Spermidine trihydrochloride powder: Can the conversion of "salt form" to enhance bioavailability unlock the full potential of polyamine therapy?
Spermidine trihydrochloride powder is the pharmaceutically stable salt form of the endogenous aliphatic polyamine spermidine, widely present in human cells, animal and plant tissues, and microorganisms. It participates in regulating a series of fundamental life activities, including DNA stability, chromatin structure, cell proliferation and differentiation, mitochondrial function, and immune inflammation balance. With aging, spermidine levels in the human body show a significant progressive decline. This phenomenon has been confirmed by multiple large-scale population cohort studies and is closely related to accelerated aging, cognitive decline, increased risk of cardiovascular events, enhanced chronic low-grade inflammation, and metabolic disorders. Free spermidine is a highly viscous alkaline liquid with drawbacks such as strong hygroscopicity, easy oxidation, easy degradation, and difficulty in formulation, making it unsuitable for direct use as a raw material. However, the trihydrochloride structure, through protonation, transforms the molecule into a stable white crystalline powder, significantly improving water solubility, storage stability, and formulation compatibility, while retaining all biological activity. This makes it the most industrially valuable polyamine raw material in the fields of anti-aging, neuroprotection, cardiovascular health, adjuvant cancer treatment, and anti-inflammatory repair.
A "morphological revolution" from viscous liquids to crystalline powders
The chemical nature of spermidine trihydrochloride can be summarized in one sentence: each of the three amino groups in the spermidine molecule combines with one hydrogen chloride molecule to form an ionic compound of C₇H₂₂Cl₃N₃. Its CAS registry number is 334-50-9, and its molecular weight is 254.63 g/mol, which is three times the molecular weight of hydrogen chloride compared to the 145.25 g/mol of free spermidine. Structurally, spermidine trihydrochloride retains all the characteristics of free spermidine. It is a linear polyamine composed of seven carbon atoms and three nitrogen atoms, with the specific sequence H₂N-(CH₂)₄-NH-(CH₂)₃-NH₂. The three nitrogen atoms are located at the two ends and the center of the chain, respectively, and are all positively charged under physiological pH conditions. When free spermidine is converted to trihydrochloride, each of these three nitrogen atoms forms an ionic bond with a chloride ion, resulting in a "trication-trichloride" ion pair structure for the entire molecule. This structural feature determines the behavior of spermidine trihydrochloride in water: upon dissolution, the ion pairs rapidly dissociate, releasing a positively charged spermidine cation and a negatively charged chloride ion. The spermidine cation is the active form that actually enters the cell and exerts its biological function.
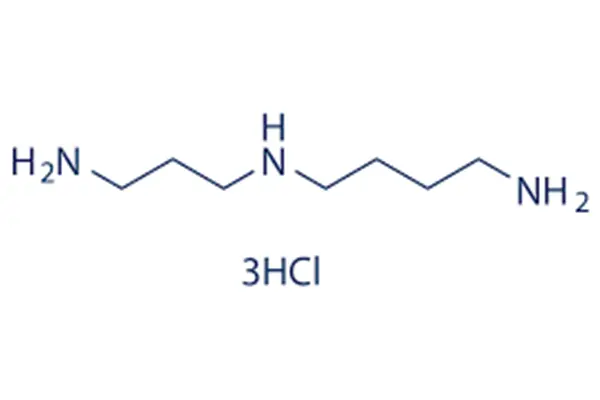
This "salt form" transformation brings about revolutionary changes in physicochemical properties. Free spermidine is a colorless to pale yellow viscous liquid at room temperature, with a melting point of only 22 to 25°C, meaning it is liquid even at room temperature in summer. More problematic is its extremely strong hygroscopicity—it absorbs moisture and becomes viscous within minutes of exposure to air, posing significant difficulties for accurate weighing and long-term storage. In the laboratory, handling free spermidine typically requires low-temperature operations under inert gas protection; even then, its weight often introduces non-negligible errors due to moisture absorption. Spermine trihydrochloride, on the other hand, is a white crystalline powder with a significantly increased melting point of 248 to 258°C, and it does not exhibit any "melting" problem under normal storage conditions. This change means that, under room temperature, light-protected, and sealed conditions, the trihydrochloride powder can be stably stored for more than two years, while the free base may degrade within weeks due to moisture absorption and oxidation under the same conditions. For active pharmaceutical ingredient (API) manufacturers, this stability translates to lower storage costs and longer product shelf life; for formulation developers, the crystalline powder means it can be accurately weighed, mixed with excipients, and compressed into tablets or capsules.
