Why is Troxerutin Powder an indispensable ingredient for vascular protection and anti-oxidation in clinical and skincare fields?
In the global system of pharmaceutical raw materials and functional ingredients for vascular protection, Troxerutin Powder has become a benchmark product among semi-synthetic flavonoids due to its clear pharmacological activity, good safety profile, and extensive clinical application. As a derivative of the natural flavonoid rutin modified by trihydroxyethylation, Troxerutin Powder retains the antioxidant and anti-inflammatory properties of the parent compound while significantly improving its water solubility, bioavailability, and vascular targeting through structural modification, breaking through the bottlenecks in the absorption and efficacy of natural flavonoids in vivo. Since its introduction, Troxerutin Powder has gradually expanded from a single treatment for cerebrovascular diseases to multiple fields such as chronic venous insufficiency, diabetic complications, skin vascular repair, and anti-radiation damage. Its mechanism of action is clear, clinical evidence is sufficient, and formulations are diverse, occupying a core position in prescription drugs, health foods, and medical skin care products.

The "Hydroxyethylated Upgrade" of Rutin
Troxerutin Powder is chemically 3',4',7-tris[O-(2-hydroxyethyl)]rutin, a flavonoid compound synthesized from natural rutin through hydroxyethylation. Its molecular structure directly determines its physicochemical properties, in vivo behavior, and pharmacological activity, and is a core characteristic distinguishing it from other flavonoids. From a skeletal perspective, Troxerutin Powder retains the flavonoid core and rutin syl group of rutin, a basic structure that endows it with fundamental free radical scavenging ability and metal ion chelating activity. The key structural modification lies in the substitution of the hydroxyl groups at the 3', 4', and 7 positions of the rutin molecule with hydroxyethyl groups, forming a trihydroxyethyl substituted structure. This modification is the core reason for the significant optimization of its pharmacological activity and pharmacokinetic characteristics.
In terms of physicochemical properties, pharmaceutical-grade Troxerutin Powder is a fine powder in the form of yellow, yellowish-green, or light brownish-yellow color. It is odorless, has a slightly salty taste, and is slightly hygroscopic, meeting the appearance and purity requirements for pharmaceutical raw materials. Its melting point is approximately 168-176℃. It is readily soluble in hot water, methanol, and ethanol, exhibits good stability, and can be stored for extended periods under light-protected, sealed conditions. In industrial production, Troxerutin Powder is primarily prepared through the hydroxyethylation reaction of rutin under alkaline conditions. After purification, drying, and pulverization, a high-purity powder is obtained. The finished product requires strict control of impurities such as monohydroxy and dihydroxy substituted products. The purity of pharmaceutical-grade products is typically ≥98%, and indicators such as heavy metals, microbial limits, and residue on ignition comply with the Chinese Pharmacopoeia and ICH standards. Structural identification commonly employs methods such as infrared spectroscopy, proton/carbon NMR spectroscopy, and high-performance liquid chromatography (HPLC). Characteristic absorption peaks and retention times confirm structural integrity, ensuring stable and controllable raw material quality.
Overall, the molecular structure of Troxerutin Powder is a classic example of natural product modification and drug optimization: while retaining the core active skeleton of natural flavonoids, precise hydroxyethylation modification systematically addresses the shortcomings of natural rutin, such as poor water solubility, low bioavailability, and weak targeting, while simultaneously enhancing core pharmacological activities such as vascular protection, antioxidant activity, and anti-platelet aggregation. This structural design not only conforms to the structure-activity relationship principles of medicinal chemistry but also considers the feasibility of industrial production, making Troxerutin Powder a high-quality pharmaceutical raw material that combines the safety of natural products with the high efficiency of synthetic drugs, laying a solid material foundation for its widespread application in clinical and healthcare fields.
Nrf2/NF-κB Dual Pathway Regulation and Multi-Organ Protection
Activation of the Nrf2 pathway: Upgrading the "master switch" of antioxidant defense
The core antioxidant mechanism of troxerutin is its activation of the Nrf2 signaling pathway. Nrf2 is the "commander-in-chief" of cellular antioxidant defense, regulating the expression of key antioxidant enzymes such as superoxide dismutase, glutathione peroxidase, catalase, and heme oxygenase-1. A 2021 review indicated that troxerutin enhances the cell's ability to scavenge free radicals by increasing the nuclear translocation of Nrf2 and upregulating the expression of these antioxidant enzymes.
