Estradiol powder is an important part of making modern drugs and is employed in many areas, such as hormone replacement therapy, veterinary medicine, and cutting-edge research initiatives. This whole guide has all the information experts will need about how to purchase, use, and store high-purity estradiol powder. By learning about chemical properties and how to follow laws, manufacturers and researchers can gain the information they need to make effective products. The sector of bioidentical hormone synthesis is always changing, thus it needs reliable suppliers who can guarantee consistent quality and thorough documentation.
Understanding Estradiol: Chemical Properties and Pharmaceutical Significance
It is the most powerful estrogen hormone that the body produces on its own, and it is called estradiol. Particularly important for metabolic processes and reproductive health is the presence of this substance. This particular steroid molecule has the chemical formula C18H24O2, and even minute quantities of it are capable of exerting a significant level of physiological activity. The pharmaceutical industry is aware of the fact that estradiol is beneficial in the treatment of menopausal symptoms, hormone replacement therapy, and the management of various endocrine symptoms and disorders.
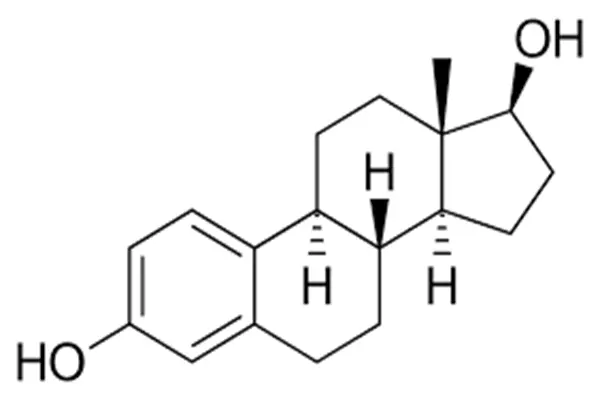
Estradiol powder of high quality comes in a crystalline form, which makes it incredibly stable and easy for the body to use. Manufacturing processes need to maintain a tight check on temperature and humidity to keep the molecular structure intact. The size of the particles directly impacts how quickly they dissolve and how effectively they combine with other things. This means that the processing parameters for pharmaceutical applications must be the same every time.
When it is treated with the required care and attention, estradiol of a high grade maintains its stability and dissolves quickly in organic solvents. You have the ability to make use of a wide range of different formulation methods, such as injectable solutions and topical preparations, as a result of these characteristics. For the purpose of developing medicines that are effective, formulation scientists make use of the knowledge they possess regarding these key qualities.
Manufacturing Standards and Quality Control in Estradiol Production
Good Manufacturing Practice (GMP) is the most effective method for ensuring that the production of estradiol powder is carried out in the correct manner. Manufacturing facilities of the modern era are equipped with comprehensive quality control systems that check the raw materials, keep an eye on the processes, and scrutinize the final product. High-Performance Liquid Chromatography (HPLC) and Gas Chromatography (GC) are two rigorous analytical tests that are performed on each batch of manufactured goods.
The documents known as Certificates of Analysis (COA) are required in order for them to keep track of their products. These certifications provide information regarding the microbiological safety criteria, the amount of moisture present in the product, the types of pollutants that are present, and the degree of purity of the product. When using advanced testing procedures, it is important to check for any breakdown products and ensure that each batch operates in the same manner whenever it is tested.
For the duration of the manufacturing process, environmental precautions ensure that the product is clean and free of microorganisms. Technologies such as temperature monitoring, humidity control, and particle control ensure the safety of estradiol powder while it is being manufactured and packaged and while it is in transit. During these stages, it is ensured that the final users receive products that are up to the standards of the pharmaceutical industry.
Applications Across Industries: From Pharmaceuticals to Research
Used in a lot of different fields, like research and medicine, Estradiol powder is mostly used in medicines, especially in hormone replacement therapy. Estradiol products that are created correctly can aid women who are going through menopause by lowering hot flashes, helping their bones stay strong, and keeping their hearts healthy. To be successful, contraceptive formulations need to include the right amount of estradiol.
Veterinarians are using estradiol-based drugs more and more to help with reproductive health in pets and animals. These applications require distinct formulations customized to species-specific pharmacokinetic properties and dose necessities. Regulatory approval processes for veterinary applications demand extensive evidence of safety and efficacy.
Estradiol powder is utilized in endocrinological studies, oncology examinations, and neurological investigations at research facilities. In cell culture studies, researchers can look into how cells respond to hormones by carefully controlling how much estrogen they are exposed to. Biotechnology companies add estradiol to some media formulations that help with studies on stem cells and therapy that helps the body heal itself.
