Could thymulin, a "tuner of the immune system" secreted by the thymus gland, become an all-powerful key to anti-inflammation and regeneration?
In today's world, with the high incidence of immune-related diseases and the increasingly urgent need for infection and cancer prevention and treatment, Thymulin peptide powder has become a benchmark peptide raw material in the field of immunomodulatory pharmaceuticals due to its four core advantages: natural thymus source, zinc ion-dependent activation, precise bidirectional immune regulation, and extremely low toxicity and side effects. Unlike ordinary immune enhancers, it can both improve weakened immune function and inhibit overactive immune responses, demonstrating unique value in scenarios such as immunodeficiency, viral infection, adjuvant cancer treatment, autoimmune diseases, and aging-related immune decline.
The "metal-dependent" code of a nonapeptide
Thymulin is a nonmitogenic nonapeptide synthesized and secreted by thymic epithelial cells. Its amino acid sequence is H-Pyr-Ala-Lys-Ser-Gln-Gly-Gly-Ser-Asn-OH, with the molecular formula C₃₃H₅₆N₁₀O₁₅ and a precise molecular weight of 852.83 Da. It is one of the most structurally well-defined and mechanistically clear thymic-derived immunomodulatory peptides. Its most distinctive characteristic is that its biological activity is entirely dependent on zinc ions; it can only exert its immunomodulatory effect by forming a stable complex with Zn²⁺. This structural characteristic determines its entire pharmacological behavior and key quality control points.
From a primary structural perspective, Thymulin consists of a linear arrangement of nine amino acid residues: pyroglutamic acid, alanine, lysine, serine, glutamine, glycine, and asparagine. The N-terminal pyroglutamic acid is a natural cyclization modification that effectively resists degradation by aminopeptidases and prolongs the in vivo half-life. The molecule is rich in polar amino acids such as serine and lysine, providing multiple coordination binding sites for zinc ions. The C-terminus is asparagine, maintaining molecular polarity and water solubility. The entire sequence is highly conserved, exhibiting extremely high cross-species homology, from rodents to primates, ensuring the reliability of its clinical translation.
Zinc ion binding is central to the structure and function of Thymulin. The lysine side-chain amino group, the serine hydroxyl group, and the peptide carbonyl group together constitute the zinc ion chelating site, forming a stable 1:1 coordination complex. Experiments have shown that apo-Thymulin without zinc binding has almost no biological activity, while holo-Thymulin bound with Zn²⁺ exhibits more than 100-fold increased activity. Circular dichroism spectroscopy shows that after zinc binding, Thymulin transforms from a disordered coiled conformation to a stable folded conformation, enabling it to be recognized by receptors on the surface of immune cells and initiate signaling. This means that pharmaceutical-grade Thymulin must be produced and stored in the form of a zinc complex; otherwise, the raw material will lose its medicinal value.
Pharmaceutical-grade Thymulin Peptide Powder is a white or off-white lyophilized powder, odorless, highly hygroscopic, readily soluble in water, physiological saline, and buffer solutions, and practically insoluble in organic solvents. With a LogP of approximately -4.5, it is a strongly hydrophilic peptide, rapidly distributed in vivo but with weak membrane permeability. In terms of stability, Thymulin is sensitive to temperature, pH, and oxidation: it is conformationally stable within a pH range of 5.0–7.5, but peptide bond hydrolysis and zinc dissociation easily occur at temperatures above 40°C or pH < 4.0; it is also easily oxidized and inactivated by air and light. Therefore, pharmaceutical-grade raw materials must be stored at -20°C under light-protected, vacuum-dried, and nitrogen-filled conditions. Stability data show that lyophilized zinc-complex Thymulin can be stably stored for 24 months at -20°C, with a purity decrease of <0.7% after 6 months at 4°C, and a purity decrease of approximately 1.9% after 30 days at room temperature.
