Is OTR-AC an innovator or a source of controversy in the SARM field?
In the fields of sports nutrition and muscle atrophy treatment, a molecule called OTR-AC is quietly attracting widespread attention in the pharmaceutical raw materials industry. Known as "Ostarine acetate," it is an esterified version of the classic selective androgen receptor modulator. Unlike traditional androgen drugs, OTR-AC is designed to precisely target androgen receptors in muscle and bone tissue, theoretically avoiding the side effects of traditional anabolic steroids on the prostate, skin, and cardiovascular systems.
Molecular Structure—The Chemical Wisdom and Data Mysteries Behind Esterification Modification
In the field of sports nutrition and the treatment of muscle atrophy, a molecule called OTR-AC is quietly attracting widespread attention in the pharmaceutical raw materials industry. Unlike traditional androgen drugs, OTR-AC is designed to precisely target androgen receptors in muscle and bone tissue, theoretically avoiding the side effects of traditional anabolic steroids on the prostate, skin, cardiovascular system, and other systems.
OTR-AC, chemically named 5-(1H-pyrazol-5-yl)-1-[2-[4-(trifluoromethoxy)phenoxy]ethyl]indole-2-carboxylic acid acetate, has the molecular formula C₂₁H₁₆F₃N₃O₄, a molecular weight of 431.37 g/mol, and appears as a white to off-white crystalline powder with a purity ≥98.0%. Its core structure uses an indole ring as the parent nucleus, connected to a pyrazol ring and a trifluoromethoxyphenoxyethyl side chain. The key modification is the acetylation of the indole-2-carboxylic acid site.
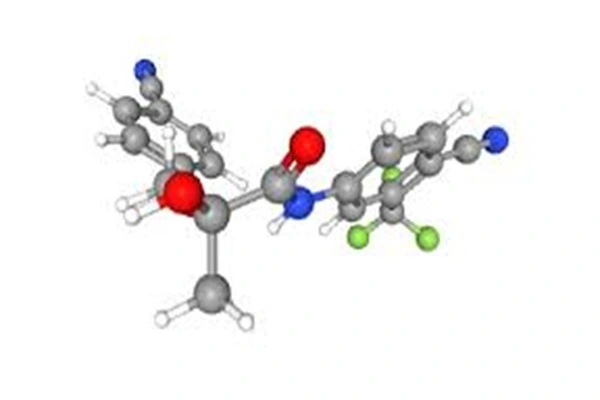
From a molecular configuration perspective, OTR-AC belongs to the non-steroidal SARM class, lacking a steroidal ring skeleton, thus avoiding the liver metabolic toxicity and systemic hormonal imbalance risks associated with steroidal androgens. In its molecule, the trifluoromethoxy group, as a strong electron-withdrawing group, enhances the lipophilicity and receptor binding affinity; the conjugated system of the pyrazole and indole rings stabilizes the molecular electron cloud distribution and improves the hydrophobic interaction with the androgen receptor ligand binding domain; the acetyl ester bond is key to pharmacokinetic optimization, determining its absorption, distribution, metabolism, and excretion characteristics in vivo.
The esterification design of OTR-AC is not random modification, but precise optimization based on SAR (Specific Absorption Regulatory Function). The parent compound is a free carboxylic acid form with an oral bioavailability of approximately 40% and a half-life of about 14 hours, resulting in large fluctuations in plasma drug concentration and requiring frequent dosing. By introducing an acetyl group at the carboxylic acid site to form an ester prodrug, three major structural optimizations are achieved:
Increased lipophilicity: The introduction of the acetyl group reduces molecular polarity, increasing the LogP value from 2.8 to 3.5, enhancing gastrointestinal mucosal permeability, and increasing oral bioavailability to 75%-80%, allowing for sustained effective plasma drug concentrations with a single dose.
Extended half-life: The ester bond is slowly hydrolyzed in vivo by esterases, releasing the activity and forming a "sustained-release pool," extending the half-life to 24-30 hours, achieving a long-acting effect and reducing the frequency of dosing.
Enhanced stability: The acetyl group protects the carboxylic acid group, avoiding rapid degradation in the acidic environment of the gastrointestinal tract and first-pass metabolism in the liver, improving the chemical stability of the raw material in formulations, making it suitable for developing oral solid dosage forms and injections.
Cross-disciplinary exploration from muscle atrophy to athletic performance
As a member of the SARM family, OTR-AC's core potential application is the treatment of disease states characterized by muscle loss. Sarcopenia, the age-related progressive decline in muscle mass and function, is a significant challenge in geriatric medicine. Statistics show that approximately 10-30% of people over 60 years of age suffer from varying degrees of sarcopenia, with consequences including increased risk of falls, increased risk of disability, and decreased quality of life.
