what is Dapoxetine HCL raw powder?
1. What is dapoxetine API?
Let's start with the basics-what is an API? For example, if you want to make a bowl of tomato and egg noodles, you need fresh tomatoes, eggs and noodles first, right? These "core ingredients" are "raw materials"; The process of frying tomatoes into sauce and eggs into pieces is "processing". Corresponding to the pharmaceutical industry, "raw materials" are the "core ingredients" of medicines and the real "active ingredients" in products; What is pressed into tablets and coated with sugar is called "preparation".
Simply put, it is a "stable form" of dapoxetine, but pure dapoxetine is unstable and easy to absorb moisture and deteriorate, so pharmaceutical factories will "combine" it with hydrochloric acid into "hydrochloride" and turn it into white crystalline powder, which is the "Dapoxetine HCL raw powder".
Further, the role of dapoxetine is to "control the brakes in the brain":
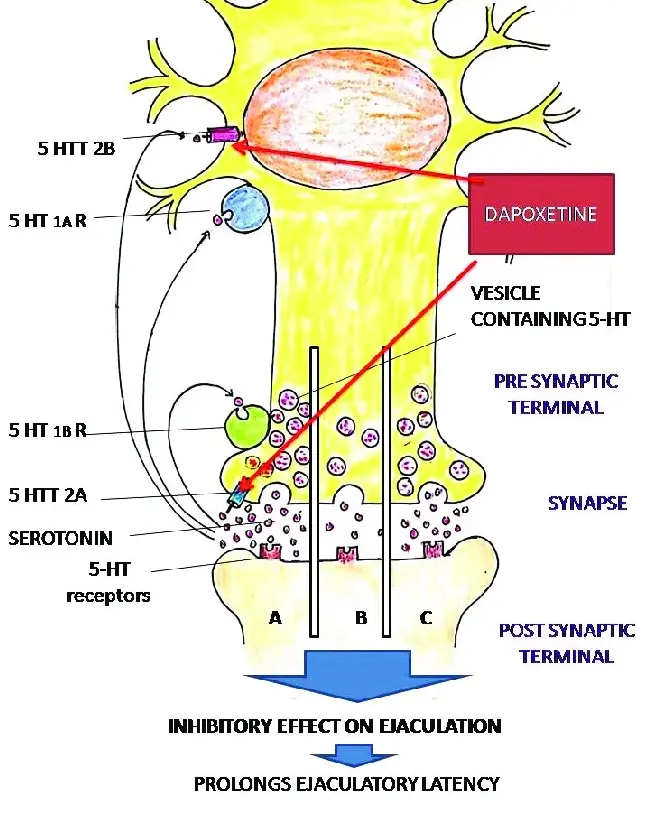
There is a "signalman" in our brain called 5- hydroxytryptamine, which is responsible for transmitting the signal that we are about to shoot. Under normal circumstances, the signalman's "work efficiency" is fixed, but some signalmen are too "anxious" to send an "emergency stop" command as soon as they feel a little, and the result is premature ejaculation. Its function is to temporarily "hold down" the signalman-make its concentration in the synaptic cleft higher, so that the brain can receive clearer "delay instructions", which is equivalent to stepping on the "brake" more stably.
This is why its function is not "slow" like antidepressants, but to accurately solve the problem of "falling off the chain at a critical moment". And the "source" of all this is this white powder of dapoxetine.
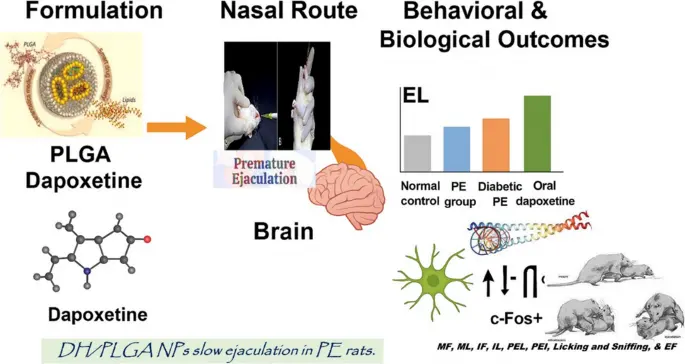
2.How difficult is the "transformation journey" of APIs?
You may think that API is "mixing the chemical raw materials together"? All wet. Its synthesis is a chemical magic "accurate to four decimal places", and every step has to be "perfectly stitched". Let's break it down into five steps, and you will understand why it can't be done by any factory:
Step 1: Find the right "road map"-synthesis process is the core competitiveness.
To turn a pile of basic chemical raw materials into it, we have to find the "shortest path" like walking a maze. For example, different compounds were used in the early process, and then gradually optimized into the "optimal route"; Some processes need 10 steps to react, while others only need 6 steps-the fewer steps, the lower the cost and the less impurities.
For example, it took the R&D team of a pharmaceutical factory three years to change the original eight-step synthesis method into five steps, which not only increased the yield from 60% to 85%, but also reduced the content of "harmful impurities" to 1/10 of the original. This step is the "technical barrier"-not a "shortcut" that everyone can think of.
The second step: purification-cut the "impurities" to "one in a million"
Even if it is synthesized, it will be mixed with all kinds of "impurities": raw materials that have not been completely reacted, by-products of the reaction, solvent residues ... even a little of these things may affect the efficacy and even safety. For example, if there are harmful substances in impurities, it may affect its function in the body, leading to unstable function.
How to purify it? The most commonly used method is recrystallization-the crude product is put into a solvent to be heated and dissolved, and then the temperature is slowly lowered to allow it to re-form crystals, while the impurities remain in the solution. But the key is to "choose the right solvent": it can not only dissolve well, but also "squeeze out" impurities.