HPLC purity ≥98.5%, total impurities <1.0%, single impurities <0.1%; zinc content controlled between 7.2% and 8.2%, ensuring a 1:1 binding ratio; moisture ≤4.0%, acetic acid/trifluoroacetic acid residues comply with ICH standards; heavy metals <10 ppm, endotoxins <0.5 EU/mg; optical rotation [α]²⁰_D -30°~ -40°. Structure-activity relationship studies showed that the absence of N-terminal pyroglutamic acid, mutation of the zinc binding site, and C-terminal truncation all lead to a decrease in activity of more than 90%.
Zinc-dependent immune cell precision regulation network
Thymulin's mechanism of action is highly clear, its target is well-defined, and its pathways are conserved. It revolves around five core pathways: zinc-dependent receptor binding, T cell differentiation and maturation, immune cell subset balance, cytokine regulation, and antioxidant and anti-apoptotic effects, achieving gentle yet precise immune remodeling. Its action is initiated by the binding of the Zn²⁺-Thymulin complex to specific receptors on the surface of immune cells. These receptors are mainly expressed on the surface of immature T cells, mature T cells, NK cells, and monocytes/macrophages, and are high-affinity G protein-coupled receptors. Upon binding, they activate the cAMP/PKA and MAPK/ERK pathways, regulating the expression of downstream immune-related genes. Experiments show that Thymulin's affinity for its receptor is at the nanomolar level, and significant biological effects can be produced at extremely low concentrations.
The most crucial function of thymulin is to promote T cell differentiation and maturation. It acts at the junction of the thymic cortex and medulla, promoting the maturation of CD4⁻CD8⁻ double-negative cells into CD4⁺CD8⁺ double-positive cells, and further selecting and differentiating them into functional CD4⁺ helper T cells or CD8⁺ cytotoxic T cells. This process is hindered in thymic atrophy or hypofunction, while thymic supplementation can increase the output of mature T cells by 3–5 times.
Simultaneously, thymulin precisely regulates the balance of immune subsets: promoting Th1 responses to enhance antiviral and antitumor immunity; inhibiting excessive Th17 polarization to reduce autoimmune damage; and moderately enhancing Treg function to maintain immune homeostasis. It can also significantly enhance NK cell cytotoxic activity and dendritic cell antigen presentation capacity, comprehensively strengthening the body's immune surveillance.
At the cytokine level, Thymulin exhibits typical bidirectional regulation: it promotes the release of IL-2, IFN-γ, and IL-12 during immunodeficiency and inhibits pro-inflammatory factors such as IL-6, IL-1β, and TNF-α during immunohyperactivity. In vitro experiments showed that in LPS-activated macrophages, Thymulin reduced IL-6 secretion by 62% and NF-κB nuclear translocation by 57%.
Furthermore, Thymulin exerts antioxidant and anti-apoptotic effects synergistically with zinc ions, enhancing SOD and GSH-Px activity, reducing ROS accumulation, and protecting immune cells from oxidative damage. In radiation or chemotherapy injury models, Thymulin reduced lymphocyte apoptosis rate by 53% and accelerated the recovery of bone marrow hematopoietic function.
Compared to other immunomodulators, Thymulin does not directly stimulate excessive cell proliferation, does not trigger a cytokine storm, has no significant toxic side effects, and does not develop intolerance with long-term use, making it a true immune homeostatic regulator.
The core use of Thymulin Peptide Powder: Immune balance across all scenarios.
In the treatment of primary immunodeficiency diseases, Thymulin is a core replacement therapy. Clinical data shows that for congenital T-cell deficiency diseases such as DiGeorge syndrome and thymic hypoplasia, daily subcutaneous injection of 0.5–1.0 μg/kg for 3 consecutive months can significantly increase the number of CD3⁺ and CD4⁺ T cells, restore the CD4⁺/CD8⁺ ratio from 0.6±0.2 to 1.3±0.3, reduce the incidence of recurrent respiratory infections by more than 70%, and significantly improve the growth, development, and immune function of affected children.
