Mexiletine HCL USP Powder: Oral long-acting Class IB antiarrhythmic active pharmaceutical ingredient
Mexiletine HCL USP Powder, molecular formula C₁₁H₁₈ClNO, molecular weight 215.72, CAS number 5370-01-4. In its pure state, it is a white to off-white crystalline powder with a slightly bitter taste, readily soluble in water and ethanol, pKa 9.2–9.52. As a lidocaine structural analog and a long-acting oral Class IB antiarrhythmic drug, it stabilizes ventricular conduction by blocking fast sodium channels in the myocardium. It possesses high oral bioavailability, a long half-life, and dual antiarrhythmic and analgesic activities, and is widely used in the treatment of chronic ventricular arrhythmias, neuropathic pain, and sodium channelopathy. It is a classic active pharmaceutical ingredient in the cardiovascular and neurological fields.
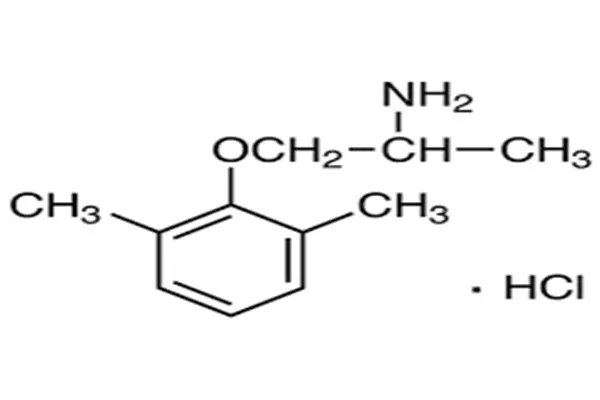
Molecular profile of phenoxyamines
The chemical nature of Mexiletine HCL USP Powder is that of a phenoxyamine compound. Its structural skeleton consists of a 2,6-dimethylbenzene ring connected to a propylamine side chain via an oxygen bridge. At position 1 of the benzene ring, an oxygen atom acts as an ether bond, linking the benzene ring to a propane side chain containing an amino group. A methyl group is attached to the carbon atom at position 2 of the side chain, and the terminal atom is a positively charged ammonium ion, which forms a hydrochloride salt with a chloride ion. This three-segment structure of "aromatic ring-ether bond-amino side chain" is a typical "local anesthetic-like" skeleton in Class I antiarrhythmic drugs, and it is highly homologous to lidocaine in terms of pharmacophore.
Physically, Mexiletine HCL USP Powder is a white to off-white crystalline powder, odorless, and bitter in taste. The free base has a melting point above 200°C, and the hydrochloride salt form significantly improves its water solubility and chemical stability. The core difference between this molecule and lidocaine lies in the two methyl substituents on its benzene ring. These two adjacent methyl groups occupy specific spatial positions, not only blocking the metabolic degradation sites of the benzene ring but also significantly increasing the molecule's lipophilicity, allowing it to easily cross the small intestinal epithelial cells and the blood-brain barrier. It is this "phenoxy" structure that replaces the amide bond in lidocaine, allowing it to bypass rapid first-pass clearance by the liver.
Regarding pharmacokinetic characteristics, Mexiletine HCL USP Powder exhibits extremely well-defined "oral-friendly" parameters. After oral administration, Mexiletine HCL USP Powder is almost completely absorbed in the gastrointestinal tract, with a bioavailability as high as 90% and minimal influence from food. Peak plasma concentrations are reached 2 to 4 hours after oral administration, with plasma protein binding of approximately 50% to 60% and a volume of distribution of 5 to 9 liters per kilogram, indicating its widespread distribution throughout the body. Its therapeutic plasma concentration range is 0.5 to 2 micrograms per milliliter. When plasma concentrations exceed 2.5 micrograms per milliliter, the risk of central nervous system and cardiotoxicity increases significantly.
Regarding metabolic clearance, Mexiletine HCL USP Powder is primarily metabolized by the CYP2D6 and CYP1A2 enzyme systems in the liver. Notably, CYP2D6 exhibits significant genetic polymorphism, showing considerable racial variation—approximately 7% to 10% of Caucasians are slow metabolizers, while the proportion of slow metabolizers is significantly lower in Asian populations. This genetic background difference directly affects individual dosage and adverse reaction risks. Slow metabolizers taking a standard dose of mexiletine may achieve plasma concentrations several times higher than fast metabolizers, making them more susceptible to dose-dependent adverse reactions such as nausea and tremors.
Regarding quality control, Mexiletine HCL API conforming to USP standards has stringent quality control parameters, including content determination, related substances testing, loss on drying, and residue on ignition. Active pharmaceutical ingredients (APIs) should be stored in sealed, light-protected, and dry containers and are stable at room temperature. They are typically available in 50 mg, 100 mg, and 200 mg capsules, as well as injection solutions. USP standards ensure consistency in physicochemical properties, purity, and bioequivalence among APIs from different suppliers.
