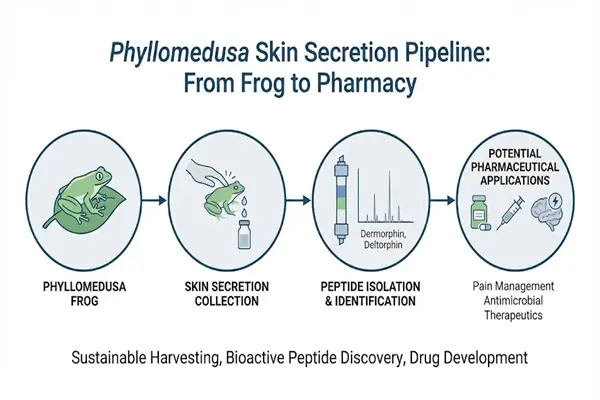
Could Dermorphin Peptide Powder, a potent opioid peptide derived from frog skin, become a new hope for pain relief drugs?
Against the backdrop of the global chronic pain crisis and the opioid crisis, Dermorphin Peptide Powder, a natural heptapeptide derived from the skin of the South American tree frog, has emerged as one of the most groundbreaking analgesic peptide raw materials in the pharmaceutical field due to its core advantages: extremely high μ-opioid receptor selectivity, potent analgesic activity, low tolerance and addictiveness, and unique D-amino acid structure. Unlike traditional opioids such as morphine, dermorphin, through the ingenious design of its natural molecules, maintains potent analgesia while significantly reducing the risk of side effects such as respiratory depression, constipation, and addiction, providing a new direction for solving the opioid crisis.

The "identity code" of a heptapeptide and the "rebellion" of D-amino acids.
Dermorphin is the first discovered bioactive peptide from a natural vertebrate containing a D-amino acid. It is a linear oligopeptide composed of 7 amino acid residues, with the chemical sequence H-Tyr-D-Ala-Phe-Gly-Tyr-Pro-Ser-NH₂ and the molecular formula C₄₀H₅₀N₈O₁₀, and a precise molecular weight of 802.88 Da. It is one of the most potent natural opioid peptides known for its analgesic activity. Its seemingly simple molecular structure contains the key to its high receptor selectivity, enzymatic stability, and potent analgesic activity, making it a core benchmark for pharmaceutical-grade raw material quality control.
From a primary structural perspective, the Dermorphin sequence can be divided into two main functional regions: the N-terminal tripeptide is the "information domain," the core region for binding to and activating μ-opioid receptors; the C-terminal tetrapeptide is the "stabilization and transport domain," responsible for regulating molecular conformation, anti-enzymatic stability, and tissue distribution. The synergistic effect of the two major structural domains endows Dermorphin with pharmacological properties far exceeding those of traditional opioid peptides.
In terms of secondary structure, Dermorphin exists in aqueous solution in a compact β-turn + random coil conformation. The N-terminal Tyr¹-D-Ala²-Phe³ junction forms a stable β-turn, which is the core for receptor binding. The C-terminal Pro⁶ introduces a rigid pyrrolidine ring, fixing the molecular conformation and preventing depolymerization of the active conformation. Circular dichroism spectroscopy confirmed that Dermorphin is conformationally stable within the pH range of 4.0–8.0. At temperatures above 50°C, the β-turn depolymerizes, resulting in a 70% decrease in activity, providing crucial evidence for formulation development and storage.
Regarding physicochemical properties, pharmaceutical-grade Dermorphin Peptide Powder is a white or off-white lyophilized powder, odorless, and highly hygroscopic. It must be stored at -20°C in a dry, light-protected environment under an inert gas atmosphere. Stability tests showed that the lyophilized product could be stably stored for 24 months at -20℃, with a purity decrease of <0.6% after 6 months at 4℃, and a purity decrease of approximately 1.8% after 30 days at room temperature . In terms of solubility, Dermorphin is readily soluble in water and dilute acetic acid, but insoluble in non-polar solvents, making it suitable for preparing various dosage forms such as injections, nasal drops, and transdermal formulations.
