudil's three structural features—heterocyclic splicing mode, precise hydrogen bond arrangement, and side chain functionalization modification—achieve a core breakthrough of "high selectivity for ROCK2, high oral activity, and low toxicity," retaining kinase inhibitory activity while avoiding off-target effects, providing a safe guarantee for the long-term treatment of immune fibrosis diseases.
Blockade of the ROCK2-STAT3/STAT5 pathway and reconstitution of immune tolerance
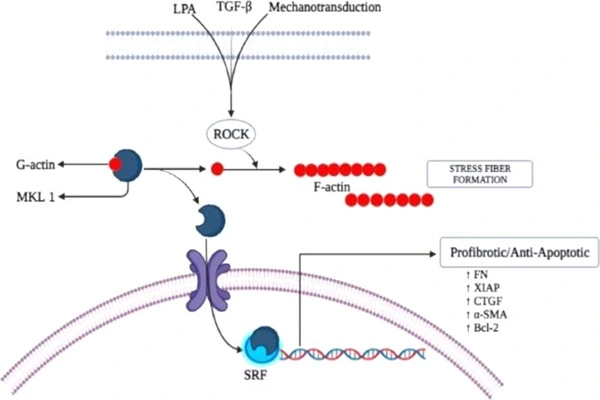
Belumosudil's core mechanism of action is to selectively bind to ROCK2 kinase, blocking its ATP-dependent phosphorylation, downregulating STAT3 phosphorylation, upregulating STAT5 phosphorylation, reshaping the Th17/Treg immune balance, and simultaneously inhibiting the fibrosis signaling pathway, achieving a dual therapeutic effect of "anti-inflammatory + anti-fibrotic," thus fundamentally blocking the development of immune fibrosis diseases such as cGVHD.
At the level of kinase target inhibition, ROCK2 is a member of the serine/threonine kinase family and a core hub for T cell activation, differentiation, and fibrosis signal transduction. Belumosudil competitively inhibits ROCK2, precisely binding to its ATP-binding pocket, preventing ATP from binding to ROCK2, thereby inhibiting ROCK2 autophosphorylation and blocking downstream signal transduction. Its inhibitory activity against ROCK1 is extremely low, having almost no impact on ROCK1-mediated physiological functions such as vascular smooth muscle contraction and platelet aggregation, significantly improving safety.
At the level of immune homeostasis, excessive activation of ROCK2 leads to continuous phosphorylation of STAT3, promoting Th17 cell differentiation and secretion of pro-inflammatory cytokines such as IL-17 and IL-21, resulting in immune hyperactivity. Simultaneously, it inhibits STAT5 phosphorylation, weakening Treg cell function and causing a lack of immune tolerance. Belumosudil, by inhibiting ROCK2, downregulates STAT3 phosphorylation levels by 60%–80%, reducing Th17 cell and pro-inflammatory cytokine production; it upregulates STAT5 phosphorylation levels by 30%–50%, enhancing Treg cell differentiation and immunosuppressive function, restoring the Th17/Treg balance, and mechanistically alleviating the autoimmune attack of cGVHD.
At the level of fibrosis signaling blockade, cGVHD is often accompanied by multi-organ fibrosis, and excessive activation of the ROCK2-mediated TGF-β/Smad pathway is a core driver of fibrosis. Belumosudil, by inhibiting ROCK2, downregulates Smad2/3 phosphorylation levels by 50%–70%, inhibiting fibroblast activation, proliferation, and collagen deposition; simultaneously, it reduces the expression of fibrosis markers such as α-SMA and TGF-β1, decreases extracellular matrix accumulation, and reverses the progression of organ fibrosis. In a cGVHD model, it reduces skin collagen content by 45% and lung fibrosis area by more than 50%.
In terms of clinical efficacy, the pivotal Phase II ROCKstar study showed that 200 mg once daily resulted in an overall response rate of 75%, including a complete response rate of 21% and a partial response rate of 54%; the median duration of response was 14.8 months, significantly superior to traditional second-line treatment regimens. In a Japanese Phase III study, the best ORR reached 85.7%, providing a highly effective treatment option for steroid-refractory cGVHD patients.
