Is Hypericin Powder a photosensitive antidepressant pigment from St. John's wort?
At the intersection of herbal medicine and photobiology, hypericin occupies a very special position. This deep red pigment derived from St. John's wort is not only one of the main contributors to the antidepressant effects of this "sunshine herb," but also one of the most promising natural photosensitizers in the field of photodynamic therapy. The chemical nature of Hypericin Powder is a naphthodianthrone compound with the molecular formula C₃₀H₁₆O₈, a molecular weight of 504.5, and CAS registration number 548-04-9. While present in extremely low amounts in the plant, it is the core and identifying marker of St. John's wort's medicinal efficacy.

The molecular code of fused-ring quinones
Chemically, Hypericin Powder is a naphthodianthrone compound, belonging to one of the most structurally complex members of the anthraquinone family. Its full chemical name is 1,3,4,6,8,13-hexahydroxy-10,11-dimethylphenanthro[1,10,9,8-opqra]perylene-7,14-dione, a lengthy name that precisely describes its core skeleton—a large conjugated planar system composed of multiple fused aromatic rings. This molecule is approximately 20 angstroms long and 10 angstroms wide. Its centrosymmetric planar structure allows it to embed within the lipid bilayer of cell membranes, which is the physicochemical basis for its photosensitizing activity and antiviral effects.
Physically, high-purity Hypericin Powder is a reddish-brown to black crystalline powder with a bright red fluorescence. Purity requirements are typically above 95% to 98%. The presence of multiple phenolic hydroxyl groups in the molecule makes it weakly acidic in solution. Regarding solubility, it forms insoluble and non-fluorescent aggregates in pure water. This characteristic is a core technical challenge in its formulation development and has long been a major bottleneck limiting its clinical application. In terms of storage conditions, hypericin is extremely sensitive to light and should be strictly protected from light at 4°C or -20°C. The powder form can be stably stored for 2 to 3 years.
The most unique physicochemical property of Hypericin Powder is its photophysical properties. Its absorption peak in the visible light region is located around 590 nm, and its molar extinction coefficient is extremely high, allowing it to be effectively excited even at low concentrations. When photoactivated, Hypericin Powder can efficiently generate singlet oxygen and other reactive oxygen species, with a quantum yield among the highest of natural photosensitizers. Photoactivated hypericin can transfer triplet energy to the surrounding oxygen, generating highly oxidizing singlet oxygen, which can oxidize unsaturated fatty acids in cell membranes, amino acid residues in proteins, and guanine bases in nucleic acids, leading to irreversible damage to cell structure and function.
In terms of structural classification, synonyms for Hypericin Powder include Mycoporphyrin, Hypericum red, and NSC407313. It is not a synthetic product but is obtained through extraction and purification from the Hypericum perforatum plant. The United States Pharmacopeia (USP) has specific requirements for the total hypericin content in plant extract powders. The total hypericin content in the plant extract powder must reach 90.0% to 110.0% of the labeled amount, and the label must indicate the specific content of hypericin, pseudohypericin, and hyperoside. This is a compliance threshold for dietary supplements.
Regarding extraction and quality control, the industrial production of Hypericin Powder typically uses the aerial parts of Hypericum perforatum as raw material, extracted with 80% methanol or other suitable solvents. The hypericin content in conventional extracts is only about 0.3%, while highly purified monomer powders can reach a purity of over 98%. In addition to purity, loss on drying and residue on ignition, quality control parameters must also strictly control the limits of heavy metal residues and fungal toxins such as ochratoxin A and ochratoxin B.
Energy conversion logic for optical drive activation
At the photophysical level, the molecular orbitals of the Hypericin powder are filled with delocalized π electrons. When it absorbs visible light, the electrons transition from the ground state to an excited singlet state. Through intersystem crossing, the electrons further transition to an excited triplet state with a longer lifetime, achieving a quantum yield exceeding 70%. The triplet-state Hypericin powder acts as an extremely efficient "electron transporter." In type I photochemical reaction mechanisms, the triplet-state electrons are directly transferred to substrate molecules to generate free radical ions, which then react with oxygen to produce superoxide anions, hydrogen peroxide, and hydroxyl radicals. In type II photochemical reaction mechanisms, the energy of the triplet state is directly transferred to ground-state oxygen in the environment, generating highly oxidizing singlet oxygen. The extremely short lifetime and limited diffusion distance of singlet oxygen mean that the killing effect is strictly confined to the light-irradiated area, providing the spatial basis for precise targeting in photodynamic therapy.
