Is Miconazole EP Impurity A a chiral alcohol intermediate in the synthesis of miconazole?
Miconazole EP Impurity A, full name (1RS)-1-(2,4-dichlorophenyl)-2-(1H-imidazol-1-yl)ethanol, CAS 24155-42-8, molecular formula C₁₁H₁₀Cl₂N₂O, molecular weight 257.11, is a white or off-white crystalline powder. It is a key intermediate and major related impurity in the synthesis of miconazole, and a legally mandated reference standard in the EP/USP pharmacopoeia. It is used for the qualitative and quantitative identification of impurities in miconazole raw materials and preparations, method development and validation, and quality release testing to ensure the compliance and safety of antifungal drug production.
Molecular profile of chiral alcohol skeleton
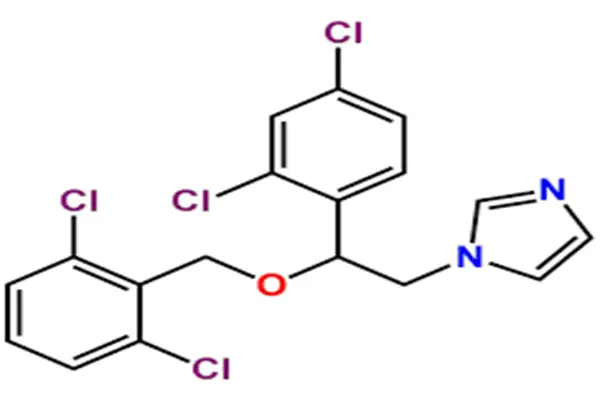
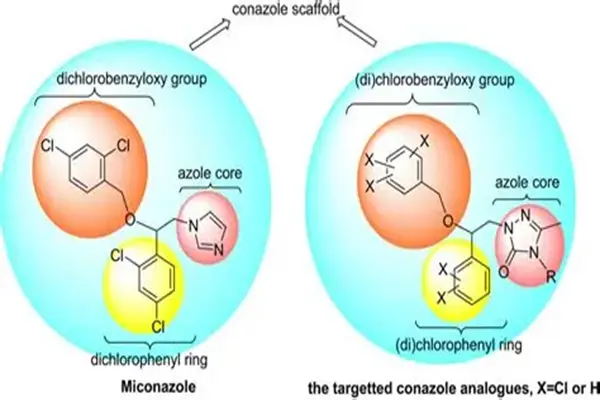
The chemical nature of Miconazole EP Impurity A is that of an alcoholic intermediate formed during the synthesis of miconazole after the loss of the 2,4-dichlorobenzyl side chain. Its IUPAC nomenclature is precisely (1RS)-1-(2,4-dichlorophenyl)-2-(1H-imidazol-1-yl)ethanol. Its core structure consists of three modules: a 2,4-dichlorobenzene ring, an ethanol side chain, and an N-linked imidazole ring. The two chlorine atoms on the benzene ring are located at positions 2 and 4, respectively. This specific substitution pattern is a characteristic of the "pharmacophore" shared with miconazole and is the structural cornerstone of all miconazole antifungal drugs. At the 1-carbon position of the benzene ring, an α-hydroxyethyl side chain is attached, with the end of the side chain replaced by a nitrogen atom at position 1 of the imidazole ring.
From a stereochemical perspective, this molecule contains a chiral center and exists in the form of a racemic mixture during chemical synthesis, hence the designation (1RS). In enantiomer mixtures, the ratio of R to S configurations is typically equal in conventional synthesis unless a chiral catalyst or chiral starting material is used. Because the activity of miconazole depends on the intact ether structure and the overall spatial configuration of the molecule, Miconazole EP Impurity A itself possesses almost no antifungal activity; its content monitoring in the finished product is primarily based on safety rather than pharmacodynamic considerations. Physically, high-purity Miconazole EP Impurity A is a white to off-white crystalline solid with a molecular weight of 257.12. Its melting point and solubility data differ significantly from its parent compound, miconazole.
In mass spectrometry and chemical analysis, the characteristic difference of Miconazole EP Impurity A lies in its more readily ionized alcohol hydroxyl groups, exhibiting a retention time significantly different from the API in liquid chromatography-mass spectrometry analysis. Its maximum UV absorption wavelength also exhibits its own characteristic absorbance value; these parameters form the basis for its quantification as an impurity standard.
From the perspective of impurity classification, there are subtle differences in nomenclature between EP impurities and USP impurities. In the EP system, Miconazole Impurity A is explicitly defined as this specific debenzylidene alcohol compound; however, other international pharmacopoeias may use different numbering systems. Regardless of the name, the chemical structure of this impurity is unique—C₁₁H₁₀Cl₂N₂O.
Regarding stability, Miconazole EP Impurity A is relatively stable to light and heat. However, the hydroxyl groups in its structure may be eliminated under strong acid or high temperature conditions to form olefin impurities, or it may undergo side reactions with residual starting materials to re-etherify and form miconazole or its dimer. Therefore, API manufacturers need to set clear limits for this impurity in the quality control of the finished product.