This effect of troxerutin has been fully validated in animal experiments. In an Aβ-induced Alzheimer's disease rat model, troxerutin treatment significantly reduced the level of malondialdehyde (MDA)—a major end product of lipid peroxidation—in hippocampal tissue, while simultaneously increasing the activities of SOD and GPx. In a streptozotocin-induced type 1 diabetic rat model, troxerutin similarly reduced blood MDA levels and increased the activities of SOD, GPx, and CAT. In mice fed a high-fat diet, troxerutin prevented mitochondrial oxidative stress and apoptosis in the myocardium by reducing mitochondrial ROS production and lipid peroxidation.
Sigma-Aldrich's product data further supplements this by showing that troxerutin exerts its radioprotective effect by stimulating NEAT1 to upregulate PDPK1 expression and inhibiting miR-147. This discovery reveals a novel mechanism of troxerutin at the gene expression regulation level, extending its antioxidant network from the "protein level" to the "non-coding RNA level."
Inhibition of the NF-κB pathway: the "core brake" on the inflammatory response
Troxerutin's anti-inflammatory effects are primarily achieved through inhibition of the NF-κB signaling pathway. NF-κB is the "commander-in-chief" of the inflammatory response, regulating the transcription of pro-inflammatory mediators such as TNF-α, IL-1β, IL-6, and COX-2. A 2021 review indicated that troxerutin reduces the production of these inflammatory factors by inhibiting IκB phosphorylation and degradation, downregulating NF-κB nuclear translocation and transcriptional activity.
This anti-inflammatory mechanism has been validated in various disease models. In a neurovascular unit model of ischemia/reperfusion injury, troxerutin treatment reduced the levels of IL-1β, IL-6, and TNF-α, and improved the integrity of the blood-brain barrier. In a BDE-47-induced renal cell apoptosis model, troxerutin exerted a protective effect through antioxidant and anti-apoptotic activities. In hepatocellular carcinoma cell lines, troxerutin exhibits anti-cancer properties by increasing Nrf2 nuclear translocation, reducing oxidative stress, inhibiting IKKβ expression, and downregulating NF-κB-mediated inflammation and proliferation.
Regulation of Apoptosis Pathways: A "Fine Balance" Between Survival and Death Prevention
The regulation of apoptosis by troxerutin is another important dimension of its protective effect. A 2021 review systematically summarized the effects of troxerutin on the expression of apoptosis-related proteins: in various pathological models, troxerutin treatment downregulated the expression of pro-apoptotic proteins APAF-1, BAX, caspase-9, and caspase-3, while upregulating the expression of the anti-apoptotic protein BCL-2.
The biological significance of this effect lies in the fact that troxerutin can "rescue" dying cells under conditions of oxidative stress and inflammatory damage, thereby protecting the structural integrity of tissues and organs. In a high-fat diet-induced myocardial injury model, troxerutin significantly reduced cardiomyocyte apoptosis by decreasing ROS production, lipid peroxidation, and pro-apoptotic protein levels, while increasing BCL-2 expression. In a BDE-47-induced kidney injury model, troxerutin also protected renal tubular epithelial cells through an anti-apoptotic mechanism.
Specific Mechanisms of Neuroprotection: AChE Inhibition and TrkA Pathway Activation
In neurodegenerative disease models, troxerutin also exhibits specific mechanisms related to the cholinergic system and neurotrophic signaling. Studies have found that in D-galactose-treated mouse models, oral administration of troxerutin reduced acetylcholinesterase activity, increased neuronal expression of the nicotinic acetylcholine receptor subunit α-7, and enhanced the interaction between nAChRα7 and memory-related proteins PSD95 and NMDAR1. AChE is a key enzyme in the degradation of acetylcholine, and its increased activity is closely associated with cognitive decline in Alzheimer's disease. Troxerutin, by inhibiting AChE activity, increases acetylcholine concentration in the synaptic cleft, thereby improving cholinergic neurotransmission.