The improvement in solubility is equally significant. While free spermidine is soluble in water, its dissolution rate is slow, and high-concentration solutions require prolonged stirring to achieve complete clarification. In contrast, spermidine trihydrochloride has a solubility in water exceeding 100 mg/mL, dissolving rapidly with gentle agitation. This means it can be easily formulated into high-concentration stock solutions to meet the needs of cell experiments at concentrations ranging from nanomolar to millimolar, and is also convenient for administration via gavage or drinking water in animal experiments. Furthermore, it exhibits good solubility in ethanol, facilitating its applications in organic synthesis and materials science. From a thermodynamic stability perspective, spermidine trihydrochloride has a sublimation temperature of approximately 110°C and does not decompose during conventional heating and drying processes. This provides a broad operating window for determining the loss on drying and for heat sterilization processes.
Regarding safety and ease of handling, spermidine trihydrochloride is classified as a skin and eye irritant. Gloves and safety goggles are recommended during handling to avoid inhaling dust. However, compared to free spermidine, its dust emission is lower, making it safer for routine weighing and dispensing operations. Free spermidine, due to its viscous liquid state and high hygroscopicity, easily contaminates balances and lab surfaces during operation, making cleaning difficult; while trihydrochloride powder can be transferred using a weighing spoon like most solid chemicals, and only a wipe of the work surface is required after handling. From the perspective of raw material quality control, the key indicators for spermidine trihydrochloride include: purity not less than 98%, usually determined by argentometric titration or high-performance liquid chromatography; moisture content less than 5%, determined by Karl Fischer titration; heavy metals less than 10 ppm; and endotoxins less than 10 EU/mg. These are the requirements for injectable grade raw materials. Storage conditions are 4°C, sealed, protected from light, and dry. During transportation, it can be stored at room temperature for short periods, but high temperature and high humidity environments should be avoided.
This stable physicochemical property makes it suitable for various formulations, including oral tablets, capsules, sustained-release granules, sterile powders for injection, and high-purity reagents for cell biology, solving the industry pain point of difficulty in formulating free spermidine. Compared with other polyamine compounds such as putrescine and spermine, spermidine trihydrochloride has a moderate molecular weight, stronger ability to penetrate cell membranes and the blood-brain barrier, and significantly improved bioavailability, providing a physicochemical basis for its application in the intervention of systemic multi-system diseases.
Furthermore, spermidine trihydrochloride has a safer metabolic pathway. In vivo, it can rapidly dissociate into spermidine cations, participate in the body's polyamine cycle, and be excreted as non-toxic products after oxidation and acetylation metabolism, without the risk of tissue accumulation or the production of cytotoxic metabolic intermediates. This structural safety feature gives it a significant advantage in long-term intervention drugs and health products, and is one of the core reasons why it has passed the safety certification of authoritative institutions.
A "molecular platform" for autophagy induction and multi-target regulation
Spermidine trihydrochloride rapidly dissociates into biologically active spermidine molecules upon entering the body. Its core pharmacological effects revolve around autophagy induction, epigenetic regulation, mitochondrial quality control, oxidative stress inhibition, and inflammation homeostasis regulation. These pathways are highly conserved and exhibit high consistency across multiple species, forming a unique multi-pathway synergistic regulatory mechanism network.
- Its most fundamental and extensively studied mechanism is the potent induction of autophagy, primarily achieved by inhibiting the activity of acetyltransferase EP300. Spermidine can directly bind to the catalytic domain of EP300, reducing its acetylation modification level on autophagy-related proteins, relieving the inhibitory effect of acetylation on the ATG protein complex, thereby promoting LC3-II generation, autophagosome formation, and autolysosomal degradation. This accelerates the clearance of damaged organelles, misfolded proteins, lipofuscin, and intracellular toxic substances. This process is the core basis for its anti-aging, neuroprotective, and anti-inflammatory effects, and has been confirmed by numerous cell and animal experiments.