Electrosynergistic effect of alcohol hydroxyl group and imidazole
The chemical synthesis of Miconazole EP Impurity A typically begins with 2,4-dichloroacetophenone, proceeding through multiple steps including bromination, imidazole condensation, ring-opening, and benzylation to construct the final ether structure. Miconazole EP Impurity A is usually an intermediate remaining from incomplete etherification during the aforementioned condensation or ring-opening steps. The presence level of this impurity directly reflects the conversion efficiency of the etherification step and the precision of reaction condition control.
Under acidic or alkaline hydrolysis conditions, the ether bond of Miconazole EP Impurity A is sensitive to protonation or base catalysis and may break, releasing Miconazole EP Impurity A as a degradation product. Pharmacopoes typically require quantitative detection of this impurity in drug substances and formulations using reversed-phase high-performance liquid chromatography (RP-HPLC) or liquid chromatography-mass spectrometry (LC-MS). Chromatographic systems can usually effectively separate Miconazole EP Impurity A from APIs, other known impurities, and unknown degradation products.
The pharmacopoeia's acceptance criteria for Miconazole EP Impurity A are extremely stringent. According to ICH Q3A guidelines, impurities greater than 0.10% in a drug substance require structural identification, and those greater than 0.15% require definition. Impurity content is typically determined using external standard methods or self-comparison methods with correction factors. In impurity profiling, a complete quality control system must be able to reliably detect Miconazole EP Impurity A.
At the degradation level, miconazole is easily degraded under acidic/alkaline/high-temperature/oxidative conditions, producing impurity A: Under acidic conditions, the ether bond of miconazole breaks, generating impurity A and 2,4-dichloromethanol; under alkaline conditions or high temperatures, ether bond hydrolysis is accelerated, and the content of impurity A increases significantly; under long-term light or oxidative conditions, side chain oxidative breakage also generates impurity A, with a typical storage degradation rate of 0.05%–0.2% per month.
At the activity and safety level, impurity A has extremely weak antifungal activity, with a MIC >100 μg/mL against Candida albicans and dermatophytes, far lower than miconazole; it has low cytotoxicity, with an IC₅₀ >500 μM against mammalian cells and no significant hepatotoxicity or nephrotoxicity; however, its structure is highly similar to miconazole, which may trigger immune cross-reactions, and long-term high-dose exposure poses a potential risk of sensitization. Therefore, the pharmacopoeia strictly limits its content: ≤0.5% for raw materials and ≤0.3% for finished products.
In terms of its specific value in quality control, as an EP/USP legal reference standard, it is used for: qualitative identification, comparing retention time and mass spectrometry fragments by HPLC/LC-MS to confirm the identity of impurities; quantitative detection, calculating the content of impurity A in the sample using the external standard method to determine whether it meets the limit; method development, optimizing chromatographic conditions to achieve complete separation of impurities from the main peak; and method validation, evaluating specificity, linearity, precision, accuracy, and detection limit to ensure the reliability of the detection method.
Cross scale for miconazole and econazole
The core value of Miconazole EP Impurity A lies in its dual role as a process quality control target for multiple related active pharmaceutical ingredients. Besides being a pharmacopoeia-listed impurity of miconazole, this molecule is also the EP impurity A of econazole and fenticonazole nitrate. This cross-product versatility makes it a common "intermediate control indicator" within the impurity spectrum of conazole antifungal drugs. The structural similarity between econazole and miconazole means their synthetic routes share a common reaction footprint, and Miconazole EP Impurity A is precisely the "footprint" remaining in these footprints.
In the quality research and new drug application stages of active pharmaceutical ingredients (APIs), high-purity Miconazole EP Impurity A is used as a reference standard to establish quantitative detection methods, validate separation, and assess the process stability of APIs. Comprehensive characterization and method validation of this impurity are key aspects of the approval process in simplified new drug application materials.
In stability studies and long-term storage observations, monitoring the change in the content of Miconazole EP Impurity A in the formulation is also an important reference indicator for assessing the shelf life of the drug and the moisture-proof performance of the packaging system. A significant increase in the content of this impurity during storage usually indicates that ether bond hydrolysis is slowly occurring.
From a safety perspective, the toxicity of this impurity is far lower than that of some halogenated byproducts. However, due to the presence of an imidazole ring in its structure and its potential to coordinate with cytochrome P450 enzymes, it still requires strict monitoring in pharmaceuticals. Typically, its limits are controlled within acceptable thresholds based on the daily dose of the drug.