What's even more powerful is chiral resolution-it has a "chiral center" (just like the left hand and the right hand, which look alike but can't overlap), and only the "dextrorotatory" is effective, while the levorotatory is useless and may even have side effects. In the past, the resolution depended on "luck", but now, with "asymmetric catalysis technology", only the D-isomer is generated directly at the time of synthesis, and it is in one step.
The third step: crystallization-make the powder "look exactly the same"
The same is the Dapoxetine HCL raw powder. Why do some manufacturers make powders that are "fluid" while others "stick together"? The secret lies in the crystal form.
The same chemical substance can exist in different "crystal shapes"-just like water can be ice (solid), water (liquid) and water vapor (gas). Its stable crystal form is "triclinic system", and the powder of this crystal form is "uniform in particle size" and "moderate in solubility". If the crystal form is wrong, for example, it becomes "amorphous", it may absorb moisture and agglomerate after a month, which will affect its efficacy.
Therefore, the raw material pharmaceutical factory has to spend a lot of time "growing crystals"-controlling the temperature, stirring speed and solvent ratio, so that each product "grows" into the same crystal.
The fourth step: detection-use "critical eye" to find faults.
Every batch of raw materials has to go through the "inspection", and nothing is missing. Test items can be filled with a piece of A4 paper:
Content: dapoxetine accounts for more than 99.5%, even less than 0.1%;
Related substances: known impurities should not exceed 0.1% and total impurities should not exceed 0.5%;
Residual solvent: it must meet the standard.
Moisture content: no more than 0.5%, otherwise it is easy to absorb moisture;
Microbes: There should be no more than 100 bacteria and no more than 10 molds per gram of powder.
Step 5: packaging-put "protective clothing" on the powder.
The last step is packaging-the raw materials must be packed in "aluminum foil bag+cardboard bucket" with desiccant in it. Why? Because it is particularly afraid of moisture: when the humidity in the air exceeds 60%, it will slowly absorb water and become a "sticky block" and cannot be used.
You see, from "a pile of raw materials" to "qualified raw materials", we have to go through process design → synthesis → purification → crystallization → testing → packaging, and every step has to be "detailed". This is why reliable Dapoxetine HCL raw powder suppliers can keep the "qualified rate" above 99.9%-this is not by luck, but by the technical accumulation of "grinding a sword for ten years".
3.Future: from "can do" to "do well",
Now, from "who can do it" to "who can do it better". In the future competition, we are fighting for these three points:
1. A greener process: from "reduced toxicity" to "zero toxicity"
The previous synthesis process will produce a lot of "three wastes" (waste water, waste gas and waste residue), and the treatment cost is high and it is not environmentally friendly. Now the industry is promoting "green chemistry"-for example, using "biocatalysis" instead of "chemical catalysis", using "water" instead of "organic solvent" as the reaction medium, and even turning "by-products" into useful raw materials.
2. More "accurate" crystal form: from "stable" to "better"
Now everyone is studying "eutectic technology"-combining it with another "safe molecule" to form a new crystal form. This eutectic has higher "solubility", which can make the efficacy play faster or have a longer "half-life".
3. More "complete" service
In the past, raw material pharmaceutical factories only "sold the powder", but now they have to provide more comprehensive services and quality.
Adjust the "particle size distribution" to make the powder easier to disperse; Provide more comprehensive technical materials and technical services.
Improve production efficiency and service quality
Conclusion
Some people say that the pharmaceutical industry is a "conscience industry"-because every product you make is related to people's life and health. Dapoxetine is no exception: it has no gorgeous packaging, no overwhelming advertisements, and even its name is rarely known, but it is quietly changing the lives of countless people.
Xi'an Faithful BioTech Co., Ltd. combines cutting-edge production technology with comprehensive quality assurance to provide high-quality Dapoxetine HCL raw powder that meets international pharmaceutical standards. Our commitment to excellent, competitive prices and technical support makes us the preferred partner of global healthcare providers and researchers. Please contact our technical team in sales11@faithfulbio.com to find out how our products can improve your formula.
This is a list of the names of the core scientific research documents that I referred to and relied on in the process of writing a soft article. These documents provide solid scientific evidence for the efficacy and mechanism mentioned in this paper.
- Cavallini, G., et al. (2004). "Dapoxetine, a novel treatment for premature ejaculation, does not interact with phosphodiesterase type 5 inhibitors." The Journal of Sexual Medicine, 1(2), 191-197.
- U.S. Patent No. 5,859,000 B1. (1999). "Use of dapoxetine for the treatment of premature ejaculation."
- B. S. K. K. Prasad, et al. (Eds.). (2016). "Novel Drug Delivery Systems for Selective Serotonin Reuptake Inhibitors (SSRIs)." In Advances in Pharmaceutical Technology and Drug Delivery.
- European Pharmacopoeia (Ph. Eur.) Monograph: Dapoxetine Hydrochloride. (Latest Edition, e.g., 11th Edition).
- United States Pharmacopeia (USP) Monograph: Dapoxetine Hydrochloride. (Latest Edition).
- Li, J., & Wang, Y. (2018). "Impurity profiling of dapoxetine hydrochloride by high-performance liquid chromatography coupled with quadrupole time-of-flight mass spectrometry (HPLC-QTOF-MS)." Journal of Pharmaceutical and Biomedical Analysis, 156, 123-130.
- Zhang, H., et al. (2020). "An improved and efficient synthesis of dapoxetine hydrochloride via a one-pot process." Organic Process Research & Development, 24(5), 789-795.
- M. Levin (Ed.). (2011). Pharmaceutical Process Scale-Up(3rd ed.). Informa Healthcare.
- Evaluate Pharma. (2022). "World Preview 2022, Outlook to 2028."