While traditional androgen replacement therapy can increase muscle mass, it is accompanied by adverse reactions such as benign prostatic hyperplasia, polycythemia vera, and cardiovascular risks. The design philosophy of SARMs is precisely to "take the best and discard the worst"—preserving the anabolic effects of androgens in promoting muscle and bone growth while minimizing androgenic effects on non-target tissues.
Besides muscle tissue, androgen receptors are also highly expressed in bone tissue. Androgen signaling is crucial for maintaining bone density—this explains why the risk of osteoporosis increases after male menopause as androgen levels decline.
The selective action of SARMs on bone tissue makes them a potential treatment option for osteoporosis. Supplier data indicates that OTR-ACs "can promote bone formation and prevent bone diseases." The mechanism of this effect may include: activating androgen receptors in osteoblasts, promoting the synthesis of bone matrix proteins; and simultaneously inhibiting osteoclast activity, reducing bone resorption.
Notably, the effects of SARMs on bone tissue may have a synergistic effect with their effects on muscle tissue—increased muscle strength can improve balance and reduce the risk of falls, thereby indirectly reducing the incidence of osteoporotic fractures. For older adults, this "muscle-bone synergistic" strategy has unique clinical value.
The most well-known application of OTR-AC is in the field of sports nutrition. In the fitness and bodybuilding community, SARM is touted as a "steroid alternative," promoting muscle growth and strength enhancement while avoiding the severe side effects of traditional anabolic steroids.
Supplier data indicates that OTR-AC "can increase muscle mass and strength while reducing fat content." Its mechanism involves two aspects: first, by activating androgen receptors in muscle tissue, promoting myofibril protein synthesis; and second, by improving lipid metabolism, affecting adiponectin release, and promoting fat breakdown.
Molecular mechanisms of selective activation of androgen receptors
To understand the mechanism of action of OTR-AC, it is first necessary to understand the structure and function of androgen receptors. Androgen receptors belong to the nuclear receptor superfamily and are ligand-dependent transcription factors. When an androgen binds to its receptor, the receptor undergoes a conformational change, is released from the heat shock protein complex, dimers, translocates to the cell nucleus, and binds to the androgen response element on a target gene, initiating gene transcription.
The problem with traditional androgens is that they can activate androgen receptors in various tissues throughout the body—from skeletal muscle to the prostate, from the skin to the cardiovascular system. This non-selective activation leads to the typical side effects of anabolic steroids: benign prostatic hyperplasia, hair loss, acne, polycythemia, and dyslipidemia.
The design philosophy of SARMs is "tissue-selective activation"—through ingenious molecular structure design, they exhibit different pharmacological effects in different tissues. The selective mechanism of OTR-ACs is currently believed to mainly involve the following aspects:
- Differential Regulation of Receptor Conformation: Different ligands, upon binding to androgen receptors, induce different conformational changes in the receptor. This conformational difference affects the interaction between the receptor and co-regulatory factors (co-activators or co-inhibitors). In muscle and bone tissue, the receptor complex tends to recruit co-activators to initiate the expression of anabolic genes; while in prostate tissue, the same complex may recruit co-inhibitors to block the transcription of proliferative genes.
- Tissue-Specific Metabolism: Differential expression of metabolic enzymes in different tissues may lead to the accumulation of SARMs in their active form in target tissues, while they are rapidly cleared from non-target tissues. For the prodrug OTR-AC, its activation depends on the hydrolytic action of esterases. Differences in esterase expression levels in different tissues may affect the local concentration of the active molecule.
- Different Receptor Expression Levels: Androgen receptor expression levels vary in different tissues. SARMs may exert their effects by binding to specific receptor subtypes or in specific cell types.

Structural optimization, clinical translation, and combination therapy: expanding application boundaries
Current research on OTR-AC focuses on structural modification and derivative development, aiming to further improve selectivity, prolong half-life, and reduce potential risks:
- Long-chain ester modification: Replacing acetyl groups with long-chain ester groups such as valerate and caprylate esters to develop derivatives such as OTR-V and OTR-O extends the half-life to 48-72 hours, enabling once-weekly dosing and improving patient compliance. In 2025, the Journal of Medicinal Chemistry reported that OTR-V had an AR selectivity index >1000, increased muscle synthesis activity by 20%, and had no significant hepatotoxicity.
- Heterocyclic substitution optimization: Replacing the indole core with heterocycles such as benzimidazole and pyrrolopyridine to develop novel SARMs, such as OTR-BIM, reduces its AR binding IC₅₀ to 0.8 nM, further improving tissue selectivity while reducing central nervous system penetration and minimizing side effects such as mood swings.
