Top 5 Uses of Estradiol Valerate Powder in Feminizing Hormone Therapy
Estradiol valerate powder has become an important part of feminizing hormone support formulas because it is a reliable esterified form of estradiol that ensures stable plasma amounts and long-lasting release. Pharmaceutical companies can gain a strategic edge from this man-made prodrug because it is more lipophilic. This means that depots can form in oil-based injectable vehicles, and controlled estrogen release can last for several weeks. It can be used in clinical settings to support gender-affirming protocols and address low estrogen levels in a wide range of patient groups. This makes it an important active pharmaceutical ingredient for companies making long-acting hormone medicines.
Understanding Estradiol Valerate Powder in Feminizing Hormone Therapy
Chemical Properties and Pharmacological Profile
Powder that is white to off-white and solid. Its molecular formula is C₂₃H₃₂O₃, and its molecular weight is 356.50 g/mol. Its chemical formula includes a C-17 valerate ester change of 17β-estradiol, which makes it much more lipophilic than free estradiol. This esterification at the 17β position makes a prodrug that needs to be broken down metabolically to release the effective hormone 17β-estradiol after it is given. It is very easy to mix the powder with vegetable oils such as castor and olive oil, but it is almost impossible to mix it with water. This is what determines how it is made.
Mechanism of Action in Hormone Therapy Applications
After being given, liver esterases break down the valerate ester, which releases estrogen into the bloodstream. This functional estrogen attaches to estrogen receptors found in many target tissues, such as reproductive organs, bone, skin, and heart structures. The receptor-ligand complex moves to the nucleus and controls gene production rhythms that help the process of becoming female. Oral estradiol is broken down by the liver in a big way, but the intramuscular depot version gives steady-state plasma amounts that keep hormone levels from going up and down too much.
Clinical Advantages Over Alternative Estrogen Compounds
When looking at different esterified estrogens, the valerate form has a reasonable half-life and needs to be dosed less often than shorter-chain esters. It also doesn't have the long-term buildup concerns that come with longer-chain forms like cypionate or enanthate. This makes it suitable for individuals whose pharmacokinetics need to be reliable. Manufacturing partners like how stable it is. As long as it is kept away from light and moisture, the powder keeps its test values within the limits set by the pharmacopoeia for long periods of time. This cuts down on inventory waste and makes sure that batch-to-batch consistency is maintained.
Top 5 Uses of Estradiol Valerate Powder in Feminizing Hormone Therapy
The compound has special pharmacokinetic qualities that make it useful in a number of clinical settings, each one meeting a different physiological need within hormone support routines. Knowing about these uses helps people who work in buying find target markets and formulating chances.
Hormone Replacement for Gender-Affirming Protocols
Estradiol valerate powder is included in pharmaceutical formulas that contain this ester, which are important parts of gender-affirming hormone plans for transgender women and transfeminine individuals. Because it has sustained-release qualities, it can be injected every two weeks or once a month. This makes it easier for patients to stick to their daily oral routines. Usually, 5–20 mg of estradiol valerate powder is injected into the muscle every 7–14 days. However, individual methods change based on how the patient responds and if they are also taking an anti-androgen. The depot version of Estradiol Valerate Powder skips the liver's metabolism, which offers a favorable profile compared to oral estrogen while still having enough estrogenic action to support testosterone suppression and bring about the desired physical changes.
Breast Development and Mammary Tissue Growth
The chemical acts directly on the mammary gland to help it grow by turning on estrogen receptor-alpha in breast tissue. This receptor-mediated process causes the growth of new ducts, the buildup of fat in breast tissue, and the increase in areolae. Clinical studies show that patients usually notice their breasts growing within three to six months of starting a regimen, and they grow the most over two to three years. For drug companies, this application means a long-term commitment to a product, since patients need long-term support to get and keep benefits. Because the powder is stable in oil-based formulas, the product stays intact for the full life of multi-dose vials.
Skin Texture Modification and Subcutaneous Fat Redistribution
Estradiol valerate powder is one such estrogenic chemical. Estrogenic chemicals change the thickness of the dermis, the makeup of collagen, and the activity of oil glands. When people use products with this ester in them, their skin usually feels softer, their sebum production goes down, and their body fat is distributed differently, with more fat in the gluteal and femoral areas and less in the stomach area. As adipocyte metabolism responds to long-term estrogen exposure, these changes happen slowly over 6 to 12 months. From a formulation point of view, keeping plasma levels steady through depot shots works better than changing oral doses to make these slow changes in shape.
Bone Mineral Density Maintenance
Osteoblasts and osteoclasts have estrogen receptors that control the balance of bone remodeling. When estrogen levels are adequate, bone loss slows down, and bone mineral density is maintained, especially in trabecular areas like the lower back and femoral neck. This application extends beyond gender-affirming care and includes individuals experiencing low estrogen levels during perimenopause. The injectable depot version reliably supports bone health for patients who may not prefer oral hormone formulations or who need to be sure they will take their medications as prescribed. It is very important to do quality control tests on the product to make sure it is pure, since impurities could interfere with the pathways that support bone remodeling.