- Simultaneously, spermidine can further enhance autophagy initiation signals upstream by inhibiting the mTORC1 signaling pathway and activating the AMPK signaling pathway, forming a dual regulatory network. mTORC1 is a core inhibitor of autophagy, and spermidine relieves this inhibition by suppressing its activity. Meanwhile, AMPK, as an energy-sensing kinase, promotes the expression of autophagy-related genes upon activation. The synergistic effect of these two pathways ensures a stable increase in autophagy levels without causing cell damage due to excessive autophagy, achieving precise regulation of autophagy.
- At the epigenetic regulatory level, protonated spermidine molecules can embed into chromatin structures through electrostatic interactions, stabilizing the DNA double helix conformation, reducing DNA strand breaks and oxidative damage, and protecting genome stability. Furthermore, it can regulate histone acetylation and methylation patterns, inhibit the expression of genes related to aging-related secretory phenotypes, and upregulate the transcriptional activity of longevity-related genes, thus delaying cellular aging at the gene expression level. This is one of the important mechanisms by which it can extend the lifespan of model organisms.
- In terms of mitochondrial function maintenance and oxidative stress and inflammation regulation, spermidine can specifically promote mitophagy, clear dysfunctional mitochondria, reduce reactive oxygen species (ROS) accumulation, enhance respiratory chain complex activity and ATP production efficiency, and improve cellular energy metabolism. This is particularly important for high-energy-demand nerve cells, cardiomyocytes, and skeletal muscle cells. Simultaneously, it can directly scavenge some ROS and upregulate the expression of antioxidant proteins such as superoxide dismutase and glutathione peroxidase by activating the Nrf2/ARE pathway. It also inhibits NF-κB nuclear translocation and NLRP3 inflammasome activation, reduces the release of pro-inflammatory factors such as IL-1β, IL-6, and TNF-α, and alleviates chronic low-grade inflammation. Chronic inflammation is a common driver of aging, cardiovascular disease, neurodegeneration, and metabolic disorders.
Furthermore, spermidine can regulate cell membrane fluidity, maintain calcium homeostasis, promote neurotransmitter release and synaptic transmission, and enhance cognitive function and mood regulation. Its multi-pathway synergistic regulation rather than single-target blockade makes it significantly superior to most synthetic small molecule drugs in terms of safety and broad spectrum, and also provides solid mechanistic support for its application in the intervention of complex chronic diseases.
It is worth emphasizing that spermidine trihydrochloride, at conventional supplementation doses, does not cause significant fluctuations in polyamine concentrations in blood and tissues. A human clinical trial published in 2024 by Keohane et al. found that after 28 consecutive days of supplementation with 40 mg of high-purity spermidine trihydrochloride daily, no significant changes were observed in serum and urinary polyamine concentrations in subjects. This finding has important clinical implications: it indicates that spermidine supplementation does not simply "increase polyamine concentrations," but rather exerts its biological effects through more sophisticated mechanisms, such as regulating the hydroxybutyrination modification state of EIF5A or the acetyltransferase activity of EP300. In other words, spermidine acts more like a "molecular switch" than a "fuel supplement." This shift in understanding has profound implications for the positioning of spermidine trihydrochloride as a functional raw material: it is no longer simply regarded as a "polyamine precursor" but has been redefined as a "signaling pathway modulator."
Core Uses and Industrial Applications of Spermidine Trihydrochloride Powder
Spermidine trihydrochloride powder, with its stable physicochemical properties and powerful biological functions, has been widely used in drug development, health foods, special medical purpose foods, and basic life science research. Its applications are highly concentrated in age-related diseases, neurodegenerative diseases, cardiovascular diseases, metabolic disorders, and adjuvant cancer treatment, forming a diversified industrial application pattern.
In the field of aging and healthy lifespan extension, this raw material is currently the most thoroughly researched autophagy-inducible natural endogenous substance. Multiple animal experiments have confirmed that it can significantly extend the lifespan of yeast, nematodes, fruit flies, and mice, while improving muscle function, exercise endurance, cognitive ability, and immune response levels in older individuals, and delaying the appearance of age-related phenotypes. Based on this characteristic, it has become a core ingredient in anti-aging health foods and age management products, with product forms including oral capsules, tablets, and oral liquids, occupying an important position in the global health consumer market.
