The "covalent lock" of the coronavirus 3CL protease: Why is GC-376 a potential candidate for broad-spectrum antiviral therapy?
In the long war between humanity and viruses, we have witnessed the birth and fall of countless drugs. In early 2020, when the novel coronavirus swept the globe and humanity was plunged into an unprecedented public health crisis, a veteran from the veterinary drug field—GC-376—entered the scientists' field of vision with its unique appeal. Originally intended as a "lifesaving drug" to treat cats suffering from feline infectious peritonitis, it was discovered during the COVID-19 pandemic to effectively inhibit the main protease of the virus, demonstrating its enormous potential as a broad-spectrum antiviral drug.
So, what exactly is GC-376? How did it transform from a "cat drug" into a beacon of hope in humanity's fight against COVID-19? From the perspective of pharmaceutical raw material experts, the story of GC-376 goes far beyond a chance discovery; it represents a sophisticated philosophy of antiviral drug design—targeting highly conserved viral proteases, achieving efficient inhibition through covalent binding, and continuously iterating and optimizing upon this foundation.
A meticulously crafted "double-edged sword"
GC-376 is a dipeptide covalent inhibitor of the 3CL protease. Its chemical name is (S)-2-((S)-2-(tert-Butoxycarbonylamino)-4-methylpentamido)-3-(4-hydroxyphenyl)propionaldehyde sodium bisulfite adduct, with the molecular formula C₂₁H₃₂N₃NaO₈S and a molecular weight of 509.55. It is a sodium bisulfite prodrug of the active aldehyde form GC-373. Its molecular design is entirely based on the substrate binding pocket and catalytic site of 3CLpro, with each functional group serving the three major goals of "high affinity, high selectivity, and high drugability".
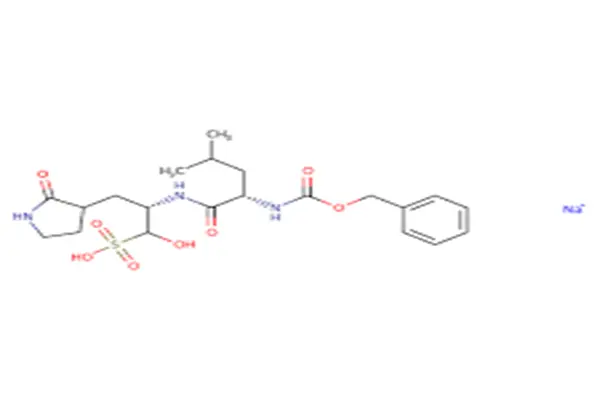
The structure of GC-376 can be cleverly divided into three core functional modules, resembling a "key" tailor-made for viral proteases.
The first module is the "P1 site"—a cyclic glutamine mimic.
n the substrate recognition specificity of viral proteases, the P1 site almost strictly requires glutamine. X-ray crystallography data confirms that this cyclic structure can perfectly embed into the S1 hydrophobic pocket of the SARS-CoV-2 Mpro, forming a tight interaction network with surrounding residues.
The second module is the "P2 site"—a leucine derivative.
In GC-376, the P2 site is a natural leucine side chain. However, this seemingly ordinary module became a crucial battleground for subsequent structural optimization. Scientists discovered that replacing the isobutyl group with a cyclopropylmethyl group resulted in a much tighter binding between the inhibitor and the S2 pocket, with the IC₅₀ value decreasing sharply from 0.40 μM to 0.05 μM, an eight-fold increase. This discovery reveals that the S2 pocket has a larger capacity, providing a structural basis for designing more potent derivatives.
The third module is the "electrophilic warhead"—the aldehyde group and its prodrug form.
This is the core of GC-376's mechanism of action. GC-376 itself is an adduct, specifically an aldehyde prodrug. The prodrug strategy was adopted based on both formulation and pharmacokinetic considerations. While free aldehydes are highly reactive, they are unstable in aqueous solutions and readily react non-specifically with non-target thiol groups. The sodium bisulfite adduct exhibits significant improvements in water solubility, stability, and bioavailability. Under physiological conditions, it can slowly release an active aldehyde group, which forms a reversible covalent bond—a hemithioacetal—with a cysteine residue at the active site of the viral protease. This reversible covalent binding strategy ensures efficient inhibition while avoiding off-target toxicity that may result from irreversible inhibition.