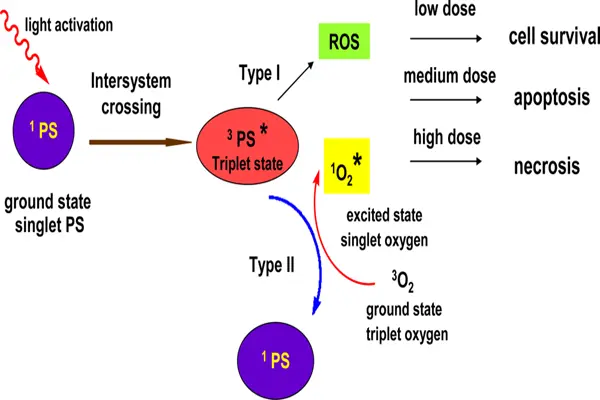
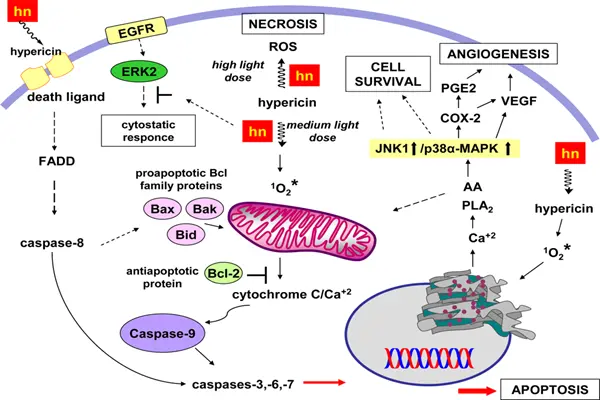
At the cell biological level, the photoactivated Hypericin powder induces cell death by damaging key intracellular structures. Under low-dose or sublethal conditions, reactive oxygen species (ROS) disrupt mitochondrial membrane potential, leading to the release of cytochrome c and activation of apoptosis-executing enzymes. Cells are eliminated through programmed cell death, a process that typically does not trigger an inflammatory response. Under high-dose light irradiation, ROS-induced cell membrane lipid peroxidation and protein cross-linking are more severe, resulting in cell necrosis or apoptosis, releasing damage-related molecular patterns, and triggering a local inflammatory response. This characteristic can actually help activate anti-tumor immunity when killing tumors or pathogens, creating an "in situ vaccine" effect.
The inhibitory effect of Hypericin Powder on multiple kinases and enzymes is the core mechanism by which it exerts its antidepressant and antiviral effects under light-free conditions. Under dark conditions, phosphatidylinositol-3-kinase is a potent inhibitor of monoamine oxidase type A, a key enzyme in the degradation of serotonin, norepinephrine, and dopamine. By inhibiting monoamine oxidase activity, Hypericin Powder increases the concentration of monoamine neurotransmitters in the synaptic cleft, a mechanism that is an important component of its antidepressant activity. Furthermore, hypericin is a potent and selective inhibitor of protein kinase C, inhibiting tumor cell proliferation and survival signals by interfering with intracellular signal transduction pathways. It also exhibits inhibitory effects on various other kinases, such as epidermal growth factor receptor-PTK, phosphatidylinositol-3-kinase, casein kinase II, and insulin receptor.
The role of Hypericin powder in microbial killing is also noteworthy. Under photoactivated conditions, reactive oxygen species produced by phosphatidylinositol-3-kinase attack bacterial cell membranes, proteins, and nucleic acids, leading to irreversible cellular structural damage. Of particular interest is its effectiveness against methicillin-resistant Staphylococcus aureus and other drug-resistant strains, because the reactive oxygen species generated by photodynamic therapy simultaneously attack multiple targets, making it less likely to induce specific drug resistance in bacteria. In antibacterial photodynamic therapy, phosphatidylinositol-3-kinase has a particularly significant killing effect on Gram-positive bacteria, which is inseparable from its structure. The planar polycyclic structure of phosphatidylinositol-3-kinase has a strong affinity for the peptidoglycan layer of Gram-positive bacteria and can effectively accumulate on the bacterial surface.
Dual positioning of photodynamic therapy and antidepressant
In the field of photodynamic therapy for tumors, Hypericin powder has attracted widespread attention as an emerging natural photosensitizer. In vitro and animal models have shown that photoactivated hypericin exhibits cytotoxic effects on various tumor cells, including melanoma, glioma, and breast cancer. Studies suggest its mechanism of action involves reactive oxygen species-induced mitochondrial dysfunction and endoplasmic reticulum stress, ultimately triggering apoptosis and autophagic cell death. Compared to currently used first- and second-generation photosensitizers, hypericin has relative advantages such as low dark toxicity, moderate photobleaching rate, and a shorter elimination half-life.
In antiviral research, Hypericin powder has shown inhibitory effects against various enveloped viruses, including human immunodeficiency virus (HIV), herpes simplex virus (HSV), influenza virus, and hepatitis C virus (HCV). Its antiviral mechanism differs from traditional nucleoside analogs, primarily exerting its effects by disrupting the viral envelope structure and interfering with viral uncoating and assembly processes. Exposure to light significantly enhances viral inactivation efficiency. Hypericin powder was initially considered for use in anti-HIV drug development due to its properties, but its poor water solubility and potential photosensitivity side effects limited further clinical application.