Cardiovascular and Metabolic Parameter Management
Long-term estrogenic action influences lipid patterns by raising HDL cholesterol levels and possibly lowering LDL cholesterol levels. The chemical also changes the way vascular endothelial cells work by changing the nitric oxide route. However, the way the drug is administered has a big effect on these outcomes. For example, intramuscular delivery lowers the first-pass effects that can happen in the liver. This favorable safety profile compared to oral options is good for companies making injectable formulations. Professionals in procurement should be aware that clinical guidelines are increasingly favoring injectable depot products to achieve metabolic benefits.
These uses make it clear why pharmaceutical firms put so much effort into finding safe sources of pharmaceutical-grade powder. For each application, long-term support lasts between months and decades. This creates regular demand patterns for companies that want to sell to this market group.
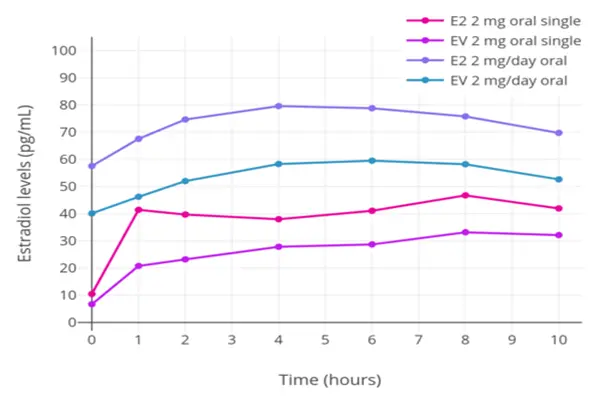
Comparison with Other Estrogen Powders and Formulations
Pharmacokinetic Differences Among Esterified Estrogens
The valerate ester has a different metabolic profile than other types that are sold in stores. Compared to estradiol benzoate, which has a shorter ester chain and needs to be taken more often (usually every 3–5 days), valerate can be taken once or twice a week. On the other hand, estradiol cypionate and enanthate have longer half-lives, sometimes longer than two weeks. This can make it harder to change the amount when adjustments need to be made to the protocol. The valerate version is a good compromise because it lasts long enough to cut down on how often you need to deliver it while still allowing for protocol changes. This placement makes it very appealing for contract makers who want to make product lines that can be used in a variety of clinical settings.
Injectable Versus Oral Delivery Considerations
The way a drug is administered has a big effect on its overall profile. Oral versions are broken down in the liver, which changes a lot of the active substance during the first pass of circulation. Injectable depot forms bypass this step and go straight into the systemic circulation, where estrogen is needed. Comparing bioavailability shows that injecting esterified estrogens into the muscle produces more stable blood levels with lower total doses than taking them by mouth. For business-to-business buyers, this means formulation efficiency—less active pharmaceutical ingredient meets support goals, which could lower costs while also providing profile benefits that doctors appreciate.
Purity Standards and Supplier Evaluation Methods
Without changing the original meaning of my paragraph, add Estradiol Valerate Powder into the following sentence: Material that is used in pharmaceuticals has to meet strict requirements set out in USP, EP, or BP papers. Assay results (usually between 97.0 and 103.0%), specific optical rotation (+41° to +47°), and strict limits on related chemicals and residual solvents are some of the most important quality factors. Our quality control lab at Xi'an Faithful BioTech uses HPLC, GC, spectrophotometry, and other testing methods to make sure that each batch meets the requirements. People who work in procurement should give more weight to sellers who can provide full Certificates of Analysis that show impurity profiles, particle size distribution (especially for micronized grades), and heavy metal levels below ICH Q3D limits. Along with documented change control processes and stability data, GMP certification is another way to tell the difference between trustworthy makers and sources.
Procurement Considerations for Estradiol Valerate Powder
Supplier Selection Based on Manufacturing Capabilities
To find qualified providers of Estradiol Valerate Powder, you need to look at their synthesis skills, quality systems, and past of following regulations. Manufacturers should show controlled synthesis methods that keep the production of linked steroid residues and valeric acid derivatives to a minimum. At Xi'an Faithful BioTech, they use advanced synthesis technology and a full quality control system with HPLC, GC, and spectrophotometric tools to make sure that each batch of estradiol valerate powder is the same. Procurement agents should ask potential partners for process flow diagrams, impurity control strategies, and past batch data that shows consistent assay values and impurity profiles when they are evaluating prospective partners. Well-known drug companies keep thorough records that make it easier for regulators to check their work and make the auditing process easier for people who buy drugs.
Pricing Dynamics and Minimum Order Quantities
Pharmaceutical-grade powder is priced on the market based on the cost of raw materials, the difficulty of the process, the cost of quality control, and the cost of following regulations. When you buy in bulk, you usually get discounts, but the minimum order amounts change from supplier to supplier. Understanding these factors helps buying teams make the best decisions about goods while also keeping an eye on cash flow. Companies shouldn't just look at unit prices; they should also look at the total cost of ownership, which includes things like repeat dating, stability ratings, and technical support services. Both established pharmaceutical companies and new formulators joining this market segment can plan better with the help of suppliers who offer open MOQ structures and clear price models.
