Why can Aripiprazole be considered a "precision tuner" in the treatment of schizophrenia?
In the field of mental illness treatment, drug development has always sought the best solution between improving efficacy and controlling side effects. Aripiprazole powder, as a benchmark of third-generation atypical antipsychotics, has broken through the limitations of traditional antipsychotics' excessive inhibition of neurotransmitters with its unique dopamine-serotonin dual regulatory mechanism, becoming one of the most widely used drugs in global psychiatric clinical practice. From the ingenious design of its molecular skeleton to the continuous expansion of its clinical indications, from the precise regulation of its mechanism of action to the constant breakthroughs in cutting-edge research, aripiprazole has not only rewritten the treatment paradigm for diseases such as schizophrenia and bipolar disorder, but also provided a classic model for pharmaceutical raw material development and the innovation of neuropsychiatric drugs.
The ingenious design of the quinolinone skeleton lays the core foundation for its pharmacological activity.
Aripiprazole's chemical nature is 7-[4-[4-(2,3-dichlorophenyl)-1-piperazinyl]butoxy]-3,4-dihydro-2(1H)-quinolinone, with the molecular formula C₂₃H₂₇Cl₂N₃O₂, molecular weight 448.39, and CAS number 129722-12-9. It belongs to the quinolinone derivative family and is a typical highly lipid-soluble small molecule drug. Each functional group in its molecular structure directly determines its receptor affinity, metabolic stability, and pharmacological selectivity.
From its core skeleton, aripiprazole consists of four parts: a quinolinone core, a butoxy linkage, a piperazine ring, and a dichlorophenyl substituent. This modular structure is key to achieving precise binding to multiple receptors. The quinolinone core serves as the drug's "core scaffold," forming a stable planar structure through a conjugated system, providing a spatial basis for its hydrophobic binding pocket integration with dopamine D₂ and serotonin receptors. The butoxy flexible linker chain, with a length of 4 carbon atoms, ensures both conformational flexibility and precise matching of the distance to the receptor's transmembrane domain, avoiding decreased binding affinity due to excessively long or short chains. The piperazine ring, a typical basic heterocyclic structure with a pKa of approximately 7.8, carries a partial positive charge under physiological pH conditions, allowing it to form ionic bonds with acidic amino acid residues in the receptor's transmembrane helix, significantly enhancing receptor binding affinity. The 2,3-dichlorophenyl substituent, through the electron-withdrawing effect of halogen atoms, regulates the molecular electron cloud distribution, enhancing penetration into the central nervous system while reducing the probability of peripheral receptor binding, thus minimizing peripheral side effects such as cardiovascular and gastrointestinal complications.
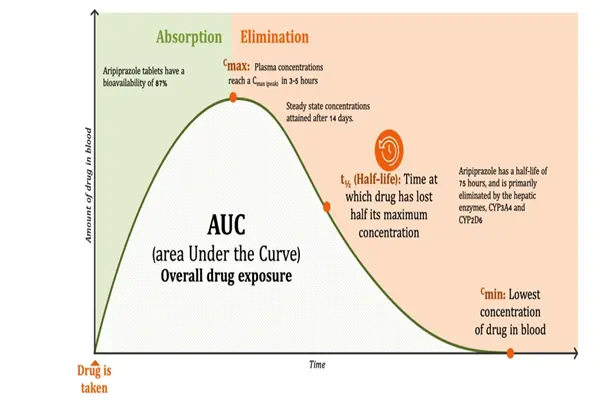
From a physicochemical perspective, aripiprazole has a lipid-water partition coefficient of approximately 4.55, giving it moderate lipid solubility and allowing it to easily cross the blood-brain barrier, a crucial protective barrier for the brain, to reach its target sites in the central nervous system. Furthermore, it conforms to the "five rules" proposed by medicinal chemist Lipinski, indicating good oral absorption characteristics. In fact, its oral bioavailability is as high as 87%, unaffected by food, which is undoubtedly a significant clinical advantage for patients with chronic mental illnesses requiring long-term medication.