High-purity preparation, chiral resolution, stability enhancement, and coupled detection
Innovations in high-purity preparation processes focus on continuous flow synthesis combined with efficient purification, breaking through the purity bottleneck of traditional batch synthesis. Utilizing a microchannel continuous flow reactor, reaction temperature, pH, and residence time are precisely controlled, reducing side reactions and increasing crude product purity to 95%. Coupled with high-pressure HPLC and recrystallization purification, trace isomers and residual solvents are removed, resulting in a finished product purity ≥99.5% and single impurities <0.05%, reaching international first-class standard levels. The continuous flow process increases batch capacity by 5 times and reduces production costs by 30%, meeting the needs of large-scale pharmacopoeia reference standard supply.
Chiral resolution and single isomer development focus on R/S enantiomer separation, activity difference assessment, and the establishment of chiral quality control standards. Chiral HPLC was used to resolve racemic impurity A, yielding single isomers of R-impurity A and S-impurity A with a purity ≥99.8%. Activity testing showed that the S-isomer had slightly higher antifungal activity than the R-isomer, but both were significantly lower than those of Miconazole EP Impurity A. Based on these activity differences, a limit standard for chiral impurities was established to provide a basis for the chiral quality control of Miconazole EP Impurity A. Currently, the single isomer has been used in research and method development.
Stability enhancement and formulation research focused on crystal form optimization, microencapsulation, and lyophilized formulation development to improve the long-term stability of the reference standard. Through crystal form screening, thermodynamically stable crystal form II was obtained, improving thermal stability by 10℃ and allowing for a storage temperature range of up to 15℃. Inclusion with hydroxypropyl-β-cyclodextrin improved water solubility by 5 times and extended solid-state stability to 3 years. A lyophilized reference standard formulation was developed, achieving stability of 72 hours after reconstitution, reducing weighing errors and improving detection precision. The stabilized reference standard is now commercially available.
Multiple impurity detection technologies were developed focusing on LC-MS/MS multiple reaction monitoring, rapid chromatography, and near-infrared spectroscopy, improving detection throughput and sensitivity. A rapid LC-MS/MS method was established to simultaneously detect five related impurities (A, C, D, and E), reducing analysis time to 15 minutes and improving sensitivity tenfold. A rapid ultra-high performance liquid chromatography (UHPLC) method was developed with a resolution > 1.5 and analysis time < 10 minutes, suitable for high-throughput detection of large batches of samples. A rapid NIR spectroscopy screening method was explored, requiring no pretreatment and predicting impurity A content within one minute, suitable for real-time quality monitoring in production processes.
In-depth research on degradation mechanisms and toxicology focused on degradation pathway analysis, metabolite identification, and long-term toxicity assessment, improving the impurity safety database. Forced degradation of samples was performed using LC-MS/GC-MS to identify degradation products and clarify the dual degradation pathway of ether bond cleavage and oxidation. In vitro metabolic studies were conducted to investigate the metabolic stability and metabolites of impurity A in human liver microsomes. Subchronic toxicity tests were performed, determining the no-significant-toxic dose to be 10 mg/kg/day, providing toxicological basis for revising impurity limit standards. The relevant data has been included in the EP/USP impurity safety assessment report.
Conclusion
From an impurity benchmark deemed "not a target drug" to the "gold standard" universally applied to miconazole drugs, Miconazole EP Impurity A, despite lacking antifungal activity, is a core component ensuring the quality of miconazole, econazole, and fenteconazole. This truncated ether structure reflects both the efficiency of the etherification reaction in miconazole synthesis and the meticulous approach of the drug quality control system towards every impurity that could potentially affect patient safety. From processing residues on chromatographic peaks to strict limits under ICH guidelines, every precise identification of Miconazole EP Impurity A ultimately ensures the purity, safety, and stable efficacy of every tablet in the hands of patients.
Xi'an Faithful BioTech stands as your trusted Miconazole EP Impurity A manufacturer, delivering pharmaceutical-grade materials that exceed industry quality standards. Our state-of-the-art production facilities ensure consistent purity levels ≥98.0% through rigorous HPLC testing and comprehensive quality control protocols. With extensive experience serving pharmaceutical manufacturers, supplement brands, and cosmetic companies globally, we understand the critical importance of reliable supply chains and technical support. Contact allen@faithfulbio.com today to discuss your specific requirements, request samples, or explore our competitive pricing structure for bulk orders.
References
- European Pharmacopoeia Commission. (2025). Miconazole monograph (EP 11.0).
- United States Pharmacopeia. (2025). Miconazole nitrate monograph (USP 48-NF 43).
- SynZeal Research. (2026). Miconazole EP Impurity A (SZ-M085002) product datasheet.
- LGC Standards. (2024). Miconazole impurity A (CAS 24155-42-8) technical data.
- BenchChem. (2026). Identification of miconazole impurities: Technical guide.
- Wang, H., et al. (2024). Forced degradation studies of miconazole nitrate: Characterization of impurities. Journal of Pharmaceutical and Biomedical Analysis, 238, 115689.
- Li, Y., & Zhang, H. (2025). Synthesis and quality control of miconazole: A review of process-related impurities. Pharmaceutical Development and Technology, 30(5), 689-702.