In the field of neurodegenerative diseases, spermidine trihydrochloride can significantly protect against Alzheimer's disease, Parkinson's disease, cerebral ischemia-reperfusion injury, and post-traumatic cognitive impairment by inhibiting neuroinflammation, reducing β-amyloid protein aggregation, decreasing tau protein hyperphosphorylation, and promoting synaptic plasticity and neurotrophic factor expression. Multiple preclinical studies have shown that it can improve learning and memory abilities in animal models, reduce neuronal apoptosis, and protect the integrity of the blood-brain barrier, demonstrating its potential as an adjunct to cognitive repair. Several candidate drugs have already entered early clinical trials.
In the field of cardiovascular diseases, epidemiological studies have shown a significant negative correlation between dietary spermidine intake and cardiovascular mortality. This ingredient can protect against heart failure, hypertension, atherosclerosis, and myocardial ischemia-reperfusion injury by improving myocardial mitochondrial function, reducing myocardial fibrosis, inhibiting vascular endothelial oxidative stress, and regulating lipid metabolism and blood pressure homeostasis. As an important candidate ingredient for cardiovascular protective drugs, it has been used in the development of various innovative drugs and is expected to provide new options for the adjunctive treatment of cardiovascular diseases.
Furthermore, spermidine can regulate cell membrane fluidity, maintain calcium ion homeostasis, promote neurotransmitter release and synaptic transmission, and enhance cognitive function and mood regulation. Its multi-pathway synergistic regulation rather than single-target blockade makes it significantly superior to most synthetic small molecule drugs in terms of safety and broad-spectrum activity, providing solid mechanistic support for its application in the intervention of complex chronic diseases.

Latest Research Directions and Industry Development Trends of Spermidine Trihydrochloride Powder
Research and industrialization development of Spermidine trihydrochloride powder mainly focuses on five areas: high-purity green manufacturing, novel delivery systems, expansion of clinical indications, combination therapy strategies, and in-depth analysis of its mechanism of action. This is driving its transition from basic research raw material to standardized drug development, with continuously improving industry maturity.
In the raw material production field, traditional plant extraction methods suffer from low content, high cost, large batch-to-batch variations, and difficulty in controlling impurities, failing to meet the large-scale requirements of pharmaceutical-grade raw materials. The current mainstream approach has shifted to synthetic biology fermentation preparation. Researchers have modified the metabolic pathways of engineered strains such as *E. coli* and *Saccharomyces cerevisiae*, enhancing the expression of key enzymes in polyamine synthesis and blocking branched metabolic pathways to achieve high-yield, low-cost, and sustainable biosynthesis. Combined with green purification processes such as membrane separation, ion exchange chromatography, low-pressure chromatography, and low-temperature recrystallization, a stable 99.9% ultra-high purity product can be obtained, meeting the high requirements of injectable formulations, ophthalmic preparations, and central nervous system drugs. These processes have gradually achieved industrial-scale scaling up to hundreds of kilograms, significantly reducing raw material production costs.
In terms of formulation and delivery technology, conventional oral formulations suffer from significant first-pass effects, limited bioavailability, and large fluctuations in blood drug concentrations, which restrict their clinical application. Currently, researchers are actively developing enteric-coated microspheres, phospholipid complexes, lipid nanoparticles, polymer sustained-release carriers, and nasal delivery systems. These formulation technologies optimize intestinal absorption efficiency, enhance blood-brain barrier penetration, prolong in vivo action time, and reduce dosing frequency. Brain-targeted delivery systems, particularly for neurodegenerative diseases, can achieve specific enrichment in the brain by modifying targeting ligands, significantly increasing intracranial drug concentrations, reducing systemic dosage and potential side effects, and providing a new technological pathway for the treatment of central nervous system diseases.
In terms of clinical research, an increasing number of randomized controlled trials are exploring new indications, breaking through traditional aging, neurological, and cardiovascular fields, and gradually expanding to areas such as post-COVID-19 cognitive impairment, sarcopenia, optic nerve protection in glaucoma, age-related hearing loss, ulcerative colitis, and autoimmune diseases. Partial Phase II clinical data show that spermidine trihydrochloride can effectively alleviate cognitive fatigue, improve muscle mass and function, delay optic nerve damage, and reduce intestinal inflammation, providing high-quality clinical evidence for its expanded clinical application and promoting its transformation towards standardized drugs.