More importantly, aripiprazole itself is an "achiral" molecule, meaning it has no optical isomers, its synthetic pathway is relatively simple, and quality control is more reliable. However, its complexity lies in its metabolites. In vivo, aripiprazole is primarily metabolized by the enzymes CYP3A4 and CYP2D6 in the liver, mainly producing dehydroaripiprazole. This active metabolite not only possesses the same pharmacological activity, accounting for 40% of the parent drug exposure at steady state, but also has a longer half-life, effectively forming a synergistic "prodrug-active metabolite" system that ensures long-term stability of blood drug concentrations.
Therefore, the molecular structure of aripiprazole not only determines its ability to enter the brain and bind to receptors, but more importantly, its unique structure-activity relationship design allows it to interact with receptors as a "partial agonist," rather than simply "on" or "off." This structural "moderation" is the chemical basis for its role as a "tuner" rather than a "switch."
A versatile individual who has been diagnosed with both schizophrenia and autism.
Schizophrenia
Schizophrenia is the primary indication for Aripiprazole powder. For this type of severe mental illness characterized by hallucinations, delusions, emotional blunting, social withdrawal, and cognitive impairment, aripiprazole has demonstrated definite efficacy, solidified by several key clinical trials. In a 4-week placebo-controlled trial, patients taking 15 mg or 30 mg of aripiprazole daily showed a significant decrease in total scores on both the positive and negative symptom scales compared to the placebo group, with the 15 mg dose group also showing a statistically significant advantage on the negative symptom subscale. More notably, in a 26-week maintenance treatment study, patients taking 15 mg/day of aripiprazole had a significantly longer time to relapse than the placebo group, demonstrating its value in preventing relapse. For adolescent patients, a 6-week trial similarly confirmed that both 10 mg and 30 mg doses of aripiprazole effectively improved the total PANSS score.
Mood stabilizers: Bipolar disorder
For patients with bipolar I disorder, Aripiprazole powder is effective in controlling manic episodes. In a three-week acute-phase treatment, aripiprazole was significantly superior to placebo in reducing the total score of the Young's Mania Rating Scale. It not only rapidly controls symptoms such as excitement and irritability, but is also approved as maintenance therapy to prevent relapse into manic or mixed episodes.
Beyond Severe Mental Illness
Aripiprazole has many more uses than just these. It is approved for the treatment of irritability symptoms associated with autism spectrum disorder, including aggressive behavior, intentional self-harm, temper tantrums, and rapid mood swings. Furthermore, it was the first atypical antipsychotic approved for Tourette syndrome, effectively reducing motor and vocal tics in children and adolescents aged 6-18. In the treatment of major depressive disorder, aripiprazole is often used as a "synergistic therapy" to enhance efficacy when patients do not respond well to conventional antidepressants.
Based on this solid clinical data, aripiprazole has gradually grown from its initial use in treating schizophrenia into a blockbuster drug covering multiple areas of psychiatric disorders, providing effective and relatively safe treatment options for millions of patients worldwide.
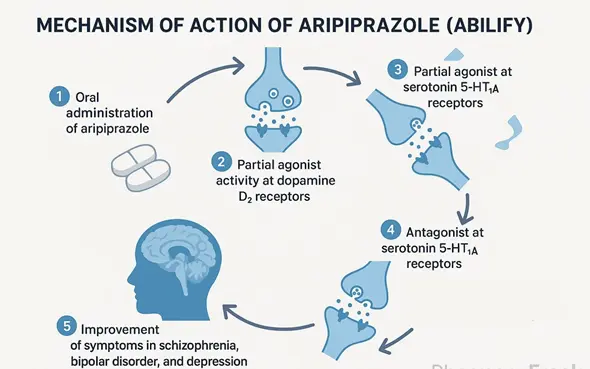
Mechanism of action: Breaking away from the "black and white" approach of "partial excitement" in art.