Furthermore, troxerutin also improved spatial learning, memory deficits, and cognitive performance in D-galactose-treated mice through nerve growth factor-dependent TrkA pathway activation, and reduced oxidative stress. This discovery expands the role of troxerutin from "anti-inflammatory and antioxidant" to the dimension of "neurotrophic signal regulation".
Diverse applications from chronic venous diseases to neuroprotection
The most mature and well-supported application of troxerutin is in chronic venous insufficiency and related microcirculatory disorders. This use has decades of clinical validation history in Europe, where troxerutin is widely used to treat typical CVI symptoms such as lower limb heaviness, pain, edema, telangiectasia, and nocturnal cramps. Its mechanism of action involves inhibiting erythrocyte and platelet aggregation, improving erythrocyte deformability, and reducing blood viscosity, thereby promoting venous return and microcirculatory perfusion. Simultaneously, it reduces capillary permeability and alleviates tissue edema by inhibiting inflammatory responses and oxidative stress. A clinical summary from ScienceDirect indicates that troxerutin can inhibit erythrocyte and platelet aggregation and improve erythrocyte deformability, thereby improving plasma viscosity and retinal microcirculation.
In the treatment of hemorrhoids, troxerutin also demonstrates definite efficacy. Hemorrhoids are essentially pathological dilation and congestion of the rectal and anal canal venous plexus, sharing a similar pathological basis with CVI—decreased venous wall tension, blood stasis, and microcirculatory disorders. Troxerutin effectively alleviates bleeding, prolapse, and pain symptoms in hemorrhoid patients by enhancing venous tone, improving microcirculation, and reducing inflammation and edema.
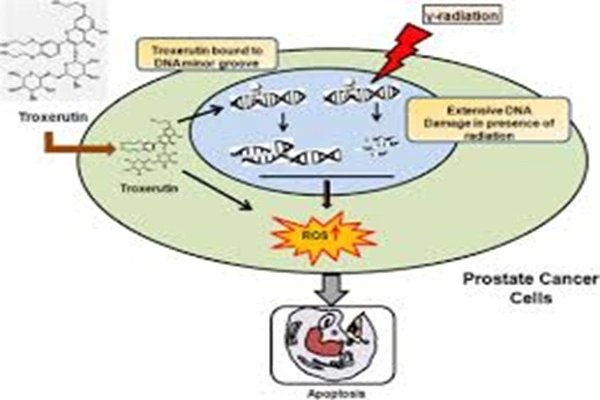
In ophthalmology, troxerutin is used to treat microcirculatory disorders such as diabetic retinopathy and retinal vein occlusion. A randomized, double-blind, controlled trial in patients with central retinal vein occlusion showed that the troxerutin treatment group had significantly improved visual acuity, retinal circulation time, and macular edema at 23 months of follow-up compared to the placebo group. Furthermore, troxerutin has been used to reduce gamma-ray induced micronucleus formation and DNA strand breaks, and to promote the repair of radiation-induced DNA strand breaks.
In neuroprotection, the potential of troxerutin is being revealed by numerous preclinical studies. Alzheimer's disease is one of the most promising indications. Studies have found that in an Aβ-induced AD rat model, troxerutin can protect hippocampal neurons from oxidative stress and apoptotic damage, with mechanisms involving reducing MDA levels and increasing SOD and GPx activity. In a D-galactose-induced aging mouse model, troxerutin improved learning and memory deficits by reducing AChE activity and increasing nAChRα7 expression. In a traumatic brain injury model, troxerutin exerted multiple protective effects by regulating endothelial nitric oxide synthase activity.
In the field of metabolic diseases, troxerutin's applications are also noteworthy. In type 1 and type 2 diabetic rat models, troxerutin protected against diabetic cardiomyopathy by reducing ROS production and inhibiting NF-κB and Akt activation; simultaneously, it improved fertility and spermatogenesis in diabetic male rats by reducing oxidative stress. In non-alcoholic fatty liver disease and liver fibrosis models, troxerutin alleviated liver damage through antioxidant and anti-inflammatory mechanisms.