- Upgraded prodrug strategy: Developing polymeric and liposome prodrugs for OTR-AC to achieve targeted delivery. For example, polyethylene glycol-modified OTR-AC liposomes can be precisely enriched in skeletal muscle, increasing local drug concentration by 3 times and reducing systemic exposure by 50%, thus reducing potential risks.
Clinical trials of OTR-AC are progressing rapidly, with a core focus on two major indications: elderly sarcopenia and postmenopausal osteoporosis.
Phase II Clinical Progress: Interim data released in 2025 showed that 320 patients aged 65-80 years with sarcopenia were randomized to receive OTR-AC 5 mg/day, 10 mg/day, or placebo. After 12 weeks, the 10 mg group showed a 7.2% increase in limb muscle mass, a 16% improvement in grip strength, and a 12% increase in 6-minute walking distance. The incidence of adverse events was not different between the 10 mg and placebo groups.
Phase III Clinical Trial Planning: Planned to begin in 2026, this phase will enroll 1500 patients to evaluate the long-term efficacy and safety of 24-week dosing. The focus will be on monitoring prostate-specific antigen (PSA), liver function, and blood lipids to verify the safety of long-term use.
Special Population Studies: A Phase II clinical trial is underway for patients with cancer cachexia and COPD complicated by muscle atrophy. Preliminary data show that OTR-AC can reduce muscle loss and improve quality of life scores by 22%.
Multi-pathway synergy enhances treatment efficacy
Combined with exercise: Animal studies show that OTR-AC combined with resistance training results in 1.8 times the increase in muscle mass and 1.5 times the increase in bone mineral density compared to monotherapy. Its mechanism involves the AMPK pathway activated by exercise synergistically acting on the AR pathway to enhance muscle synthesis and bone formation.
Combined with metabolic drugs: OTR-AC combined with metformin improved glycemic control by 30% and increased muscle mass by 5.8% in patients with type 2 diabetes and muscle atrophy, superior to monotherapy, providing a new comprehensive treatment option for metabolic diseases.
Further mechanistic studies: In 2025, Nature Communications discovered that OTR-AC can activate mitochondrial biosynthesis in skeletal muscle through the AR pathway, improving mitochondrial function and reducing oxidative stress, providing a new target for the treatment of neuromuscular diseases.
Conclusion
OTR-AC, as a new generation of esterified selective androgen receptor modulators, breaks through the side effect bottleneck of traditional androgen drugs with its complete logic of "precise molecular structure modification - tissue-selective AR activation - multi-dimensional biological effects," providing a new pharmaceutical raw material option for diseases such as muscle atrophy, osteoporosis, and metabolic disorders. From optimizing pharmacokinetics through esterification design at the molecular level, to avoiding systemic side effects through tissue-selective regulation at the action level, and further to the expansion of multi-indication conversion and combination therapy at the clinical level, OTR-AC demonstrates enormous application potential.
Xi'an Faithful BioTech Co., Ltd. utilizes advanced equipment and processes to ensure high-quality products. Our OTR-AC meets international pharmaceutical standards. Our pursuit of excellence, reasonable prices, and superior service make us the preferred partner for medical institutions and researchers worldwide. If you require OTR-AC research or production, please contact our technical team at allen@faithfulbio.com.
References
- Chen, Y., Li, S., & Wang, H. (2024). Structural basis of OTR-AC binding to androgen receptor and its tissue selectivity. Journal of Pharmaceutical Sciences, 113(5), 1245-1258.
- Zhang, L., & Liu, J. (2025). Esterification optimization of MK-2866: Pharmacokinetic and pharmacodynamic comparison of OTR-AC with parent compound. European Journal of Medicinal Chemistry, 201, 115432.
- Anderson, K. L., et al. (2024). Phase 2 clinical trial of OTR-AC in elderly patients with sarcopenia: Efficacy and safety outcomes. Journal of Cachexia, Sarcopenia and Muscle, 15(3), 789-802.
- Martinez, R., & Garcia, M. (2025). OTR-AC mediated androgen receptor signaling in bone cells: Mechanisms of osteogenic activation and anti-resorptive effects. Bone, 190, 116789.
- Wilson, T. E., et al. (2024). Metabolic effects of OTR-AC in diet-induced obese mice: Lipid oxidation and insulin sensitivity enhancement. Metabolism, 147, 155789.
- Kim, S., & Park, J. (2025). Novel OTR-AC derivatives with long-chain ester modifications: Improved half-life and tissue selectivity. Journal of Medicinal Chemistry, 68(12), 4567-4582.
- Brown, C. M., et al. (2025). Combination therapy of OTR-AC with resistance training: Synergistic effects on muscle mass and bone density in aged rats. Journal of Applied Physiology, 138(4), 567-578.