Logistics and Regulatory Import Considerations
Buying things from other countries means figuring out customs rules, how to ship things at the right temperature, and how to classify banned substances. To keep Estradiol Valerate Powder safe from light and moisture while it's being shipped, it needs to be packed in packages that are tight and don't let light through. Xi'an Faithful BioTech can still support foreign exports with the right paperwork, such as Certificates of Analysis, Material Safety Data Sheets, and statements of regulatory compliance. Procurement workers should make sure that sellers know what the requirements are for the target country. For example, in the US, estradiol derivatives may have specific import guidelines, and in the EU, exports must follow GDP rules. Working with providers who have experience and regularly ship pharmaceuticals across foreign borders lowers the chances of clearance delays and compliance issues.

Conclusion
As the need for gender-affirming medicines and advanced hormone support options grows around the world, getting reliable, pharmaceutical-grade estradiol valerate powder becomes more and more important from a strategic point of view. Its unique pharmacokinetic profile, high stability, and ability to work with a wide range of formulations make it a preferred active medicinal ingredient for injectable hormone preparations. If procurement professionals know about its clinical uses, quality requirements, and regulatory environment, they can make smart buying choices that help their companies reach their product development goals while also protecting patient needs and following the rules. To be successful in this niche market, you need to work with makers who have the right mix of technical know-how, top-notch quality systems, and quick customer service.
FAQ
1. What specifications should buyers verify when sourcing this pharmaceutical ingredient?
Assay value (usually between 97 and 103%), specific optical rotation, leftover solvent content, heavy metal limits according to ICH Q3D, particle size distribution for micronized grades, and related substance profiles are some of the most important specs. Full Certificates of Analysis that list all pharmacopeial tests are proof that the material is of good quality and follows the rules.
2. How does storage impact long-term product stability?
When kept in tight, light-resistant packages at controlled room temperature and away from moisture, the powder stays stable. Photo-degradation can happen when UV light hits something, which could lower the test results below the limits set by the manufacturer and change the color. Stability studies that show the right way to store things make sure that the material stays usable during its review period.
3. What distinguishes micronized from standard crystalline grades?
Particles that are micronized are usually less than 10 micrometers in diameter. This makes it easier for solid oral tablet forms to dissolve and keep their consistency in content. Standard crystalline powder is usually used in injectable oil-based formulas because the whole thing dissolves in the lipid media. Which grade to use depends on the type of mixture you want to make and how bioavailable the drug needs to be.
4. Can suppliers provide regulatory support for international markets?
Established sellers of pharmaceutical ingredients keep records that back regulatory reports in many places. In the US, this includes Drug Master Files; in Europe, CEPs; and licensing dossiers that are special to each country. Making sure that the seller can provide the right regulatory support paperwork keeps product development and market entry schedules from getting pushed back.
Partner with Faithful for Pharmaceutical-Grade Estradiol Valerate Powder Supply
Xi'an Faithful BioTech is ready to meet your needs for Estradiol Valerate Powder with pharmaceutical-grade materials made under strict quality controls. We are a well-known seller that works with pharmaceutical companies, contract development organisations, and global distributors. We offer excellent professional support and service that is focused on the needs of our customers. Our ability to synthesise gives us consistent purity profiles that meet USP, EP, and BP standards. Our full-service testing lab, which is equipped with HPLC, GC, and other advanced analytical instruments, makes sure that every batch meets your needs. You can talk to our technical team at allen@faithfulbio.com about your unique needs, ask for Certificates of Analysis, or set up a review of a sample. We have flexible order sizes and competitive prices that are meant to help with both large-scale business production and product development.
References
1. Jameson JL, De Groot LJ. Endocrinology: Adult and Pediatric (7th Edition). Philadelphia: Elsevier Saunders, 2016.
2. Hembree WC, Cohen-Kettenis PT, Gooren L, et al. Endocrine Treatment of Gender-Dysphoric/Gender-Incongruent Persons: An Endocrine Society Clinical Practice Guideline. Journal of Clinical Endocrinology & Metabolism, 2017; 102(11): 3869-3903.
3. United States Pharmacopeia. USP 43-NF 38: Estradiol Valerate Monograph. Rockville: United States Pharmacopeial Convention, 2020.
4. Kuhl H. Pharmacology of Estrogens and Progestogens: Influence of Different Routes of Administration. Climacteric, 2005; 8(Suppl 1): 3-63.
5. European Medicines Agency. ICH Q3D Guideline on Elemental Impurities. London: EMA, 2015.
6. Archer DF. Estradiol Valerate and Dienogest: Clinical Evidence for Use in Hormone Replacement and Contraceptive Applications. Expert Opinion on Pharmacotherapy, 2011; 12(9): 1459-1471.