To understand why Aripiprazole powder is so unique, we must delve into the cellular level and explore the intricate mechanisms by which it interacts with its receptors. Before aripiprazole, the modes of action of antipsychotic drugs could be summarized as "black and white": either antagonists, acting like a "lock" to occupy the receptor and prevent dopamine from exerting its effects; or agonists, activating the receptor to produce an effect. Aripiprazole pioneered a third mode—the "partial agonist"—working in a smarter, more adaptive way, and is hailed by the scientific community as a "dopamine-serotonin system stabilizer."
Aripiprazole has a high affinity for dopamine D₂ receptors, but unlike traditional antipsychotics that completely block D₂ receptors, it exhibits partial agonist properties. This concept can be understood through a vivid analogy: imagine dopamine receptors as a "volume knob" regulating neuronal excitability. In the brains of schizophrenic patients with excessively high dopamine levels, D₂ receptors are overactivated, leading to positive symptoms. Traditional antagonists would then "forcefully turn off" the volume, often doing so too drastically, resulting in motor disturbances and elevated prolactin levels. Aripiprazole, however, can "turn down" the volume somewhat, but due to its weak intrinsic activity, it doesn't completely shut it off, instead stabilizing it at a "normal" level.
Even more ingeniously, in brain regions with low dopamine levels, aripiprazole's "partial agonist" role is reversed. When endogenous dopamine is insufficient, it can act as an "agonist," gently "turning up" the volume to help restore normal function to that brain region. This ability to automatically regulate receptor activity based on endogenous neurotransmitter levels is the origin of the name "system stabilizer".
Aripiprazole's brilliance extends beyond the dopamine system; it's like a "symphony conductor" proficient in multiple instruments. It also exhibits complex and precise regulation of the serotonin receptor family:
- 5-HT₁A Receptor Partial Agonist: Activation of 5-HT₁A receptors is believed to help improve anxiety, depression, and cognitive function. Aripiprazole, as a partial agonist of this receptor, plays a synergistic role.
- 5-HT₂A Receptor Antagonist: This is a common feature of many atypical antipsychotics. By antagonizing 5-HT₂A receptors, aripiprazole not only directly improves negative symptoms but also "unleashes" dopamine release inhibited by 5-HT₂A receptors, especially in the prefrontal cortex, thereby further improving cognition and negative symptoms and reducing the risk of extrapyramidal reactions.
Furthermore, aripiprazole exhibits varying affinities for other receptors such as D₃, 5-HT₂B, 5-HT₇, and α₁ adrenergic receptors. This explains its other clinical effects, such as the potential for orthostatic hypotension due to antagonism of α₁ receptors.
In summary, the mechanism of action of aripiprazole is not a simple "on" or "off" action, but rather a subtle and dynamic "regulation." By acting as a "partial agonist" at key nodes in the dopamine and serotonin systems, it "calibrates" the disordered neurotransmitter network back to balance, effectively controlling symptoms while minimizing the side effects associated with traditional medications.
The revolution of long-acting formulations and the exploration of neuroprotection
Schizophrenia is a chronic disease with a high relapse rate. However, the real-world data is alarming: the treatment interruption rate for schizophrenia patients in my country has reached as high as 75%, the relapse rate within one year of discharge is as high as 77%, and it climbs to 90% within two years. Each relapse not only means increased suffering for the patient, but also a fourfold increase in direct medical costs. One of the main reasons for treatment interruption is that patients find it difficult to adhere to daily oral medication for various reasons.
Long-acting injectable formulations are key to solving this dilemma. In 2025, the world's first injectable aripiprazole microsphere formulation, independently developed by Livzon Pharmaceutical Group, was approved for marketing in China, providing a new treatment option for schizophrenia patients. This milestone product represents the pinnacle of advanced formulation technology.