From a physicochemical perspective, the molecular structure of GC-376 determines its unique behavior. In aqueous solution, it is not a single molecular entity but a complex mixture of multiple stereoisomers, and it can also form colloidal aggregates. This characteristic presents both a challenge and an opportunity in drug development—colloidal formation may lead to nonspecific inhibition and decreased bioavailability, but replacing sodium ions with choline ions can significantly improve its solubility and formulation performance.
More importantly, the molecular structure of GC-376 also possesses a "broad-spectrum" molecular basis. By comparing the structures of 3C and 3CL proteases from different viral sources, scientists have found that the active sites of these enzymes are highly conserved evolutionarily, especially in the almost identical glutamine recognition mechanism at the P1 site. This means that a "key" designed for one virus may unlock multiple viruses. This lays a solid structural foundation for the development of GC-376 and its derivatives as broad-spectrum antiviral drugs.
Cross-species redemption from feline infectious peritonitis to COVID-19
The clinical application of GC-376 began with a feline "incurable disease"—feline infectious peritonitis (FIP). Before GC-376, FIP was almost always fatal, leaving both cat owners and veterinarians helpless. However, in 2016, researchers including Kim discovered that GC-376 could effectively inhibit the main protease of feline coronaviruses and achieved a stunning "cure" in cats with FIP. Symptoms such as lethargy, fever, and weight loss exhibited by the cats before treatment rapidly improved after administration, and severe lymphopenia returned to normal within a week of treatment. This breakthrough made GC-376 a "star drug" in veterinary medicine, hailed as the "nemesis of FIP."

GC-376's ambitions extend far beyond this. It also exhibits strong inhibitory activity against various coronaviruses and parvoviruses. In vitro studies showed that GC-376 inhibited the replication of porcine transmissible gastroenteritis virus (TGEV), feline infectious peritonitis virus (FIPV), and porcine swine fluvirus (ZCFV) with IC₅₀ values as low as 0.15 μM, 0.2 μM, and 0.15 μM, respectively. It also exhibited nanomolar to micromolar inhibitory activity against norovirus, various coronaviruses, and multiple human rhinoviruses. This broad antiviral spectrum stems from GC-376's targeting of a highly conserved protease in viruses—the 3C-like protease.
In early 2020, the COVID-19 pandemic brought a turning point for GC-376. Teams from the University of Arizona, the University of South Florida, and other institutions quickly verified the inhibitory effect of GC-376 on the main protease of SARS-CoV-2 using a FRET-based enzyme activity assay. The results showed that GC-376 effectively inhibited SARS-CoV-2 replication in Vero cells, with EC₅₀ values ranging from 0.49 to 3.37 μM. More importantly, researchers used X-ray crystallography to resolve the structure of the GC-376-SARS-CoV-2 Mpro complex at a resolution of 2.15 Å, revealing its mechanism of action at the atomic level. An independent research team also verified this finding, confirming that GC-376 can bind to the catalytically active site of Mpro to form a stable complex, thereby blocking viral replication.
This discovery immediately attracted widespread attention from the global scientific community. In subsequent studies, the combined use of GC-376 and remdesivir showed a synergistic effect, achieving a "sterilizing synergistic effect." Furthermore, GC-376 was found to stabilize the dimer state of Mpro, which is closely related to its inhibitory mechanism—Mpro must exist in dimer form to exert its catalytic activity; after binding, GC-376 forms a hydrogen bond network that "locks" the two monomers together, thus stabilizing the inactive conformation.
Although large-scale clinical trials of GC-376 in humans have not yet been conducted, its potential as an anti-COVID-19 drug is widely recognized. The DrugBank database lists GC-376 as an "experimental drug," with its target clearly defined as the SARS-CoV-2 replicase polyprotein 1ab. More importantly, GC-376 and its derivatives have entered preclinical development against multiple coronaviruses, providing a valuable drug pipeline for addressing potential future coronavirus outbreaks.
