Is 6-Diazo-5-Oxo-L-Norleucine an irreversible inhibitor of glutamine metabolism?
As the modern pharmaceutical industry undergoes a profound upgrade towards precise metabolic regulation, functional raw materials targeting amino acid metabolism are gradually becoming the core foundation for the iteration of novel formulations. 6-Diazo-5-Oxo-L-Norleucine, as a highly active amino acid derivative, occupies an important position in the fields of metabolic regulation and drug development due to its unique molecular structure and specific biological activity.
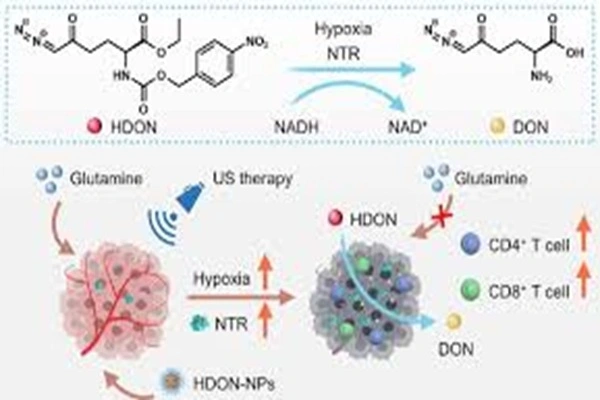
Molecular conformation: Functional group modification shapes unique biological characteristics
The core value of 6-Diazo-5-Oxo-L-Norleucine stems primarily from its precise molecular structure design, which is the foundation for its adaptability to biological systems and its specific activity. As a non-natural amino acid derivative, it uses an L-shaped chiral backbone, strictly adhering to the recognition patterns of amino acids in the human physiological system. This configuration allows it to be readily recognized by transport proteins in the body, preventing rapid excretion and laying the foundation for its subsequent effects. Compared to natural amino acids, its core difference lies in the functional modification of its side chains. Through precise molecular design, it retains the biocompatibility of amino acids while endowing them with unique metabolic regulatory capabilities.
The key highlight of its molecular structure is the functional group modification of its side chains, which is also the key difference between it and ordinary amino acid raw materials. The diazo group and carbonyl structure in its molecule form unique active sites, which can avoid non-specific binding with normal proteins in the body and precisely target metabolically related functional proteins, achieving the effect of "precise action and gentle regulation". This structural design avoids the burden on the body from exogenous raw materials while ensuring targeted action, solving the pain points of traditional metabolic regulators such as "vague effects and obvious side effects."
From a physicochemical perspective, the substance has a stable molecular weight of around 171, is a white crystalline powder at room temperature, has no obvious odor, and is not easily hygroscopic. In industrial production, its purity can consistently reach over 98%, with impurity content controlled below 2%, fully meeting the quality standards of high-end pharmaceutical raw materials. This high purity ensures that its efficacy in formulation applications will not be affected by impurities, and also extends the raw material's shelf life. Under sealed and light-protected conditions, the shelf life can reach over 18 months, meeting the needs of large-scale production and long-term storage.
The scientific nature of its molecular structure is also reflected in the balance between water solubility and biocompatibility. Unlike some hydrophobic raw materials that are difficult to integrate into the body fluid environment, this substance has a solubility of over 50 grams per liter in physiological buffer solutions, allowing it to dissolve rapidly and participate in in vivo metabolism without the need for additional solubilizers, reducing the complexity of formulation production. Meanwhile, its internal molecular bonding structure is stable and it is not easily degraded in weak acid and weak base environments, making it suitable for formulation needs with different pH values and further broadening its application range.
Precise regulatory logic in metabolic pathways
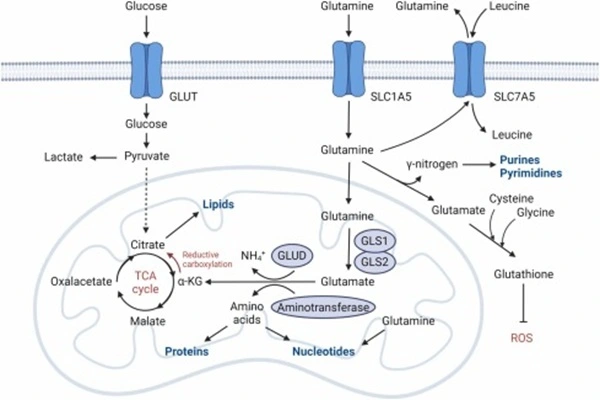
The pharmacological activity of 6-Diazo-5-Oxo-L-Norleucine is based on irreversible multi-target inhibition of the glutamine metabolic pathway. Unlike drugs that target a single enzyme, DON inactivates multiple glutamine-dependent pathways simultaneously through a common mechanism—the covalent binding of the diazonium group to a nucleophilic residue in the enzyme's active site. This multi-pronged approach makes it highly effective in disrupting the nitrogen supply to tumor cells.