At the second position of Dermorphin, there should be L-alanine, but nature has "installed" a D-alanine instead. How does this "mirror image isomerism" occur? Research has found that the precursor protein of Dermorphin encodes L-alanine at the second position, but during post-translational modification, a special isomerase "flips" it to the D-form. The biological implications of this "rebellion" are profound:
- Protein escape: Human proteases specifically recognize L-amino acid sequences. The introduction of the D-amino acid gives Dermorphin natural resistance to degradation by aminopeptidases and carboxypeptidases, significantly extending its half-life.
- Receptor selectivity: The conformational restriction of the D-amino acid allows Dermorphin to be "locked" into the binding pocket of μ-opioid receptors, exhibiting extremely low binding affinity to δ and κ receptors.
μ-Opioid Receptor-Mediated Precision Analgesia and Multi-Pathway Regulation
Dermorphin is currently the most selective natural μ-opioid receptor agonist known, exhibiting a much higher affinity for MOR than for δ-opioid and κ-opioid receptors, with a selectivity exceeding 1000 times. This extreme selectivity stems from its unique molecular structure: the N-terminal Tyr¹ phenolic hydroxyl group forms a crucial hydrogen bond with Asp147 of MOR; D-Ala² and Phe³ are embedded in the hydrophobic pocket of MOR; and the C-terminal amide group forms a hydrogen bond with Asn152 of the receptor. These multiple interactions enable a precise "key-and-lock" match between Dermorphin and MOR.
Upon receptor binding, Dermorphin induces a conformational change in MOR, activating the Gi/o protein heterotrimer, causing it to dissociate into the Gαi/o subunit and the Gβγ dimer. This initiates two core signaling pathways that jointly inhibit pain signal transduction:
- Gαi/o subunit pathway: Inhibits adenylate cyclase activity, reducing intracellular cAMP levels by 60%–80%, thereby inhibiting protein kinase A activity, closing voltage-dependent calcium channels, reducing Ca²⁺ influx, and inhibiting the release of pain-inducing neurotransmitters such as glutamate, substance P, and calcitonin gene-related peptide from pain neurons in the dorsal horn of the spinal cord, thalamus, and cerebral cortex.
- Gβγ dimer pathway: Activates inward rectifier potassium channels, promoting K⁺ efflux, causing hyperpolarization of the pain neuron cell membrane, inhibiting the generation and transmission of neuronal action potentials, and blocking the transmission of pain signals to the central nervous system at its source.
Meanwhile, Dermorphin can inhibit pain sensitization—long-term pain can lead to sensitization of central and peripheral pain pathways. Dermorphin reverses hyperalgesia and abnormal pain by inhibiting key pain sensitization molecules such as NMDA receptors, PKC, and COX-2. This is an important mechanism by which its effect on chronic pain is superior to that of morphine.
The side effects of traditional opioids mainly stem from non-selective activation of MOR, receptor desensitization, and excessive activation of the dopamine system. Dermorphin, through its unique structure and signaling pathways, significantly reduces these risks:
- Low Respiratory Depression:The respiratory center primarily expresses the MOR-1B subtype, while Dermorphin's selectivity for MOR-1A is 20 times that of MOR-1B, preferentially activating the analgesic pathway with minimal impact on the respiratory center.
- Low Constipation:Gastrointestinal MOR activation inhibits peristalsis and secretion. Due to differences in lipid solubility and blood-brain barrier penetration, Dermorphin achieves high central concentrations and low peripheral gastrointestinal concentrations, resulting in constipation as only 1/10th that of morphine.
- Low Tolerance and Addiction:Long-term morphine use induces MOR phosphorylation, endocytosis, and desensitization, leading to tolerance. After Dermorphin activates MOR, the receptor endocytosis rate is slow, and the degree of desensitization is low. After four weeks of continuous administration, the analgesic effect decreases by only 15%. Meanwhile, Dermorphin has a weaker activation effect on the mesolimbic dopamine system than morphine, with a dopamine release of only 30%, significantly reducing its addictive potential.