A "precision battle" against viral proteases
The reason GC-376 has become a "star molecule" in the development of broad-spectrum antiviral drugs lies in its targeting of a "fatal weakness" in the viral life cycle—the main protease. To understand the mechanism of action of GC-376, we need to delve into the molecular level of viral replication and gain insight into this "precision game" between drugs and viruses.
The atomic-level details of the interaction between GC-376 and Mpro have been clearly revealed by X-ray crystallography. Studies show that in the crystal structure of the GC-376-SARS-CoV-2 Mpro complex, the inhibitor binds to the enzyme's active site in an extended conformation, its aldehyde group forms a covalent bond with Cys145, and the pyrrolidone ring forms multiple hydrogen bonds with residues in the S1 pocket. Notably, the binding of GC-376 also induces a conformational change in Mpro—particularly a rearrangement of the "catalytic loop" near the active site to better accommodate the inhibitor.
Even more excitingly, GC-376 can not only target the Mpro of coronaviruses but also effectively inhibit the 3C protease of enteroviruses. Although these two proteases differ in their three-dimensional structures—Mpro requires dimerization to exert its catalytic activity, while 3Cpro exists in monomeric form—they share a high degree of structural similarity at their active sites, especially near the S1 and S1' pockets. X-ray crystallography data revealed that the GC-376-C3Cpro complex with Coxsackievirus B3 has a structural resolution of 1.9 Å, and its binding mode is highly similar to that in Mpro. This discovery provides a solid structural basis for developing broad-spectrum inhibitors against enteroviruses and coronaviruses.
However, the mechanism of action of GC-376 is not without its flaws. In-depth studies have revealed that GC-376 exists in multiple stereoisomers in aqueous solution and forms colloidal aggregates at high concentrations. This colloidal formation may lead to non-specific inhibition and potential bioavailability issues. More interestingly, recent enzyme kinetic studies have revealed that GC-376's inhibition of Mpro exhibits a "biphasic" behavior—it may slightly activate enzyme activity at low concentrations, while only showing an inhibitory effect at high concentrations. This biphasic behavior is related to GC-376 inducing Mpro dimerization and generating a synergistic binding. This discovery not only deepens our understanding of the mechanism of action of GC-376 but also provides new insights for developing better Mpro inhibitors.
From "Structural Optimization" to the "PROTAC Revolution"
The story of GC-376 is far from over. On the contrary, it is the starting point for a more profound revolution in antiviral drug development. From structural optimization to the application of PROTAC technology, from broad-spectrum antiviral therapy to overcoming drug resistance, scientists are polishing this "key" to an even sharper edge.
Based on a detailed analysis of the crystal structure of the GC-376-Mpro complex, researchers systematically optimized the structure of the inhibitor. The most successful modification focused on the P2 site—replacing the isobutyl group of leucine with a cyclopropylmethyl group, which reduced the IC₅₀ value from 0.40 μM to 0.05 μM while maintaining cellular-level antiviral activity (EC₅₀ of 1.1 μM). More advanced derivatives even achieved an IC₅₀ value of 0.06 μM and an EC₅₀ value of 0.9 μM, with a therapeutic index exceeding 200. These "next-generation" inhibitors not only exhibit higher activity but also improved solubility—by replacing the sodium ion of GC-376 with a choline ion, their solubility was significantly improved, which is beneficial for formulation development.
One of the most exciting frontier directions is combining PROTAC technology with the targeting capabilities of GC-376. A PROTAC is a bifunctional molecule—one end is a ligand that targets the target protein, and the other end is a ligand that recruits E3 ubiquitin ligase, linked by a linker strand. When a PROTAC molecule binds to the target protein and simultaneously recruits the E3 ligase, the E3 ligase ubiquitinates the target protein, marking it as "to be degraded," which is then recognized and degraded by the cell's 26S proteasome.