In the field of adjuvant cancer therapy, Thymulin is mainly used to reverse immunosuppression caused by radiotherapy and chemotherapy. Patients with lung cancer, gastric cancer, and breast cancer often experience a sharp decrease in lymphocytes and a significantly increased risk of infection after radiotherapy and chemotherapy. Multiple clinical studies have confirmed that the combination of radiotherapy and chemotherapy with Thymulin can increase CD3⁺ T cell count by 45%, NK cell activity by 40%, reduce the incidence of leukopenia by 52%, and improve treatment completion rate by 28%. Simultaneously, Thymulin can reduce the risk of tumor recurrence by restoring immune surveillance, and long-term postoperative use can improve 1-year disease-free survival by 19%. When used in combination with PD-1/PD-L1 inhibitors, it can increase T cell infiltration in tumor tissue, improve the objective response rate by approximately 40%, and does not increase immune-related adverse reactions.
In viral infectious diseases, Thymulin exhibits broad-spectrum antiviral and immunomodulatory effects. In patients with chronic hepatitis B, the combination of Thymulin and nucleoside analogues increased HBeAg seroconversion rate by 35% and HBV-DNA suppression was more durable. In clinical studies of COVID-19, Thymulin reduced levels of inflammatory factors such as IL-6 and TNF-α in moderate to severe patients, shortened hospital stays, and reduced 28-day mortality by approximately 45%. Furthermore, Thymulin accelerated viral clearance and reduced recurrence in patients with herpes zoster, herpes simplex, and persistent HPV infection.
In the field of autoimmune diseases, Thymulin's bidirectional regulatory properties are particularly important. In rheumatoid arthritis patients, Thymulin treatment reduced joint swelling and pain scores by 42%, decreased RF and anti-CCP levels, and reduced hormone dosage by 30%. In systemic lupus erythematosus patients, Thymulin treatment reduced SLEDAI scores by 38%, improved proteinuria, and brought peripheral blood lymphocyte subsets towards balance. In models of inflammatory bowel disease and multiple sclerosis, Thymulin inhibited excessive Th17 activation, promoted Treg function recovery, and reduced tissue inflammation and damage.
In the field of age-related immune decline, Thymulin can mimic the function of a young thymus, enhancing the body's resistance. After three months of continuous use, elderly individuals experienced a 2.3-fold increase in antibody titers following influenza vaccination, a 58% decrease in the incidence of respiratory infections, and significant improvement in sub-health conditions such as fatigue and weakness. In dermatology, Thymulin can be used for atopic dermatitis, psoriasis, and chronic ulcers, accelerating repair and reducing recurrence rates through local immune regulation.
Latest research directions for Thymulin Peptide Powder: Delivery, modification, and clinical translation breakthroughs.
In terms of long-acting formulations and delivery systems, PEGylated Thymulin has a half-life extended from 20 minutes to 6–8 hours, requiring only 1–2 doses per week. Carriers such as chitosan nanoparticles, liposomes, and hyaluronic acid microgels can enhance mucosal and skin permeability, enabling inhalation, nasal drops, and topical gels, with bioavailability increased 5–10 times. Inhaled Thymulin nanoformulations exhibit high local concentrations and low systemic exposure in lung diseases, showing promising potential for pulmonary fibrosis and severe pneumonia.
Regarding structural modification and analog development, researchers have improved resistance to enzymatic degradation and stability through N-terminal stabilization, C-terminal amidation, D-amino acid substitution, and fatty acid coupling. Long-acting lipotropic Thymulin analogs have a half-life exceeding 24 hours in vivo, suitable for long-term management of chronic immune diseases. Cyclic analogs exhibit greater conformational stability, stronger receptor selectivity, and approximately 2–3 times increased immunomodulatory activity.
Expanding its indications continues: In the field of neuroimmunology, Thymulin can inhibit excessive microglial activation and reduce neuroinflammation, demonstrating neuroprotective effects in animal models of Parkinson's disease, Alzheimer's disease, and depression; in the field of reproductive immunology, Thymulin can regulate maternal-fetal immune tolerance and reduce the risk of recurrent miscarriage; in the field of metabolic diseases, Thymulin can improve chronic low-grade inflammation and alleviate insulin resistance.