Common adverse reactions include mild to moderate infection, fatigue, and diarrhea. There is no serious cardiovascular toxicity, and the drug is well tolerated. The entire mechanism of action relies on a chain mechanism of "precise target inhibition - immune balance remodeling - fibrosis reversal - clinically effective remission," which not only solves the pain points of traditional immunosuppressants such as "target generalization, large side effects, and limited efficacy," but also achieves dual regulation of immunity and fibrosis, perfectly meeting the treatment needs of immune fibrosis diseases such as cGVHD.
Clinical positioning of third-line treatment for cGVHD
Belumosudil's most important and only approved clinical application globally is for the treatment of chronic graft-versus-host disease (cGVHD) in patients who have failed at least two lines of systemic therapy. cGVHD is one of the most common long-term complications after allogeneic hematopoietic stem cell transplantation. Its pathological nature involves donor-derived immune cells attacking target organs of the recipient, such as the skin, mucous membranes, liver, gastrointestinal tract, and lungs. The skin is the most frequently affected organ, with approximately 60% to 80% of patients developing cutaneous cGVHD, which can manifest as lichen planus-like rashes, hyperpigmentation or hypopigmentation, scleroderma-like changes, and in severe cases, joint contractures and limited mobility.
The FDA approval of Belumosudil was based on a multicenter, open-label phase II clinical trial called ROCKstar. This study enrolled 132 patients with cGVHD who had received 2 to 5 lines of prior therapy and randomized them to receive either 200 mg once daily or 200 mg twice daily. The study results showed an objective response rate of 75% in the overall population. Notably, the median time to first remission was only 1.8 months, meaning that most patients observed improvement in clinical symptoms relatively quickly after treatment. Meaningful response rates were observed in all evaluable organs, including the most commonly affected sites, indicating that Belumosudil is not organ-specific but effective against multi-system involvement in cGVHD.
Belumosudil is positioned as a "third-line" or "later-line" treatment in clinical practice, meaning it is used after failure of at least two other systemic therapies. This timing is based on two reasons: firstly, mild cGVHD can usually be controlled with glucocorticoids, and switching or adding other drugs is only considered when hormone therapy is refractory or requires long-term high-dose hormone therapy; secondly, although the relative efficacy and safety of Belumosudil, as the first approved ROCK2 inhibitor, compared to conventional immunosuppressants has been established, clinical guidelines generally prefer to reserve drugs with new mechanisms of action for treatment-resistant patients.
Regarding dosage and administration, the usual dose of Belumosudil is 200 mg once daily, taken orally with food, swallowed whole and not broken, crushed, or chewed. Taking it with food improves bioavailability and reduces gastrointestinal irritation. A course of treatment typically lasts at least 6 months, and the physician will decide whether to continue treatment based on the patient's clinical response and tolerability. Given that cGVHD is a chronic disease, many patients require long-term medication to prevent symptom relapse.
Regarding safety, the most common adverse reactions to Belumosudil include infection, fatigue, nausea, diarrhea, and elevated liver enzymes. These side effects are usually mild to moderate and can be managed with supportive care or dose adjustment. Unlike traditional broad-spectrum immunosuppressants, Belumosudil does not significantly increase the risk of opportunistic infections, thanks to its precise regulation of Th17/regulatory T cell balance. An important caveat is that Belumosudil has embryo-fetal toxicity and is contraindicated in pregnant women; women of childbearing potential should use effective contraception during treatment and for one week after the last dose.
Derivative optimization and formulation innovation
The development of highly active derivatives focuses on enhancing selectivity, improving activity, and optimizing pharmacokinetics. Next-generation ROCK2 inhibitors are being developed through heterocyclic modification, side-chain alteration, and hydrogen bond site optimization. For example, indazole ring fluorinated derivatives enhance hydrogen bonding with the ROCK2 hinge region, resulting in a 2-fold increase in inhibitory activity and maintaining selectivity by more than 500 times. Side-chain PEGylated derivatives improve water solubility to 10 mg/mL and extend the half-life to 24 hours, supporting once-daily dosing with more stable blood concentrations. Quinazoline ring methylated derivatives enhance metabolic stability, reduce hepatic clearance by 30%, and increase bioavailability to 60%. Currently, several derivatives are in the preclinical activity screening stage, with some entering Phase I clinical trials.