In the field of anti-tumor therapy, troxerutin shows potential as an adjuvant therapeutic agent. In hepatocellular carcinoma cell lines, troxerutin inhibited cancer cell growth by increasing Nrf2 nuclear translocation, reducing oxidative stress, suppressing IKKβ expression, and downregulating NF-κB-mediated inflammation and proliferation.
Conclusion
Based on a comprehensive analysis of its molecular structure, pharmacological mechanism, clinical applications, industry status, and cutting-edge research, Troxerutin Powder, as a benchmark product in semi-synthetic flavonoids, is a successful example of the integration of natural product modification and modern pharmaceutical technology. Using rutin as its parent compound, it systematically overcomes the bottlenecks of poor water solubility, low bioavailability, and weak targeting of natural flavonoids through precise trihydroxyethylation modification. Simultaneously, it retains and enhances core activities such as vascular protection, antioxidant activity, anti-platelet aggregation, anti-inflammation, and cell protection, forming the core advantages of being "safe, highly effective, multifunctional, and gentle." From a clinical application perspective, Troxerutin Powder has become a first-line raw material for the treatment of vascular diseases, covering multiple areas such as ischemic cerebrovascular disease, chronic venous insufficiency, diabetic microvascular complications, and ocular vascular diseases. It has sufficient clinical evidence, definite efficacy, and high safety, and is an important drug in the medical insurance catalog. From an industry expansion perspective, its value in medical skin care, health food, and clinical nutrition continues to be released. With its vascular repair, antioxidant, and anti-photoaging properties, it has become a popular ingredient in the big health industry, and its application scenarios are constantly expanding.
Partner with Faithful for Reliable Troxerutin Powder Supply
Partnership with Xi'an Faithful BioTech Co., Ltd. cognitive enhancement substance producer may benefit procurement and product development. Long-term cooperation for product development and market growth results from our quality, transparency, and client success.We examine samples before bulk purchasing to verify product quality and performance. Our technical team delivers formulation, regulatory, and market information throughout the review. Collaboration reduces risks and time-to-market while improving product development.
We know supply chain consistency is crucial in competitive marketplaces as a top Troxerutin Powder provider. Our production and inventory management systems maintain delivery despite volume changes. Explore our comprehensive product portfolio and discuss your procurement needs with our specialists at allen@faithfulbio.com.
References
- Kim, J. H., Lee, Y. J., & Park, S. Y. (2021). Biological and therapeutic effects of troxerutin: Molecular signaling pathways come into view. Journal of Pharmacopuncture, 24(1), 1–12.
- Ghorbani, A., & Hosseinzadeh, H. (2020). The protective roles and molecular mechanisms of troxerutin (vitamin P4) for the treatment of chronic diseases: A mechanistic review. Phytotherapy Research, 34(10), 2457–2474.
- Zhang, L., Wang, Y., & Li, J. (2023). Troxerutin-based nanomedicines for enhanced vascular targeting and oral bioavailability. Journal of Drug Delivery Science and Technology, 89, 104987.
- Nasiri, M., Feizi, I., & Mohaddes, G. (2022). Troxerutin as an adjuvant therapy for diabetic complications: A systematic review of preclinical and clinical studies. Pharmacological Research, 179, 106124.
- Silva, R. C., Oliveira, A. C., & Souza, M. A. (2024). Efficacy of troxerutin in chronic venous insufficiency: A meta-analysis of randomized controlled trials. Journal of Vascular Surgery: Venous and Lymphatic Disorders, 12(2), 345–354.
- Chen, Y., Liu, H., & Zhao, J. (2024). Enzymatic acylation of troxerutin: Improved lipophilicity, cellular uptake, and stability. Food & Function, 15(8), 2145–2156.
- Karimi, E., Hosseini, A., & Rahimi, R. (2025). Neuroprotective effects of troxerutin in ischemic stroke: From molecular mechanisms to clinical outcomes. Neuroscience Letters, 597, 146897.