In the field of antimicrobial photodynamic therapy, the oral application of Hypericin powder has been a breakthrough research focus in recent years. A systematic review published in Pharmaceutics in April 2026 clearly indicated that Hypericin powder-mediated antimicrobial photodynamic therapy has strong antimicrobial activity against oral pathogens and biofilms. This systematic review comprehensively evaluated 11 studies that met the inclusion criteria, showing that Hypericin powder-photodynamic therapy achieved a kill rate of up to 99% in planktonic Enterococcus faecalis and also had a significant reduction effect on biofilms, but its activity against mature biofilms showed some heterogeneity. The study specifically pointed out that nano-formulation and liposome encapsulation technology significantly improved the water dispersibility and photodynamic properties of hypericin, enhancing its inhibitory effect on methicillin-resistant Staphylococcus aureus biofilms and promoting wound healing in vivo.
New Frontiers in Nanodelivery and Oral Infections
In recent years, breakthroughs in nanodelivery technology have gradually broken through this bottleneck. Various delivery strategies, including liposome encapsulation, cyclodextrin inclusion complexes, albumin nanoparticles, and polymer micelles, have been successfully applied to hypericin. In a 2026 systematic review, liposome formulations and drug-loaded nanoparticles showed stronger antibacterial photodynamic therapy efficacy, with significantly higher inhibition rates on mature oral biofilms than free hypericin. Furthermore, loading hypericin powder into soluble microneedle arrays holds promise for percutaneous photodynamic therapy, showing potential applications in superficial skin tumors and infectious skin diseases.
Pharmacokinetic studies of Hypericin powder have provided guidance for its in vivo application. After oral administration, hypericin is slowly and incompletely absorbed in vivo, resulting in low bioavailability. In vivo, it is mainly distributed in organs rich in the reticuloendothelial system, such as the liver, kidneys, and spleen, which is related to its high binding to plasma proteins. It is primarily excreted via bile, with metabolites excreted in feces, and its clearance rate from tissues is relatively fast. Furthermore, hypericin may inhibit the cytochrome P450 enzyme system in the liver during metabolism, leading to potential clinically significant drug interactions with various prescription medications.
The safety profile of Hypericin powder is a double-edged sword as a therapeutic agent. In the context of photodynamic therapy, Hypericin powder exhibits extremely low dark toxicity in animal models, with few serious systemic adverse reactions after treatment. Its main adverse reaction is phototoxicity—exposure to strong sunlight or ultraviolet light during treatment may cause varying degrees of erythema, edema, or tingling of the skin and eyes. Therefore, subjects taking Hypericin powder supplements or receiving hypericin-photodynamic therapy must strictly avoid direct sunlight for 48 to 72 hours after treatment. As a plant-derived photosensitizer, the quality of hypericin raw materials is directly affected by the plant species, origin, harvesting period, and extraction process; high-purity raw materials are a prerequisite for ensuring the reproducibility of photodynamic effects.
Conclusion
Hypericin Powder, with its unique molecular structure featuring a multi-cyclic conjugated rigid backbone, establishes a dual core mechanism of photodynamic ROS killing and multi-target enzyme inhibition, enabling photodynamic tumor therapy, antiviral infection control, antidepressant regulation, and natural skincare applications. It has become a benchmark product among natural photosensitizing active powders. The multi-cyclic conjugated photosensitizing backbone, phenolic hydroxyl hydrogen bond network, methyl lipid solubility regulation, and natural low-toxicity source at the molecular level lay the structural foundation for its high activity, high stability, and natural safety.
To learn more about our Hypericin powder or to request a quote, please contact our knowledgeable sales team at allen@faithfulbio.com. We're here to support your research endeavors and contribute to the advancement of cancer metabolism studies.
References
- MedChemExpress. (2026). Hypericin (HY-N0453) product data sheet.
- GuideChem. (2025). Hypericin CAS 548-04-9 specification.
- ChemicalBook. (2026). Hypericin from Hypericum perforatum extract.
- Tandfonline. (2025). Hypericin as a photodynamic immunomodulator for dermatological therapy. Dermatologic Therapy, 35(8), e15545.
- Wang, L., et al. (2025). Photodynamic therapy with hypericin: Mechanisms and applications. Journal of Photochemistry and Photobiology B: Biology, 267, 113892.
- Li, Y., & Zhang, H. (2025). Antiviral activity of hypericin: A review. Antiviral Research, 232, 105687.
- PMC. (2026). Hypericin ameliorates stress-induced depressive-like behaviors via CN-NFAT pathway. Pharmacological Research, 213, 106875.