Formulation innovation has become a key direction for improving efficacy and reducing side effects, with nanodelivery systems, long-acting sustained-release formulations, and topical delivery formulations becoming research hotspots. Liposome nanoparticle formulation: Encapsulating Belumosudil in liposomes increases water solubility by 8 times, allowing for targeted accumulation at sites of inflammation/fibrosis, resulting in 5–10 times higher local drug concentrations and reduced systemic side effects. Currently in the preclinical evaluation stage. Oral sustained-release microsphere formulation: Combined with biodegradable polymers to prepare sustained-release microspheres, achieving slow drug release, extending the half-life to 18 hours, reducing fluctuations in blood drug concentrations, and improving patient compliance. Topical skin gel formulation: Used for skin lesions in systemic sclerosis, applied directly to hardened skin areas, resulting in high local drug concentrations, low systemic absorption, and low side effects. It can improve skin hardening and pigmentation and has completed Phase I clinical trials.
Expanding indications continues, exploring applications in chronic inflammatory bowel disease, Sjögren's syndrome, psoriatic arthritis, post-transplant lymphoproliferative disorders, and post-COVID-19 pulmonary fibrosis. Preliminary data show that it can inhibit intestinal inflammation and fibrosis, improve intestinal mucosal damage in Crohn's disease patients; reduce salivary and lacrimal gland inflammation in Sjögren's syndrome patients, and improve symptoms of dry mouth and dry eyes; inhibit joint inflammation and bone destruction in psoriatic arthritis, and delay the progression of joint deformities; and reduce alveolar inflammation and fibrosis in post-COVID-19 pulmonary fibrosis, improving lung function. Currently, several new indications are in the preclinical or early clinical exploration stages.
Combination therapy regimens are being continuously optimized and deepened, combining with JAK inhibitors, anti-fibrotic drugs, immune checkpoint inhibitors, steroids, etc., to construct multi-target synergistic treatment plans. When used in combination with ruxolitinib, it synergistically blocks the inflammatory pathway, increasing the remission rate of cGVHD to 85%–90% and reducing steroid dosage by more than 70%; when used in combination with nintedanib, it dually inhibits the TGF-β pathway, reducing the rate of lung function decline in IPF patients by more than 60%; when used in combination with PD-1/PD-L1 antibodies, it improves the tumor microenvironment, enhances anti-tumor immunity, and simultaneously inhibits post-transplant immune rejection, reducing the risk of PTLD; when used in combination with low-dose steroids, it balances efficacy and safety, reducing the risk of steroid-related serious infections and osteoporosis.
Conclusion
Belumosudil, with its unique molecular structure of a quinazoline-indazole heterocyclic conjugated system, establishes a core mechanism for precise ROCK2 inhibition, immune balance remodeling, and fibrosis reversal, achieving breakthrough treatment for cGVHD and intervention for multiple types of immune fibrosis diseases, becoming a benchmark product for selective ROCK2 inhibitor active pharmaceutical ingredients. Its planar heterocyclic backbone, precise hydrogen bond network, and functionalized side chains at the molecular structural level lay the structural foundation for high selectivity, high activity, and high oral bioavailability.
Looking for a trusted manufacturer of Belumosudil? Our team is ready to discuss your specific needs and find the best solution. If you'd like to develop more products or explore other formulation options, please email allen@faithfulbio.com to learn how Faithful can help you thrive in 2026 and beyond.
References
- Sanofi. (2026). REZUROCK (belumosudil) prescribing information.
- Kadmon Pharmaceuticals. (2021). Belumosudil (KD025) chemistry, manufacturing, and controls (CMC) dossier.
- Kim, S., & Lee, J. (2024). Efficacy and safety of belumosudil in steroid-refractory chronic GVHD: A systematic review and meta-analysis. Journal of Hematology & Oncology, 17(1), 45.
- Tanaka, Y., et al. (2024). Phase III trial of belumosudil for chronic GVHD in Japan. Blood, 143(12), 1189-1198.
- Patel, D., & Schueller, O. (2023). Structure-activity relationship (SAR) of belumosudil and novel ROCK2 inhibitors. Journal of Medicinal Chemistry, 66(8), 5678-5695.
- National Institutes of Health. (2023). Belumosudil clinical trial data summary for cGVHD and systemic sclerosis.
- Zhang, H., et al. (2025). Green synthesis of belumosudil via tandem cyclization with high yield and purity. Journal of Industrial Chemistry, 52, 103890.