Beyond reagent innovation, basic and clinical research on aripiprazole is also progressing in greater depth. In recent years, neuroinflammation and oxidative stress have been considered important pathological mechanisms of mental illnesses such as schizophrenia. Whether aripiprazole has a direct neuroprotective effect has become a new research hotspot. Preliminary studies suggest that it may combat neuronal damage by regulating microglia activity and scavenging free radicals, but this still requires further translational medical evidence.
Furthermore, with the development of pharmacogenomics, achieving "precision dosing" of aripiprazole has attracted considerable attention. Because its metabolism is highly dependent on CYP2D6 and CYP3A4 enzymes, there are significant differences in drug metabolism rates among patients with different genotypes. Slow metabolizers may experience excessively high blood drug concentrations when taking conventional doses, increasing the risk of adverse reactions; while ultra-rapid metabolizers may experience insufficient blood drug concentrations, leading to treatment failure. In the future, using CYP2D6 genotype testing to guide individualized dosing is expected to further improve the efficacy and safety of aripiprazole.
Conclusion
Based on its sophisticated molecular structure and unique mechanism of action involving bidirectional dopamine-serotonin stabilization, Aripiprazole has overcome the efficacy and side effects bottlenecks of traditional psychotropic drugs, becoming a top choice in the field of psychiatric treatment. From the industrial synthesis of the active pharmaceutical ingredient to the development of multi-dosage formulations, from precision treatment of core indications to continuous breakthroughs in cutting-edge research, aripiprazole not only demonstrates the scientific appeal of combining pharmaceutical raw material development with neuropharmacology, but also provides a new approach to "balanced regulation" in the treatment of mental illnesses. With the continuous deepening of research on long-acting formulations, precision dosing, and new indications, aripiprazole will continue to expand its clinical application boundaries, providing safer and more effective treatment options for patients with mental illnesses worldwide while ensuring the quality of the active pharmaceutical ingredient and green manufacturing processes, and also providing important references for the development of next-generation neuropsychiatric drugs.
Xi'an Faithful BioTech Co., Ltd. cordially invites pharmaceutical companies and research institutions to learn about our comprehensive Aripiprazole powder solutions. Our technical team is ready to discuss your specific needs, provide detailed product specifications, and arrange sample testing. Please contact allen@faithfulbio.com for highly competitive pricing, technical documentation, and personalized consultation services to ensure your bulk purchasing needs are reliably met.
References
1.Casey, A. B. (2017). Classics in chemical neuroscience: Aripiprazole. ACS Chemical Neuroscience, 8(6), 1135–1146.
2.Davies, M. A. (2006). Aripiprazole: A novel atypical antipsychotic drug with a uniquely robust pharmacology. CNS Drug Reviews, 10(4), 317–336.
3.Food and Drug Administration. (2005). Abilify (aripiprazole) tablets, oral solution labeling. U.S. Department of Health and Human Services.
4.Kapur, S., & Seeman, P. (2001). Aripiprazole: A dopamine-serotonin system stabilizer. Journal of Clinical Psychiatry, 62(Suppl 13), 3–8.
5.Meltzer, H. Y., & Sumiyoshi, T. (2018). Aripiprazole for the treatment of schizophrenia: An update. Expert Review of Neurotherapeutics, 18(2), 111–124.
6.Shapiro, D. A., Renock, S., Arrington, E., Chiodo, L. A., Liu, L. X., & Sibley, D. R. (2003). Aripiprazole, a novel antipsychotic, is a high-affinity partial agonist at human dopamine D2 receptors. Journal of Pharmacology and Experimental Therapeutics, 306(2), 521–531.
7.Weiden, P. J., & Kane, J. M. (2019). Long-acting injectable aripiprazole: Clinical utility and real-world effectiveness. Journal of Clinical Psychiatry, 80(3), 19cm12870.






_1764052804092.webp)