Combination therapy studies have also become a research hotspot in recent years. Researchers have found that spermidine trihydrochloride, when used in combination with drugs such as rapamycin, memantine, and donepezil, can achieve synergistic effects on signaling pathways, while reducing the dosage of single drugs, minimizing adverse reactions, and expanding the therapeutic window. For example, combination therapy with donepezil can significantly improve cognitive function in Alzheimer's disease patients.
At the mechanistic level, the latest research reveals that spermidine can promote the proliferation of beneficial bacteria by regulating the gut microbiota structure, further increasing the body's polyamine levels, and forming a regulatory network of the gut-brain axis, gut-cardiovascular axis, and gut-metabolic axis, enriching the theoretical system of its mechanism of action. Furthermore, research on its application in stem cell regeneration, telomere homeostasis maintenance, and immune cell functional remodeling is also deepening, making its anti-aging and disease intervention theories more systematic and complete. With the increasing aging of the global population and the rapid development of aging medicine, the quality standards of Spermidine trihydrochloride powder are constantly being upgraded, and pharmacopoeias and industry standards in various countries are being established, driving its transformation from a common chemical raw material to a standardized API. In the future, it is expected to launch more approved drugs and functional products in the fields of neuroprotection, cardiovascular health, metabolic regulation and anti-aging.
Conclusion
Spermidine trihydrochloride powder, as a pharmaceutically stable form of spermidine, has become a highly valuable raw material for endogenous active substances in the modern pharmaceutical and health industries due to its unique molecular structure, excellent physicochemical stability, highly conserved autophagy activation mechanism, and broad disease intervention potential. Based on polyamines naturally present in the human body, it boasts high safety, mild action, and clear targets. It can serve as a key tool reagent in life science research and can also be used to develop drugs and functional products related to anti-aging, neuroprotection, cardiovascular health, and metabolic regulation. Against the backdrop of a high global incidence of age-related chronic diseases, it possesses broad market prospects and clinical translational value.
Although there is still room for improvement in oral bioavailability, central targeting efficiency, and large-scale long-term clinical evidence, its industrial maturity and clinical application scope are continuously expanding with the continuous advancement of green biomanufacturing technology, nanodelivery systems, and precision clinical research. From a pharmaceutical raw material perspective, spermidine trihydrochloride not only represents a successful paradigm for optimizing the salt formation of endogenous active substances but also provides a low-risk, high-potential development direction for aging intervention and adjuvant therapy for chronic diseases.
Xi'an Faithful BioTech Co., Ltd. cordially invites pharmaceutical companies and research institutions to learn about our comprehensive Spermidine trihydrochloride powder solutions. Our technical team is ready to discuss your specific needs, provide detailed product specifications, and arrange sample testing. Please contact allen@faithfulbio.com for highly competitive pricing, technical documentation, and personalized consultation services to ensure your bulk purchasing needs are reliably met.
References
- Madeo, F., Eisenberg, T., Pietrocola, F., & Kroemer, G. (2018). Spermidine in health and disease. Science, 359(6374), eaan2788.
- Eisenberg, T., Knauer, H., Schauer, A., Büttner, S., Ruckenstuhl, C., Carmona-Gutierrez, D., et al. (2009). Induction of autophagy by spermidine promotes longevity. Nature Cell Biology, 11(11), 1305–1314.
- Minois, N., Carmona-Gutierrez, D., & Madeo, F. (2021). Spermidine: a physiological autophagy inducer acting as an anti-aging drug in humans. Ageing Research Reviews, 69, 101353.
- Schwarz, C., Aumann, A., Depenbusch, M., & Spitz, U. (2023). Dietary spermidine intake and cognitive function: Results from the population-based SHIP study. Nutrients, 15(2), 348.
- Guo, C., Liu, J., & Klionsky, D. J. (2022). Spermidine induces cytoprotective autophagy in mammalian cells. Autophagy Reports, 1(1), 112–122.
- Wirth, M., Schwarz, C., & Kiehntopf, M. (2022). Spermidine: A novel biomarker and therapeutic target for cardiovascular diseases. European Journal of Heart Failure, 24(3), 388–398.
- LaRusso, N. F., & Feldstein, A. E. (2020). Spermidine as a potential therapeutic for nonalcoholic fatty liver disease. Hepatology Communications, 4(12), 1784–1796.



_1765011147056.webp)