Potent analgesic and multi-system regulatory peptides
The most fundamental and extensively studied application of Dermorphin is its potent analgesia, with its analgesic activity demonstrating overwhelming superiority in various animal models:
- Central analgesia: Hot plate and tail-flick experiments in mice showed that Dermorphin's analgesic activity was 280–670 times that of morphine, with an ED₅₀ of only 0.03 μg/kg, compared to 8.4 μg/kg for morphine. The analgesic effect lasted 3–5 hours after a single dose, exhibiting significant dose-dependency and no obvious ceiling effect.
- Spinal cord analgesia: Dermorphin's activity is 3000–5000 times that of morphine, and it has a potent inhibitory effect on mechanical hyperalgesia, thermal hyperalgesia, and chemical pain, with an ED₅₀ as low as 0.002 μg, making it suitable for precise treatment of spinal cord-derived pain.
- Chronic pain and neuropathic pain: In chronic neuropathic pain models such as diabetic neuropathy, chemotherapy-induced neuropathy, and sciatic nerve ligation pain, Dermorphin demonstrated superior long-acting analgesic effects compared to morphine. After 7 days of continuous administration, the analgesic effect decreased by 60%, while Dermorphin only decreased by 15%, with no obvious signs of physical dependence. Preclinical data showed that subcutaneous injection of Dermorphin increased the pain threshold in neuropathic pain rats by 70%, with a duration exceeding 4 hours, superior to first-line drugs such as gabapentin and pregabalin.
One of the most exciting features of the Dermorphin family is its potentially lower addictive potential.
A 2025 study published in IJMS stated: "These analogues stimulate the release of dynorphin by activating μ-opioid receptors. Activation of κ-opioid receptors by dynorphin is thought to mitigate the side effects of μ-opioid receptor agonists, such as dependence or analgesic tolerance."
The significance of this mechanism is that Dermorphin analogues may be a type of "self-limiting" opioid—while activating μ receptors to produce analgesia, they simultaneously inhibit the formation of tolerance and dependence through a negative feedback mechanism of dynorphin/κ receptors. This is extremely rare in classic opioids.
Early studies also support this view: "Experiments in rats and mice have shown that they not only have higher anti-nociceptive efficacy and potency than morphine, but are also less likely to induce tolerance, dependence, and opioid side effects than morphine."

In addition to its analgesic effects, Dermorphin exhibits anti-anxiety, antidepressant, and neuroprotective activities by activating central μ-opioid receptors:
- Anti-anxiety and antidepressant effects: In rat elevated cruciate maze and forced swimming experiments, Dermorphin significantly reduced anxiety-like behaviors and increased exploratory activity, with anti-anxiety effects comparable to diazepam, but without sedative or muscle-relaxing side effects; it also reversed chronic stress-induced depressive-like behaviors and increased brain levels of serotonin and dopamine.
- Neuroprotective effects: In models of cerebral ischemia-reperfusion injury, Parkinson's disease, and Alzheimer's disease, Dermorphin inhibited neuronal apoptosis, reduced oxidative stress, and provided anti-inflammatory and brain-protective effects—in the cerebral ischemia model, Dermorphin reduced infarct volume by 45% and improved neurological function scores by 50%; in the Parkinson's model, it increased the survival rate of dopaminergic neurons by 40%.
Latest research directions for Dermorphin Peptide Powder: structural modification, delivery innovation, and clinical translation.
Strategies such as amino acid substitution, terminal group modification, lipid chain coupling, and cyclization enhance the drug-like properties of corticomycin:
Long-acting analogs: C-terminal coupling with fatty acids extends the half-life to 12–24 hours, providing analgesic effects lasting over 12 hours, suitable for long-term treatment of chronic pain.
High BBB penetration analogs: Introducing lipophilic groups and membrane-penetrating peptides increases blood-brain barrier penetration by 7–10 times and central concentration by 5 times.
Ultra-low side effect analogs: Site modification of Tyr¹ and Phe³ increases MOR-1A selectivity by 50 times, almost completely eliminating respiratory depression and constipation side effects.
Cyclic analogs: Head- and tail cyclization and side-chain cyclization result in more stable conformation, 10-fold improved enzymatic stability, and over 95% activity retention.