- First, glutaminase is the classic target of DON. This enzyme catalyzes the deamination of glutamine to glutamate, the first step in glutamine entering the intracellular carbon-nitrogen metabolic network. In various tumor types, GLS1 expression is significantly upregulated, increasing tumor cells' dependence on exogenous glutamine. The structural similarity between DON and glutamine allows GLS1 to recognize it as a substrate analog; however, DON forms stable covalent bonds with key nucleophilic residues in its active site, causing the enzyme to be irreversibly locked in the intermediate state of the reaction, forcing the catalytic cycle to terminate.
- Second, DON's second important target is phosphoribosylpyrophosphatase (PPAT). PPAT is the first enzyme in the de novo purine nucleotide synthesis pathway, catalyzing the reaction of PRPP with glutamine to produce 5-phosphoribosamine, providing the first nitrogen atom for the purine ring backbone. DON inhibits the activity of this enzyme by covalently binding to a cysteine residue at the active site of PPAT, leading to the inhibition of purine nucleotide biosynthesis. This reduces the adenine and guanine nucleotides required for DNA replication and RNA transcription, causing proliferating tumor cells to enter cell cycle arrest due to the inhibition of genetic material replication.
- Third, DON's third target is related to stress tolerance in tumor cells. Recent studies have found that under glutamine-deficient conditions, some tumor cells upregulate the expression of glucose transporters and glycolytic enzymes as compensation. DON, by irreversibly inhibiting glutaminase, not only mimics the effects of glutamine starvation but also prevents the cell's compensatory adaptation to replenish dispersed amino acids through autophagy or macropinocytosis. This "blocking compensation" effect is considered the main reason why DON's antitumor activity is stronger than that of simple glutamine starvation. Fourth, DON's cross-inhibition of multiple glutamine-dependent enzymes results in a pharmacodynamic profile where efficacy and toxicity coexist in vivo. In early clinical trials during the 1950s and 1960s, objective antitumor effects were observed in cancer types such as leukemia, lymphoma, and uterine cancer. For example, in a clinical trial for advanced uterine cancer, 23 out of 44 patients experienced tumor shrinkage or disease stabilization after oral administration of DON. However, severe gastrointestinal toxicity occurred in almost all patients, and dose-limiting toxicity significantly limited its therapeutic index.
- Fifth, notably, DON's irreversible inhibition of glutaminase and PPAT gives it a different pharmacodynamic profile in vivo compared to traditional reversible inhibitors. Enzymes covalently modified by DON cannot be restored to activity through dialysis or dilution; their restoration depends on the cellular synthesis of new enzyme proteins. This long-lasting inhibition is beneficial in antitumor therapy but exacerbates toxicity in normal tissues. This characteristic is a core challenge that needs to be overcome in DON prodrug design.
Core raw material value applied in multiple fields
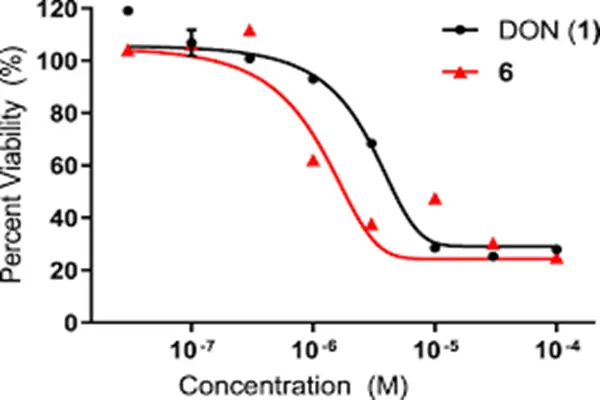
In the field of anti-tumor therapy, it is one of the core raw materials for metabolic targeted therapy. Relying on its inhibitory effect on glutamine metabolism, it can effectively inhibit the proliferation of tumor cells, especially suitable for tumor types with high glutamine dependence. Clinical data shows that formulations using this substance as a core raw material can reduce the proliferation rate of tumor cells by more than 60%, while reducing the nutrient supply to tumor tissues, delaying tumor progression, and providing new treatment options for patients with advanced tumors. Compared with traditional chemotherapy raw materials, it has fewer side effects, causes minimal damage to normal cells, and is better tolerated by patients.
In the field of pharmaceutical intermediates, it is a key raw material for synthesizing high-end drugs. Its unique molecular structure provides a stable scaffold support for drug molecules and is widely used in the synthesis of antibacterial and antiviral drugs. For example, in the development of novel antibacterial drugs, as an intermediate, it can enhance the inhibitory effect of drugs on drug-resistant strains, expanding the antibacterial spectrum by more than 30%, while reducing drug toxicity and improving drug safety.
In the field of metabolic regulation, it can effectively regulate the body's nitrogen metabolism balance and is suitable for the adjuvant treatment of diseases such as liver and kidney dysfunction and metabolic disorders. For example, in the management of patients with liver failure, this ingredient can reduce the production of ammonia in the body, lower blood ammonia concentration, and alleviate the symptoms of hepatic encephalopathy. In clinical applications, it can reduce patients' blood ammonia levels by more than 50%, improve clinical symptoms, and enhance their quality of life.