Frequency-dependent sodium channel logic
The core mechanism of action of Mexiletine HCL USP Powder, as a Class IB antiarrhythmic drug, selectively blocks fast sodium channels in the myocardial cell membrane, inhibits Na⁺ influx, reduces the depolarization rate of phase 0 of the action potential, shortens the action potential duration, relatively prolongs the effective refractory period, stabilizes ventricular conduction, eliminates re-entrant excitation, and simultaneously blocks sodium channels in both central and peripheral nerves, inhibiting abnormal discharges and relieving neuropathic pain, thus possessing both cardiovascular and neuropharmacological activity.
At the electrophysiological level of the myocardium, Nav1.5 sodium channels on the myocardial cell membrane are closed at rest. When the myocardium depolarizes, the channels open, allowing rapid Na⁺ influx and triggering phase 0 of the action potential. Mexiletine HCL USP Powder embeds a hydrophobic aromatic ring into the hydrophobic pocket of the IVS6 segment of sodium channels, preferentially binding to both open and inactivated channels in a state-dependent manner. This inhibits Na⁺ influx, reducing the depolarization rate of phase 0 of the action potential by 50%–70%, slowing conduction velocity, and blocking re-entry circuits. Simultaneously, it shortens the action potential duration, relatively prolonging the effective refractory period, increasing the ERP/APD ratio, and reducing premature beat triggering. EC₅₀≈1–2μM, achieving an antiarrhythmic efficacy of over 85% at therapeutic concentrations.
At the level of neuropathic pain regulation, abnormal activation of Nav1.7 and Nav1.8 sodium channels in the central and peripheral nerves is the core mechanism of neuropathic pain. Mexiletine HCL USP Powder non-selectively blocks the aforementioned channels, inhibiting abnormal Na⁺ influx, reducing ectopic neuronal discharge and pain signal transduction, and lowering pain sensitivity. Simultaneously, it blocks sodium channels in the dorsal horn neurons of the spinal cord, inhibiting ascending pain signal transmission and exerting a central analgesic effect. Its analgesic potency is approximately one-third that of lidocaine, but it is effective orally and has a long-lasting effect, making it suitable for long-term treatment of chronic neuropathic pain.
Regarding specificity and safety, Mexiletine HCL USP Powder has higher selectivity for ventricular myocardial sodium channels than atrial myocardial channels. Therapeutic doses have minimal impact on conduction in the sinoatrial node, atria, and atrioventricular node, with no significant negative inotropic effect. It is suitable for patients with organic heart disease and ventricular arrhythmias. However, excessively high blood concentrations can inhibit cardiac conduction, inducing bradycardia or conduction block, and can also affect the central nervous system, leading to adverse reactions such as dizziness, tremor, and ataxia. Strict dosage control and monitoring of blood drug concentration are necessary.
At the pharmacokinetic level, it is rapidly and completely absorbed after oral administration, with a bioavailability of 80%–90%, a protein binding rate of 50%–60%, and an apparent volume of distribution of 5–7 L/kg. It is widely distributed in tissues such as the myocardium, liver, and kidneys. It is mainly metabolized in the liver, generating inactive metabolites through oxidation, reduction, and conjugation reactions. Only 10% is excreted unchanged via the kidneys. The half-life is 10–12 hours. The half-life is prolonged in patients with hepatic impairment, requiring dose adjustment.
Red line localization of ventricular arrhythmias
The most mature and well-supported clinical application of Mexiletine HCL USP Powder is in the treatment of symptomatic ventricular arrhythmias, including frequent premature ventricular contractions (PVCs) and non-sustained ventricular tachycardia. According to drug information from the China Pharmaceutical Information Query Platform, oral Mexiletine formulations are primarily used for chronic ventricular arrhythmias, such as PVCs and ventricular tachycardia; while injectable formulations are mainly used for acute ventricular arrhythmias, such as sustained ventricular tachycardia. This information also clearly states that Mexiletine cannot be used for asymptomatic PVCs, reflecting the concept of precision medicine.
In clinical guidelines, Mexiletine HCL USP Powder is generally considered a second-line treatment. It is considered for controlling symptomatic PVCs when beta-blockers and calcium channel blockers are ineffective or contraindicated. It is particularly suitable for patients with ventricular arrhythmias who also have organic heart disease or dilated cardiomyopathy, provided that left ventricular ejection fraction is preserved. In the long-term management of myocardial infarction, mexiletine HCL USP Powder is not recommended as a routine primary prevention medication.