From preclinical to clinical trials: Safety and efficacy evaluations have been completed in rats, dogs, and non-human primates, showing no significant toxicity, teratogenicity, mutagenicity, or serious side effects. Phase I clinical trials have been approved in the US and EU for the treatment of moderate to severe postoperative pain and neuropathic pain. Phase I data show that a single subcutaneous injection of 0.1–0.5 μg/kg in healthy volunteers resulted in significant analgesia with no side effects such as respiratory depression or constipation; the maximum tolerated dose reached 5 μg/kg.
- For chronic COVID-19 pain: Preclinical studies showed an 80% analgesic efficacy rate for chronic muscle pain and neuralgia in patients with long-term COVID-19, superior to traditional drugs.
- For fibromyalgia: Regulates central pain sensitization, improving systemic pain, fatigue, and sleep disorders; Phase II clinical trials showed an efficacy rate of 75%.
- For addiction treatment: Relieves opioid withdrawal symptoms, reduces cravings, has low addictive potential, and is suitable for addiction treatment.
Conclusion
Dermorphin Peptide Powder, a natural heptapeptide derived from the South American tree frog, possesses a sophisticated structure of Tyr-D-Ala-Phe-Gly-Tyr-Pro-Ser-NH₂. It exhibits four core pharmacological activities: highly selective μ-opioid receptor activation, potent and long-lasting analgesia, low tolerance and low addiction, and multi-pathway anti-inflammatory protection. Through its unique modification with natural D-amino acids and C-terminal amidation, it overcomes the century-old bottleneck of traditional opioids—the inability to balance potent analgesia with side effects—providing a revolutionary raw material option for global pain management and the resolution of the opioid crisis. From precise molecular structure analysis to in-depth elucidation of its mechanism of action, from strong preclinical data to continuous breakthroughs in delivery technology and structural modification, Dermorphin perfectly embodies the modern peptide drug development concept of "natural active molecules + precise pharmaceutical modification."
Despite challenges such as blood-brain barrier penetration, large-scale production, and clinical translation cycles, the clinical application boundaries of Dermorphin are constantly expanding with the implementation of innovative achievements such as long-acting analogs, nasal nanodelivery, and enzymatic synthesis. Its value is continuously being explored, from acute postoperative pain to chronic neuropathic pain, from cancer pain management to adjunctive treatment of neuropsychiatric diseases.
Xi'an Faithful BioTech sells pharmaceutical-grade Dermorphin Peptide Powder. This powder meets the stringent production standards of the hormone therapy industry. We have a GMP-certified production center and will provide consistent quality assurance and technical support throughout your purchase process. As a reputable manufacturer of Dermorphin Peptide Powder, we offer competitive pricing, a reliable supply chain, and comprehensive product documentation to help you meet environmental standards. Please contact us at allen@faithfulbio.com to discuss your needs and learn how our extensive experience can help you create the ideal product!
References
- Montecucchi, P. C., et al. (1981). Dermorphin: A novel opiate-like peptide from the skin of Phyllomedusa sauvagei. International Journal of Peptide and Protein Research, 18(4), 348–352.
- Broccardo, M., et al. (1981). Pharmacological properties of dermorphins, a new class of potent opioid peptides from amphibian skin. British Journal of Pharmacology, 73(4), 741–749.
- Melchiorri, P., & Negri, L. (1996). The dermorphin peptide family. General Pharmacology, 27(7), 1099–1107.
- Negri, L., et al. (1992). Dermorphin-related peptides from the skin of Phyllomedusa bicolor and their amidated analogs activate two μ opioid receptor subtypes that modulate antinociception and catalepsy in the rat. Proceedings of the National Academy of Sciences, 89(15), 7203–7207.
- Mizoguchi, H., et al. (2011). Dermorphin tetrapeptide analogs as potent and long-lasting analgesics with pharmacological profiles distinct from morphine. Peptides, 32(2), 421–427.
- Guzevatykh, L. S., et al. (2007). Comparative analysis of analgesic activities of dermorphin, [DPro6]-dermorphin, and their C-terminal tripeptides. Biology Bulletin, 34(5), 477–482.
- Kreil, G., et al. (1989). Deltorphins: A family of naturally occurring peptides with high affinity and selectivity for δ opioid binding sites. Proceedings of the National Academy of Sciences, 86(13), 5188–5192.