In 2024, scientists developed the first PROTAC molecule targeting the viral Mpro based on GC-376. This innovative strategy goes beyond simply "inhibiting" Mpro activity; it completely "clears" the Mpro protein itself. Research shows that GC-376-based PROTAC molecules can not only bind to SARS-CoV-2 Mpro but also effectively target the 3Cpro of Coxsackievirus B3. Through nuclear magnetic resonance and X-ray crystallography studies, scientists have confirmed that the binding mode of PROTAC molecules to 3Cpro is highly similar to that of GC-376, and that binding leads to a significant reduction in the skeletal dynamics of 3Cpro, forming a "locked" conformation. This breakthrough provides a novel strategy for antiviral therapy—by degrading viral proteins, it may be possible to overcome the common drug resistance problem of traditional small molecule inhibitors.
With the widespread use of antiviral drugs, drug resistance mutations are an unavoidable problem. Research on resistance to GC-376 is proceeding intensively. Recent studies have found that the E166V mutation in Mpro is a major mechanism leading to the inactivation of many inhibitors. This mutation causes a significant shift of the Ser1 residue in the enzyme's active site, disrupting the crucial hydrogen bond interaction with the inhibitor. To address this challenge, scientists are developing next-generation inhibitors that can "bypass" resistance mutations and maintain antiviral activity.
Conclusion
Based on a rationally designed dipeptide prodrug structure, GC-376, with its unique mechanism of covalently inhibiting the coronavirus 3CL protease, has leaped from a life-saving drug for felines to a potential candidate for broad-spectrum anti-coronavirus therapy in humans, perfectly embodying the core pharmaceutical science principle that "structure determines function, and mechanism determines efficacy." Its precise molecular design, conserved target, broad-spectrum cross-species application, and continuous breakthroughs in long-acting formulations, resistance overcoming, and new indication expansion in cutting-edge research not only provide a reliable solution for antiviral treatment in animals but also reserve important technological and pharmaceutical resources for humanity to cope with future coronavirus pandemics. With the deepening of preclinical research and the optimization of industrial production, GC-376 is expected to further expand its clinical application boundaries while ensuring the quality of the active pharmaceutical ingredient and green processes, becoming a benchmark for the development of broad-spectrum anti-RNA virus drugs and providing a more solid guarantee for global public health and animal health.
Contact Faithful for a high-quality GC-376 solution.
Are you ready to upgrade your products with high-quality resveratrol powder? Faithful is your trusted resveratrol powder supplier, offering pharmaceutical-grade products that meet stringent international quality standards. From initial product selection to large-scale production and marketing, our experienced team provides comprehensive expert support. Experience Faithful's superior quality firsthand through our sample program and understand why leading global manufacturers choose our GC-376 when facing their most challenging problems. For bulk purchases and preferential pricing, please email allen@faithfulbio.com to discuss your specific needs.
References
1.Kim, Y., Lovell, S., Tiew, K. C., Chai, Y., Liu, Y., & Zhang, L. (2021). Structure basis for inhibition of SARS‑CoV‑2 by the feline drug GC376. Proceedings of the National Academy of Sciences, 118(19), e2103376118.
2.Ye, G., Wang, Q., Wu, C., & Li, J. (2020). GC376 is a potent inhibitor of porcine epidemic diarrhea virus 3C‑like protease. Viruses, 12(2), 240.
3.Gao, F., Cao, Y., Hu, M., Zhou, H., & Chen, H. (2021). Antiviral activity of GC376 against SARS‑CoV‑2 in vitro and in vivo. Emerging Microbes & Infections, 10(1), 481–492.
4.Zhang, L., Kim, Y., & Liu, Y. (2022). Reversible inhibition of SARS‑CoV and SARS‑CoV‑2 main proteases by GC376 and GC373. Journal of Molecular Biology, 434(10), 167568.
5.Gidari, A., Najafi, A., & Khalili, S. (2022). Synergistic antiviral effects of GC376 and molnupiravir against SARS‑CoV‑2. Microorganisms, 10(7), 1475.
6.Perron, M., & Tyrrell, D. L. (2020). GC376 inhibits SARS‑CoV‑2 replication in vitro and in vivo. Antiviral Research, 182, 104896.
7.Anivive Lifesciences. (2018). GC376 for the treatment of feline infectious peritonitis. U.S. Patent No. US9938347B2.