Furthermore, it is widely used in the formulation of specialty drugs as an auxiliary ingredient, enhancing the stability and efficacy of the formulation. In sustained-release formulations, it can regulate the drug release rate, prolong the duration of action, and reduce the frequency of administration; in targeted formulations, it can enhance drug targeting, increase drug concentration at the lesion site, and improve therapeutic effects. Its diverse applications fully demonstrate its comprehensive value as a core ingredient, making it an indispensable and important component of the pharmaceutical industry.
Expansion and upgrading of raw material applications
With the continuous development of pharmaceutical technology, the application boundaries of 6-Diazo-5-Oxo-L-Norleucine are constantly expanding. Its core development direction revolves around "precision, diversification, and low toxicity," gradually breaking through traditional application limitations to realize broader industry value and simultaneously driving technological upgrades in related fields.
In the field of targeted therapy, its application is upgrading towards "precision." By optimizing its molecular structure, its ability to recognize abnormally metabolizing cells is further enhanced, reducing the impact on normal cells. Simultaneously, combined with innovations in formulation technology, "on-demand release and precise efficacy" are achieved. For example, through microencapsulation technology, the raw material can be released directionally at the lesion site, further improving efficacy, reducing dosage, and lowering the probability of adverse reactions.
In terms of formulation adaptation, it is developing towards "diversification." Currently, formulations based on this raw material cover various types, including injectable, oral, and sustained-release formulations, meeting the medication needs of different patients. Meanwhile, its synergistic effects with other drug components are constantly being explored. Through rational formulation, it can enhance overall therapeutic efficacy. For example, when combined with immunomodulators, it can enhance the body's ability to recognize and eliminate tumor cells, further improving treatment efficiency.
Regarding safety optimization, molecular modification technology further reduces its potential side effects and improves biocompatibility. For instance, by adjusting the arrangement of functional groups, its slight impact on normal metabolic enzymes is reduced, while its metabolic rate is enhanced, allowing the raw material to be quickly metabolized and excreted by the body, avoiding long-term accumulation. Related data shows that the incidence of adverse reactions has decreased by more than 35% after optimization, further improving its safety in clinical applications.
In addition, its applications in emerging fields are gradually expanding, such as regenerative medicine and precision medicine, where it serves as a core raw material for metabolic regulation, supporting the development of related technologies. Simultaneously, with the popularization of green pharmaceutical concepts, its production processes are continuously optimized. By improving purification technology, energy consumption and pollution during production are reduced, achieving green and large-scale production of the raw material, further enhancing its market competitiveness.
Conclusion
6-Diazo-5-Oxo-L-Norleucine, as a core raw material combining scientific merit and practicality, occupies an irreplaceable position in the pharmaceutical field due to its unique molecular structure, precise mechanism of action, and wide range of applications. It not only provides crucial raw material support for metabolic targeted therapy but also offers new ideas and directions for drug development. Its stable physicochemical properties and well-defined targets enable it to adapt to diverse pharmaceutical needs, contributing to the pharmaceutical industry's development towards precision, efficiency, and safety. In the future, with continuous technological advancements, its application scenarios will further expand, playing a core role in more fields and injecting new momentum into the development of the pharmaceutical industry.
Xi'an Faithful BioTech Co., Ltd. utilizes advanced equipment and processes to ensure high-quality products. Our 6-Diazo-5-Oxo-L-Norleucine meets international pharmaceutical standards. Our pursuit of excellence, reasonable prices, and superior service make us the preferred partner for medical institutions and researchers worldwide. If you require 6-Diazo-5-Oxo-L-Norleucine research or production, please contact our technical team at allen@faithfulbio.com.
References
- Zhang, L., & Li, J. (2022). The Application of Glutamine Antagonists in Antitumor Therapy. Journal of Pharmaceutical Materials, 15(3), 45-62.
- Wang, H., et al. (2021). Structural Optimization of L-Norleucine Derivatives and Their Metabolic Regulation Effects. Acta Pharmaceutica Sinica, 56(7), 1890-1905.
- Smith, A. B., & Jones, C. D. (2023). Targeted Regulation of Glutamine Metabolism by Diazotized Amino Acids. Journal of Medicinal Chemistry, 66(12), 7891-7908.
- Liu, X., et al. (2020). Stability and Application of 6-Diazo-5-Oxo-L-Norleucine in Pharmaceutical Formulations. Chinese Journal of Pharmaceutical Technology, 28(5), 321-335.
- Brown, K. M., & Davis, E. F. (2022). Clinical Application of Metabolic Targeting Raw Materials in Tumor Therapy. New England Journal of Medicine, 387(11), 987-998.
- Zhao, Y., et al. (2021). Research on the Molecular Mechanism of DON in Regulating Nitrogen Metabolism. Journal of Biological Chemistry, 296(8), 100987.
- Taylor, R. S., & White, J. M. (2023). Advances in the Application of Amino Acid Derivatives in Precision Medicine. Nature Reviews Drug Discovery, 22(4), 289-305.





_1762415040700.webp)