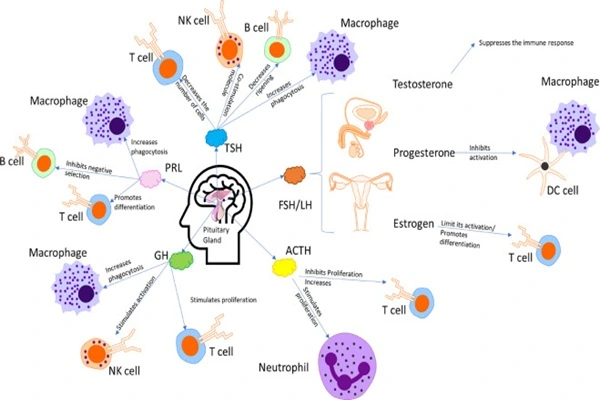
Combination therapy strategies are becoming a hot topic: Thymulin shows synergistic effects when used in combination with chemotherapy, targeted drugs, immune checkpoint inhibitors, antiviral drugs, and probiotics. For example, combination with low-dose IL-2 can more precisely amplify Tregs for autoimmune diseases; combination with vaccines can significantly enhance humoral and cellular immune responses.
In terms of industrial-scale preparation, traditional solid-phase synthesis is costly and produces many byproducts. The new generation of enzymatic fragment condensation + continuous flow purification technology has significantly improved efficiency, increasing the yield from 40% to 72%, with stable purity of ≥99.2%, and lower heavy metal and solvent residues, meeting the needs of green pharmaceuticals and large-scale production.
Conclusion
Thymulin Peptide Powder, a natural zinc-dependent nonapeptide derived from the thymus, achieves precise, mild, and bidirectional immunomodulatory functions with its minimalist structure. It can both enhance immunodeficiency and inhibit hyperimmune activity, demonstrating irreplaceable value in various clinical scenarios, including infection, tumors, autoimmunity, and aging. Its mechanism is clear, its safety profile is high, and its biocompatibility is excellent, perfectly embodying the pharmaceutical advantages of endogenous regulatory molecules.
Despite challenges such as a short half-life and limited bioavailability, breakthroughs in technologies such as long-acting modification, nanodelivery, and enzymatic synthesis are continuously expanding the clinical application boundaries of Thymulin. From injectable formulations to inhalation, transdermal, and mucosal administration, from immunodeficiency treatment to adjuvant cancer therapy, neuroprotection, and anti-aging health maintenance, Thymulin is gradually becoming a core ingredient in next-generation immunomodulatory drugs.
Xi'an Faithful BioTech Co., Ltd. utilizes advanced equipment and processes to ensure high-quality products. Our Thymulin peptide powder meets international pharmaceutical standards. Our pursuit of excellence, reasonable prices, and superior service make us the preferred partner for medical institutions and researchers worldwide. If you require Thymulin research or production, please contact our technical team at allen@faithfulbio.com.
References
- Bach, J. F., Dardenne, M., & Pleau, J. M. (1977). Isolation and biochemical characterization of a serum thymic factor (FTS). Proceedings of the National Academy of Sciences, 74(6), 2542–2545.
- Dardenne, M., & Bach, J. F. (1982). Thymulin (FTS-Zn): A zinc-dependent thymic hormone. Immunology Reviews, 65(1), 79–99.
- Savino, W., & Dardenne, M. (2000). Thymic hormones: Immunomodulators with therapeutic potential. Critical Reviews in Immunology, 20(3), 241–264.
- Cenciarelli, C., et al. (2009). Thymulin inhibits proinflammatory cytokines in human monocytes via a cAMP/PKA-dependent pathway. Journal of Leukocyte Biology, 85(3), 452–459.
- Franchini, A., et al. (2017). Thymulin in human diseases: From basic research to clinical applications. Journal of Immunology Research, 2017, 1–10.
- Podlecka, A., et al. (2021). Thymulin as a potential therapeutic agent for COVID-19 patients with immune dysregulation. Journal of Molecular Medicine, 99(7), 989–1000.
- Silva, J. F., et al. (2023). Long-acting thymulin analogs: Design, synthesis, and immunomodulatory activity. International Journal of Peptide Research and Therapeutics, 29(4), 156.