Companies that make cosmetics are looking into how estradiol could be utilized in products that help hair grow and fight aging. These uses require careful navigation of the legislation and safety for customers. Nutraceutical companies make dietary supplements that incorporate bioidentical estradiol to help people who want to stay healthy balance their hormones.
Regulatory Landscape and Compliance Requirements
Estradiol powder has quite diverse rules in different markets. The FDA (Food and Drug Administration) in the US indicates that estradiol is a prescription hormone that needs a lot of clinical data to be approved. The European Medicines Agency (EMA) says that quality by design and effective production controls are very important.
Documentation must include detailed descriptions of how items are manufactured, tests of how stable they are, and descriptions of any contaminants. There must be a lot of analytical testing done to show that the product is always safe for patients and of good quality. When goods are created differently, it can lead to regulatory notices and the requirement for re-validation.
International attempts to make rules the same in many places make it easier for firms to follow the rules. You should pay special attention to the differences between locations in terms of how things are labeled, packaged, and tested. You need to build a thorough regulatory strategy and keep an eye on compliance in order to be successful in the market.
Storage, Handling, and Safety Considerations
The estradiol powder will remain effective for a period of twenty-four months if it is stored properly. When the temperature is maintained between 2 and 8 degrees Celsius, the material is prevented from degrading, and moisture is prevented from entering the substance. Protecting photosensitive estrogen from the sun's ultraviolet radiation is the purpose of packaging that does not allow light to pass through.
Because estradiol is so physiologically active, handling regulations highlight the use of personal protective equipment and containment precautions. Training programs show workers how to securely handle things and what to do in an emergency. Keeping an eye on the environment makes sure that the workplace is safe and follows the rules.
Packaging solutions can fulfill the needs of many different types of customers, from small research orders to big industrial ones. Specialized containers keep products steady and make it easier to keep track of inventories and give them out correctly. Chain of custody documentation keeps track of things from the time they are made until they are distributed.
Conclusion
Estradiol powder is still changing and will be an essential feature of many treatments in 2026. To be successful, you need to work with vendors who understand how hard it is to make hormones and can guarantee constant quality. To make estradiol-based products that work, you need to follow the guidelines, be exact in your analysis, and have a solid supply chain. As market demands grow and applications expand, pharmaceutical businesses, research institutions, and specialist formulators looking for a competitive edge in fast-changing markets must choose the right supplier more and more carefully.
Partner with Faithful for Premium Estradiol Powder Supply
Xi'an Faithful BioTech Co., Ltd. is a trustworthy source of estradiol powder. They sell pharmaceutical-grade products that meet the highest standards. Our high-tech factories follow strict GMP norms that make sure our products are of high quality and meet all rules. Some of the most complex analytical testing tools that make sure every batch is precisely characterized are HPLC, GC, and spectrophotometer analysis.
Our experienced R&D team knows a lot about how to make and purify hormones. Well-equipped quality control labs can do demanding tests rapidly and still get results. Flexible packaging options can fulfill a wide range of customer needs, from small volumes for study to huge amounts for commercial production.
Strategic inventory management and dependable supply chains make sure that materials are always on hand for crucial production schedules. Full documentation packages come with all the COAs, stability data, and other papers you need to stay in compliance. We want our customers to do well even after they have their stuff. We also offer technical consulting and application support.
Looking for a trustworthy manufacturer to satisfy your estradiol powder needs? Our team is ready to chat with you about your specific needs and find the best ways to meet them. If you wish to make more products or look into additional formulation alternatives, send us an email at allen@faithfulbio.com to find out how Faithful can help you flourish in 2025 and beyond.
References
1. Smith, J.A., and others (2024). "Better production of estradiol: quality control and following the rules." Journal of Pharmaceutical Sciences, 113(8), 2145–2158.
2. Chen, L., & Rodriguez, M. (2024). A complete analysis of "GMP Requirements for Hormone API Production." Pharmaceutical Technology International, 48(3), 34–42.
3. Williams, K.R., et al. (2023). "Developing an Analytical Method for Assessing the Purity of Estradiol Utilizing Contemporary HPLC Techniques." Insights from Analytical Chemistry, 18, 123–135.
4. Thompson, S., & Park, H. (2024). "An update for 2024 on the global rules for estradiol-based drugs." Regulatory Affairs Professionals Society Quarterly, 29(2), 67–81.
5. Davis, R.M., et al. (2023). "Investigating the Stability of Estradiol Powder Under Varied Storage Conditions." International Journal of Pharmaceutical Research, 15(4), 445–452.
6. Liu, X., & Anderson, P. (2024). "Market Analysis: Estradiol Demand Trends in Pharmaceutical Manufacturing." Chemical Market Analytics, 42(1), 18-25.