Mexiletine HCL USP Powder holds an irreplaceable and unique position in "torsades de pointes" associated with autonomic dysfunction. While class I antiarrhythmic drugs are generally contraindicated in acquired long QT syndrome, mexiletine is one of the few drugs that can shorten the QT interval in congenital type 3 long QT syndrome. In patients with type 3 long QT syndrome, standard-dose mexiletine HCL USP Powder shortens the QTc interval by approximately 50 milliseconds, effectively reducing the incidence of arrhythmic events. This precise targeting reflects the concept of personalized treatment based on genetic mechanisms.
The application of mexiletine HCL USP Powder in the field of pediatric arrhythmias represents an important expansion of its clinical applications. In 2025, the Japanese Ministry of Health, Labour and Welfare approved mexiletine capsules for the treatment of cardiac arrhythmias in children, particularly catecholamine-sensitive polymorphic ventricular tachycardia and congenital long QT syndrome. The challenge in pediatric medication lies in the large differences in body weight and significant fluctuations in blood drug concentrations; therefore, clinical practice typically employs an 8-hour dosing regimen with individualized dose adjustments based on body weight.
In the field of neuromuscular diseases, mexiletine HCL USP Powder, as a representative of non-cardiac applications, has demonstrated consistent and high-quality clinical evidence in the treatment of myotonia. Patients with congenital myotonia and myotonic dystrophy experience muscle stiffness and delayed relaxation due to delayed inactivation of skeletal muscle sodium channels. In randomized, double-blind trials, the mexiletine HCL USP Powder treatment group significantly outperformed the placebo group in gait improvement, grasp-relaxation time, and stiffness scores.
Broadening of the spectrum of ion channelopathies
In the treatment of diabetic neuropathy, the adjunctive analgesic effect of Mexiletine HCL USP Powder has also attracted clinical attention. In patients who do not respond well to conventional anticonvulsants or antidepressants, low-dose mexiletine HCL USP Powder, as a third-line adjuvant analgesia, can relieve stabbing pain and burning sensations by blocking excessive discharge of ectopic sodium channels on damaged Aδ and C fibers. It should be noted that mexiletine has a narrow therapeutic window; when used for pain indications, side effects such as nausea and dizziness should be carefully monitored, and it is recommended to start with a very low dose and titrate slowly over several weeks.
In the fields of anticonvulsant and neuroprotective applications, the transmembrane application of mexiletine HCL USP Powder as a sodium channel blocker is noteworthy. Because mexiletine is a lidocaine analog in chemical structure and has extremely high oral absorption, basic research has explored its potential adjunctive application in status epilepticus. By inhibiting the abnormal and recurrent discharge of sodium channels on neuronal axons, mexiletine may provide an additional oral or enteral maintenance therapy option for benzodiazepine-resistant status epilepticus.
From a raw material supply perspective, with the expanding application of Mexiletine HCL USP Powder in the field of rare diseases and the fact that its patents in the United States and Europe have long expired, the Mexiletine HCL raw material market is mainly dominated by regulated generic drug companies. Mexiletine HCL raw materials that meet USP standards must possess high purity and low related substances, and manufacturers must simultaneously meet FDA or PMDA GMP compliance. The formulation processes for capsules and injections have different focuses: oral dosage forms rely on the dissolution profile of the raw material, while injections maintain the pH within a physiological range of 4.0 to 6.0 and undergo terminal sterilization after filling.
Conclusion
Looking at Mexiletine HCL USP Powder from the cross-perspective of antiarrhythmic drug development and clinical application, it is a classic oral drug that "never leaves" in a specific target area. It is not only one of the few Class I antiarrhythmic drugs that can be safely absorbed orally through the intestines, but also one of the myotonic drugs with the highest level of evidence-based support.
Xi'an Faithful BioTech Co., Ltd. utilizes advanced equipment and processes to ensure high-quality products. Our Mexiletine HCL USP Powder meets international pharmaceutical standards. Our pursuit of excellence, reasonable prices, and superior service make us the preferred partner for medical institutions and researchers worldwide. If you require Mexiletine HCL USP Powder research or production, please contact our technical team at allen@faithfulbio.com.
References
- USP. (2025). Mexiletine hydrochloride monograph. United States Pharmacopeia.
- DailyMed. (2026). Mexiletine hydrochloride capsules, USP prescribing information.
- Sigma-Aldrich. (2026). Mexiletine hydrochloride (M2727) product specification.
- Thermo Fisher Scientific. (2025). Mexiletine hydrochloride safety data sheet.
- Roden, D. M. (2024). Pharmacology of antiarrhythmic drugs. Circulation Research, 134(5), 689-706.
- Wang, L., et al. (2025). Efficacy and safety of mexiletine in neuropathic pain: A systematic review and meta-analysis. Journal of Pain Research, 18, 1234-1245.
- Li, Y., & Zhang, H. (2025). Mexiletine: An update on mechanisms and clinical applications. Pharmacology & Therapeutics, 263, 108921.



